Imperial College
Researchers have developed a bioassay that for the first time combines cells from a single donor’s blood to predict whether a new drug will cause a severe immune reaction in humans, according to an article in The
FASEB Journal
[1]
.
The assay is designed to avoid catastrophes, such as the 2006 trial of the drug TGN1412, which caused six healthy male volunteers to be admitted to intensive care with multiple organ failure. The men, who received the experimental drug at Northwick Park Hospital, experienced a profound cytokine response known as a cytokine storm, an immune reaction which is potentially fatal.
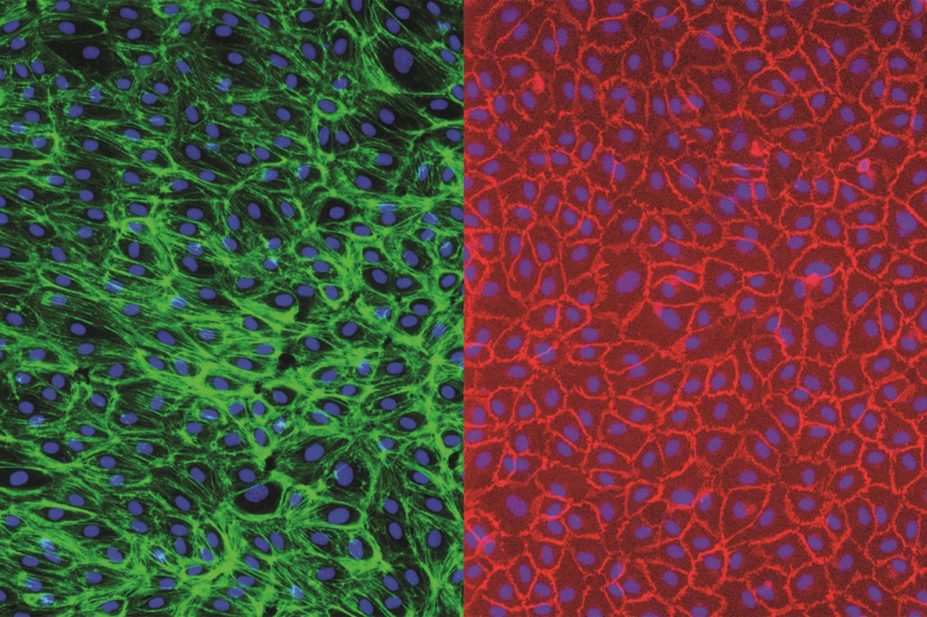
The research team, led by Jane Mitchell from the National Heart and Lung Institute at Imperial College London, used donor stem cells in the peripheral blood to create endothelial cells and combined these with white blood cells from the same donor. With this co-culture, they thus created a bioassay that responded by releasing a large amount of cytokines when TGN1412 was introduced.

Source: Imperial College London
The research team was led by Jane Mitchell, pictured with Daniel Reed, from the National Heart and Lung Institute at Imperial College London
“This is the first time that anyone has made in a dish a test using endothelial cells and white blood cells from the same donor,” says Mitchell, and she adds that the team has been developing the test for three years. “We’ve only been able to do so because of advances in stem cell research. Previously, there wasn’t any way to get enough endothelial cells from a person unless they were removed in surgery or the person was dead. Now we can grow endothelial cells from blood cells.”
Biological therapies or “biologics” account for around a third of medicines in the pharmaceutical pipeline, according to the researchers. Because these compounds contain biological material such as antibodies that are specific to humans, they can cause serious reactions that cannot be detected in animal studies.
There are currently three different approaches that scientists use to detect these cytokine storm reactions. In the first, small amounts of human blood are mixed with drug antibodies, incubated overnight and the cytokines measured afterwards. But as there are no endothelial cells used in this approach, it cannot accurately mimic what happens in human blood vessels. “It´s not very sensitive and not very discriminatory,” Mitchell says. “You get a number of false positives and might miss a drug that will cause problems.”
With the second approach, biologic drugs are air-dried to plastic and presented to monocultures of immune cells. The main drawback with this system, Mitchell points out, is its artificiality, and the fact that it fails on some counts to differentiate effectively between a very harmful drug such as TGN1412 and drugs that are currently used in clinics.
The third approach uses endothelial cells taken, for example, from umbilical veins and then adds them to white blood cells from a different donor, which can therefore result in mismatched tissues.
“The assay we have developed separates those drugs that are very toxic from those that are more mildly toxic,” says Daniel Reed, one of the study authors. The study found that the assay was able to distinguish between alemtuzumab, an anti-CD52 monoclonal antibody that can cause a mild cytokine storm in some patients, and bevacizumab, ofatumumab and trastuzumab, biologics that do not cause cytokine release in most patients.
The researchers acknowledge that a drug might cause a cytokine storm response by acting on other specialised cells, such as those in the brain. Mitchell adds that, as stem cell technology advances, the same approach could be taken with combinations of any specialised cells with white cells from the same donor.
The Imperial researchers are working with clinical trials company Quintiles to develop the technology further, and believe the new test could be available in prototype “off-the-shelf” kits within a year.