Georgia Tech
A skin patch with painless needles has the potential to transform the UK’s flu vaccination programme, providing an alternative for children and adult refuseniks, according to infectious diseases experts at Public Health England (PHE).
Their comments follow the success of the first in-human clinical trial of a microneedle patch to deliver the flu vaccine, which was carried out by researchers in the United States.
Katja Höschler and Maria Zambon from PHE’s National Infection Service said the microneedle presented a possible alternative for delivering the flu vaccine to patients both in the UK and elsewhere, particularly in developing countries.
“These early findings suggest the emergence of a promising new option for seasonal vaccination,” they write in a comment piece accompanying the research
[1]
published in The Lancet (online, 27 June 2017).
They describe patient safety, low cost and its “stability” for a year stored at 40 degrees Celsius as the “more exciting” features of the patch.
They write: “Microneedle patches have the potential to become ideal candidates for vaccination programmes, not only in poorly resourced settings, but also for individuals who currently prefer not to get vaccinated, potentially even being an attractive vaccine for the paediatric population, provided late-stage clinical development confirms vaccine efficacy.”
The US phase 1 randomised controlled trial involved 100 people aged 18–49 years who had chosen not to receive the vaccination during the 2014–2015 flu season.
The patients were divided into four groups and received a single dose of inactivated influenza vaccine.
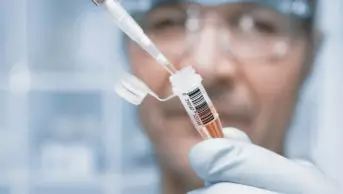
Microneedle patch applied for 20 minutes
One group was given the vaccine via a microneedle patch, another by intramuscular injection, while the third group received a placebo by microneedle patch. The vaccine in all three groups was administered by a healthcare worker. The fourth group self-administered the vaccine via a microneedle patch.

Source: Rob Felt / Georgia Tech
In the study, the microneedle patch was applied for 20 minutes on the wrist.
The injection was given in the arm while the microneedle patch was applied for 20 minutes on the wrist.
In the three microneedle patch groups patients gave an acceptability score of 4.5–4.8 out of a possible 5 immediately after it was administered, similar to the 4.4 score in the traditional needle group. Some 28 days later, 70% of the microneedle patch groups said they preferred it to the injection. At six months, there were no serious side effects reported and no cases of influenza in any of the patients.
According to latest PHE figures, the uptake of the flu vaccine in England during the 2016–2017 flu season was 70.4% in adults aged over 65 years, 48.7% in the children and adult at-risk groups, and 48.7% in pregnant women.
Meanwhile new draft guidance from the National Institute for Health and Care Excellence (NICE), England’s health technology assessment body, proposes that people should be offered the flu vaccine at every opportunity, including when they pick up a prescription, in order to reduce the number of deaths and hospital admissions from flu.
The guidance is out for consultation until 4 August 2017.
References
[1] Rouphael NG, Paine M, Mosley R et al. The safety, immunogenicity, and acceptability of inactivated influenza vaccine delivered by microneedle patch (TIV-MNP 2015): a randomised, partly blinded, placebo-controlled, phase 1 trial. The Lancet doi: 10.1016/S0140-6736(17)30575-5