Pleiotrope / Wikimedia Commons
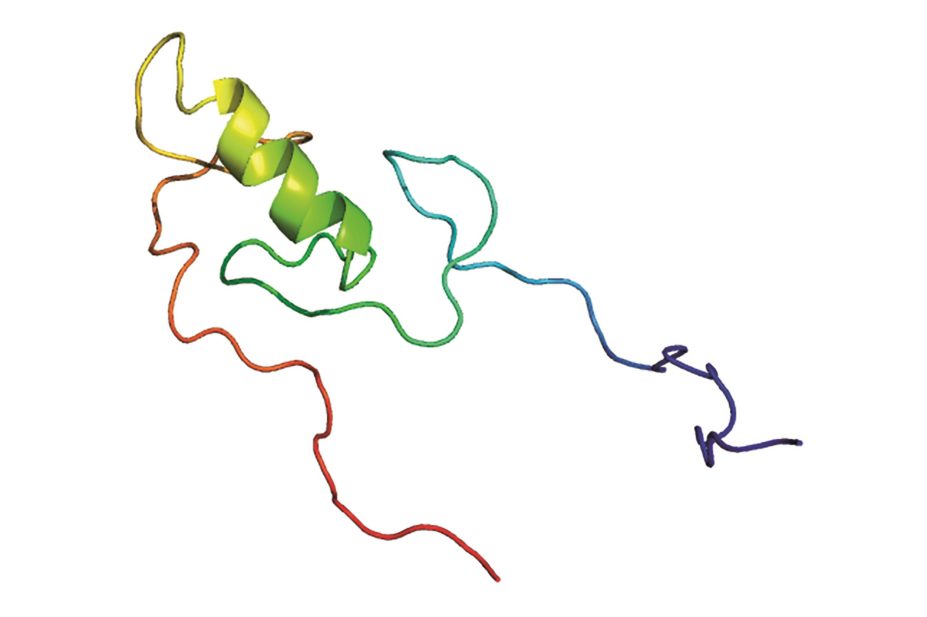
A protein found in rhesus monkeys is known to confer resistance to HIV infection, but the exact mechanism was unclear. Researchers have now shed light on how the TRIM5α protein targets the HIV-1 retrovirus and triggers its degradation and activates the innate immune system pathways.
The researchers, who report in Heliyon (online, 14 January 2016)[1]
, worked with human cells expressing the TRIM5α protein and found that it contains a motif that allows it to bind a family of proteins known as SUMO. When this binding motif was mutated, TRIM5α lost its ability to degrade HIV and stimulate the innate immunity pathway.
The team say the findings could have applications in gene therapy, for which the TRIM5α gene is a candidate.
References
[1] Nepveu-Traversy M-E, Demogines A, Fricke T, et al. A putative SUMO interacting motif in the B30.2/SPRY domain of rhesus macaque TRIM5α important for NF-κB/AP-1 signaling and HIV-1 restriction. Heliyon 2016;2:e00056. doi: 10.1016/j.heliyon.2015.e00056.