Shutterstock.com
Switching from branded to generic antiepileptic drug (AED) products can significantly lower the costs of treatment. However, there has been widespread scepticism about the bioequivalence of generic products and the safety of generic-to-generic switching.
US researchers report in The
Lancet Neurology (online, 11 February 2016)[1]
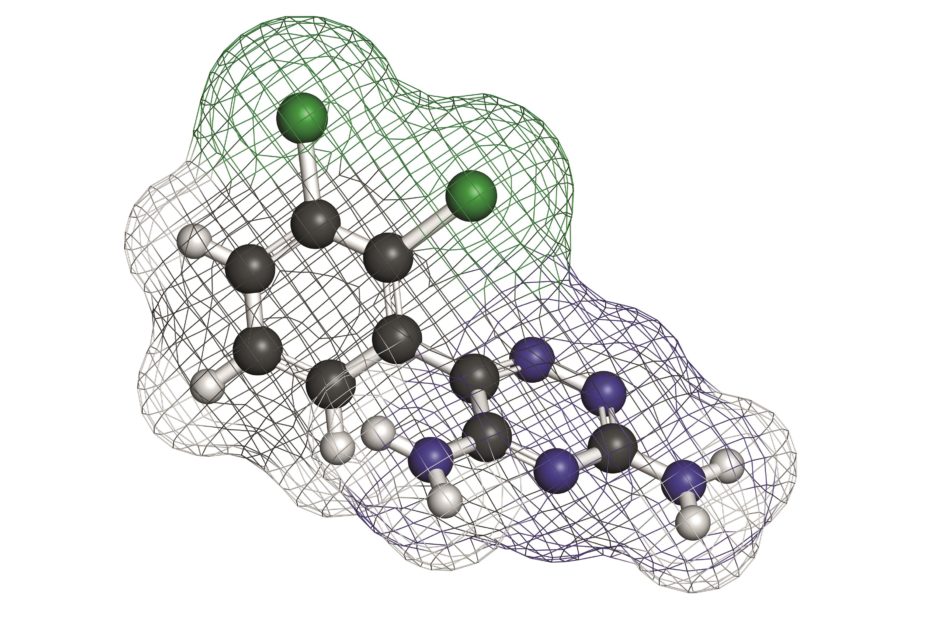
on a randomised study in which 35 patients alternated between the two most different generic lamotrigine products for four cycles of 14 days.
They found that pharmacokinetic measures of the drugs were equivalent and there were no significant changes in seizure frequency or adverse events between the two.
The team says the findings could prompt pharmaceutical bodies to reassess their opinions on generic substitution. At present in the UK, the Medicines and Healthcare products Regulatory Agency divides AEDs into three risk categories to help prescribers decide on product selection for patients.
References
[1] Privitera MD, Welty TE, Gidal BR et al. Generic-to-generic lamotrigine switches in people with epilepsy: the randomised controlled EQUIGEN trial. The Lancet Neurology 2016. doi: 10.1016/S1474-4422(16)00014-4