DR P. MARAZZI / SCIENCE PHOTO LIBRARY
Researchers have recommended reclassifying first-generation sedating antihistamines as a prescription-only medicine (POM), citing an increase in the number of deaths involving these drugs.
A team of researchers at King’s College London studied drug-related deaths reported to the National Programme on Substance Abuse Deaths (NPSAD) in England between 1999 and 2019.
They identified 1,666 antihistamine detections at post-mortem from 1,537 cases reported to NPSAD in that time, with pharmacy-only (P) sedating antihistamines detected in 85% of these cases.
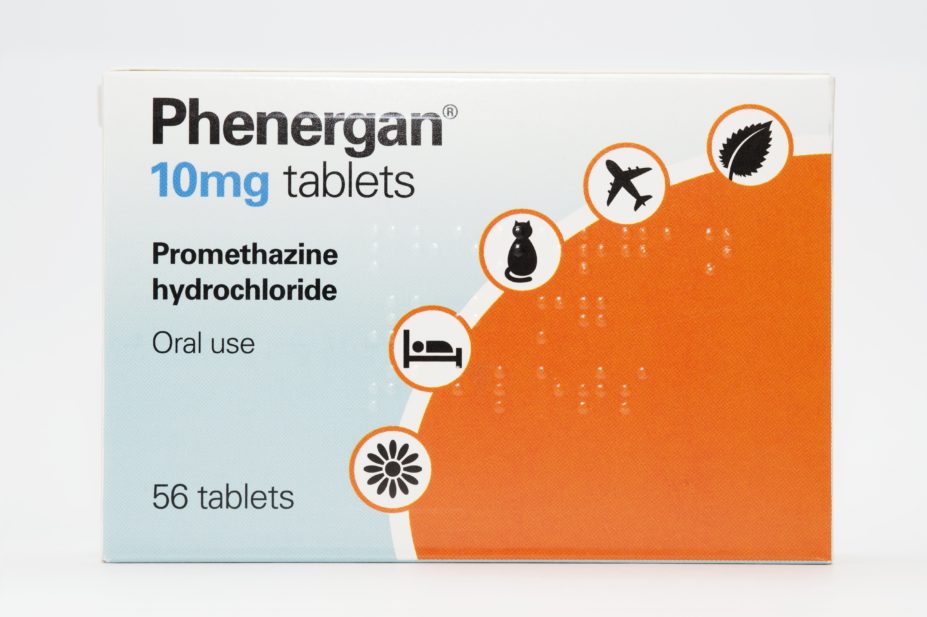
The proportion of deaths where first-generation antihistamines — those that cross the blood-brain barrier and have sedating properties — were detected has “significantly increased” over the study period, with the most notable increase relating to promethazine.
The authors said the findings, published in the British Journal of Clinical Pharmacology, comprise the first report regarding antihistamine-related mortality in England in more than 40 years.
They added that, in 95% of the cases, the drugs had been taken alongside another central nervous system depressant — a figure that the authors described as “astounding”, and potentially evidence that opioid users are specifically seeking the effects of this drug–drug interaction.
Some 21% of the cases studied were identified as suicides.
There is “concerning evidence to suggest that individuals are specifically seeking out P antihistamines for their sedative properties”, the authors said.
“An urgent review of first-generation antihistamines currently assigned under the P classification, with special attention to promethazine, is needed to achieve antihistamine harm reduction,” they continued.
“Dependent upon the assessed risk versus benefit profile of individual antihistamines, recommendations including reclassification to POM status, limitations on quantity per pack, prominent warning labels or the requirement of a formal pharmacist consultation before access to these compounds is granted, could contribute towards achieving this aim.”
However, the authors note that “some pharmacists are already taking matters into their own hands, hiding P antihistamines from open display (particularly promethazine) and refusing their sale”.
Roz Gittins, director of pharmacy at health and social care charity Humankind, said: “The provision of harm reduction information and advice is essential in helping to reduce drug-related harms, including associated deaths. The pharmacy team have a key role to play.
“If there were to be changes to POM status or restrictions on sales, then there would need to be careful consideration of how to avoid unintended consequences — such as people instead resorting to illicit supplies, which could further increase harms.”
She added that it would need to be ensured that “access is not made more difficult or expensive for those who need them for clinically appropriate reasons”.
“Better education around practical strategies, for example, reducing polypharmacy, and non-pharmacological sleep hygiene measures, also need to be considered,” she continued.
The recommendations come after the Medicines and Healthcare products Regulatory Agency said the antihistamine Allevia (fexofenadine hydrochloride) was “sufficiently safe” to move from a POM to the General Sales List in December 2020.
READ MORE: Case study: an error with a medicine containing arachis oil