National Institutes of Health. The image is in the public domain.
Antifungal drug voriconazole increases the risk of developing skin cancer by 73% in lung transplant patients given the drug to prevent infection with Aspergillus, according to research published in the American Journal of Transplantation
[1]
on 3 September 2015.
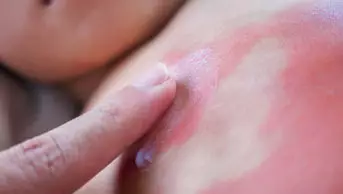
Patients who undergo a solid organ transplant are at a much higher risk of skin cancer than the general population because of the immune suppressants they have to take. For one of the most common skin cancers, cutaneous squamous cell carcinoma (SCC), the risk is 65 times higher.
Patients are also at high risk of fungal infections, which are a significant cause of disease and death. Voriconazole, an extended-spectrum triazole, has been an “important agent in our armentarium against these infections”, says Haifa Lyster, consultant transplant pharmacist at Royal Brompton & Harefield NHS Foundation Trust in London.
The drug is known to increase the risk of SCC, possibly because it makes the skin more sensitive to light. Now, researchers have defined the benefits and risks more accurately. Overall, any exposure to voriconazole increases the risk of SCC by 73% (hazard ratio [HR] 1.73; 95% confidence interval [CI] 1.04–2.88; P=0.03) — this effect is linked to the cumulative dose. For each additional month of treatment with voriconazole at a dose of 200mg twice a day, the risk of SCC increases by 3% (HR 1.03; 95% CI 1.02–1.04; P<0.001).
The study, led by researchers at the University of California, San Francisco (UCSF), evaluated 455 patients who had a lung transplant at the UCSF hospital between 1991 and 2012.
Lyster says the level of risk could be influenced by sun exposure, which was not monitored in the study. “The lack of relative sun exposure of the patients is an important confounder when assessing risks in less sunnier climates, such as the UK,” she says.
Researchers also assessed whether treatment with voriconazole affected risk of death. For patients without Aspergillus colonisation, there was no impact on the risk of death. But for patients with Aspergillus, the risk of death was reduced by 66% in those who took the drug (HR 0.34; 95% CI 0.13–0.91; P=0.03). “This new research underlines the importance of considering the risk versus benefit for individual patients; one size does not fit all,” says Lyster.
Lung transplant recipients with risk factors for SCC are mainly those who are older, white and male. The authors urge transplant physicians to reconsider the use of high cumulative doses of voriconazole in these patients and in patients lacking evidence of Aspergillus colonisation.
Lyster explains that the hospital in the United States where the study was performed uses prophylaxis with voriconazole more widely than in the UK. “We don’t use voriconazole as primary prophylaxis here,” she points out.
She says that where voriconazole is deemed beneficial, for example when Aspergillus has been identified, it is essential to continue to counsel patients on sun protection, limit the duration of courses and ensure patients are reviewed by a dermatologist as part of their follow-up after transplantation.
“In those patients where a higher risk profile exists, then consideration of an alternative antifungal agent should be considered,” she adds.
Other antifungals, such as amphotericin B and posaconazole, were not associated with an increased risk of SCC, the researchers report.
Lyster notes that the study does have a number of limitations, which the authors acknowledge, such as the lack of smoking history assessment, which is another risk factor for cancer.
References
[1] Mansh M, Binstock M, Williams K et al. Voriconazole exposure and risk of cutaneous squamous cell carcinoma, Aspergillus colonization, invasive aspergillosis and death in lung transplant recipients. American Journal of Transplantation 2015. doi:10.1111/ajt.13431.