Photofusion / Rex Features
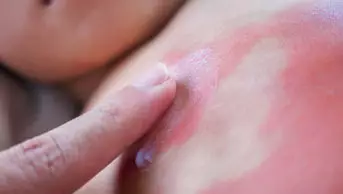
Pharmacists should routinely check a patient’s drug allergy status before they dispense a prescription, according to the first ever guideline on managing drug allergy from the National Institute for Health and Care Excellence (NICE), published on 3 September 2014.
Prescription forms should also be redesigned to include information on an individual’s drug allergy status and the drugs or drug classes to avoid, it recommends.
Whenever a person presents with a suspected drug allergy, a detailed record should be taken to include the generic and brand names of the drug, its strength and formulation and which drugs, or drug classes, should be avoided in future, according to the guideline.
NICE sets out the signs to look for when a drug allergy is suspected as well as the typical time it takes for symptoms to appear.
It details the steps to take when treating patients who are allergic to non-selective non-steroidal anti-inflammatory drugs and in what circumstances cyclooxygenase-2 inhibitors should be offered instead. NICE also makes recommendations about when patients with a suspected allergy should be referred to specialist services.
“The guideline stresses the care all healthcare professionals must take when documenting new drug allergies and the importance of sharing this information with patients and other healthcare professionals,” said Shuaib Nasser, a consultant in allergy at Addenbrooke’s Hospital, Cambridge.
“People should be provided with structured written information on drugs to avoid and be advised to check with their pharmacist before taking over-the-counter medicines,” added Nasser, the chairman of the guideline development group.
The guideline, which was out for public consultation earlier in 2014, comes as the number of recorded patient safety incidents involving a drug allergy continues to rise, and around 80% of cases occur in patients with a known drug allergy.
Data from 1996 to 2000 show that drug allergies and adverse drug reactions accounted for approximately 62,000 annual hospital admissions in England.
Between 1998 and 2005, serious adverse drug reactions rose 2.6-fold, according to NICE. Up to 15% of inpatients have their hospital stay prolonged as a result of an adverse drug reaction, it says.