Sergio Monti/Alamy Stock Photo
The National Institute for Health and Care Excellence (NICE) has recommended a monoclonal antibody treatment for generalised myasthenia gravis (gMG), an auto-immune condition, for use on the NHS in England in eligible adults.
In final draft guidance published on 29 April 2026, NICE approved rozanolixizumab (Rystiggo; UCB Pharma) for adults with gMG who tested positive for acetylcholine receptor or muscle-specific kinase antibodies and who still have poorly controlled symptoms, even after trying standard treatments.
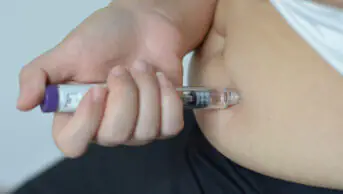
Rozanolixizumab can be self-administered at home via a short subcutaneous infusion following training by a healthcare professional, the draft guidance said.
gMG is a long-term condition that causes certain muscles to become weak and tire easily, caused by a problem with the immune system. In a statement published alongside the final draft guidance, NICE said that around 800 adults in England could benefit from the new treatment.
Patients who take rozanolixizumab will not have to attend hospital for several days of treatments, such as IV immunoglobulin (IVIg) or plasma exchange (PLEX), as they would have previously, NICE added.
In the past, treatment options have included steroids and immunosuppression, but the new treatment option will be more targeted, which reduces steroid use and side effects, as well as offers the possibility of faster-acting symptom control for patients who do not respond to other treatments, the draft guidance said.
Charlotte Carpenter, research and partnerships manager at patient charity Myaware, said the condition can cause muscle weakness, which affects movement, the ability to smile and the ability to breathe, chew and swallow.
“We know there are a lot of problems with long-term and high-dose steroids used on patients and the impact that can have on their day-to-day life,” she said.
“Myasthenia can affect so many facets of a patient’s life. Depending on age of diagnosis, it can affect careers, social, financial stability, emotional and mental health. For patients who are able to control these symptoms through this new medication, it gives them an entirely new quality of life.”
In 2023, the results of a phase III study on rozanolixizumab revealed that patients who took the drug reported greater reductions in their myasthenia gravis activities of daily living (MG-ADL) score than those in the placebo group. The most common report side effects included headache, diarrhoea and pyrexia.
“An improvement to minimal symptom expression in the MG-ADL score can mean anything from patients being able to go back to driving, being able to go back to work, not being at constant risk of what we call a myasthenia crisis, which is when the muscle weakness affects the breathing and can result in patients being emergency admitted and having to go through mechanical intubation, [which] can be deadly for some patients”, Carpenter said.
Esther Wong, lead pharmacist for neurology at Chelsea and Westminster NHS Foundation Trust, told The Pharmaceutical Journal that the recommendation was “excellent news”.
“This represents the growing number of treatment options within gMG in advancing patient care. Where traditional therapies have failed, [this] expands [clinicians’] armamentarium, allowing for a more tailored and effective treatment approach for this group of patients,” she said.
The treatment was first submitted to NICE for consideration in 2024, but rozanolixizumab was not recommended owing to uncertainty over its long-term effect and cost-effectiveness estimates.
NICE said that its recommendation followed “agreement on an improved commercial deal between NHS England and the company”.
Final draft guidance is expected in May 2026, and the treatment will be available on the NHS in England straight away through interim funding from the Innovative Medicines Fund.