KATERYNA KON / SCIENCE PHOTO LIBRARY
The first long-acting injectable treatment for HIV infection in adults has been provisionally recommended by the National Institute for Health and Care Excellence (NICE).
In draft guidance, published on 18 November 2021, cabotegravir (Vocabria; ViiV Healthcare) with rilpivirine (Rekambys; Janssen) is recommended by NICE, within its marketing authorisation, as an option for adults with HIV-1, the most common type of HIV infection.
Eligible patients include those who are already taking antiretroviral medicines that have kept the virus stable, at a low level (HIV-1 RNA fewer than 50 copies/mL of blood) and where there is no evidence of viral resistance or a previous failure of other anti-HIV-1 medicines.
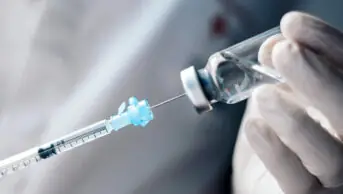
Current treatment for HIV-1 is a lifelong regimen of antiretroviral tablets each day, but both cabotegravir and rilpivirine are administered as two separate intramuscular injections every two months, after an initial oral tablet lead-in period.
Clinical trial results have shown that the two injections are as effective as oral antiretrovirals at keeping the viral load lower than 50 copies/mL of blood. The aim of HIV treatment is to keep the number of virus particles in the blood low enough that the virus cannot be detected or transmitted between people.
Meindert Boysen, deputy chief executive and director of the Centre for Health Technology Assessment at NICE, said: “Despite scientific advances, HIV is still incurable, but the virus can be controlled by modern treatment.
“However, for some people, having to take daily multi-tablet regimens can be difficult because of drug-related side effects, toxicity, and other psychosocial issues, such as stigma or changes in lifestyle.”
Boysen added that the guideline committee had heard that stigma remained an issue for people living with HIV and could have a negative impact on people’s health and relationships.
“We’re pleased therefore to be able to recommend cabotegravir with rilpivirine as a valuable treatment option for people who already have good levels of adherence to daily tablets, but who might prefer an injectable regimen with less frequent dosing,” he said.
It is estimated that around 13,000 people will be eligible for treatment with cabotegravir and rilpivirine in England.
Stephanie Tyler, a specialist pharmacist in HIV, sexual and reproductive health at Guy’s and St Thomas’ NHS Foundation Trust, said that her team was able to offer cabotegravir and rilpivirine through the compassionate use access scheme at the trust.
“The experience has been extremely positive, people have tolerated it well with minimal side effects, mostly injection site reactions, as to be expected with an intramuscular injection,” she said, adding that NICE has yet to reveal the eligibility criteria for the injectables.
“This will also be restricted by the capacity clinics have to administer these injections every two months.”
She said that “it is an exciting new formulation of [antiretroviral therapy] which I hope will change the lives of many people living with HIV and improve the quality of life of people living with HIV”.
Tyler explained that the recommendations would help work towards a target set by researchers in 2016 that would see 90% of people living with HIV have a good quality of life.
“The absence of having to take a tablet every day will help reduce the reminder of having the condition,” she said.
The final guidance is expected to be published on 5 January 2022.