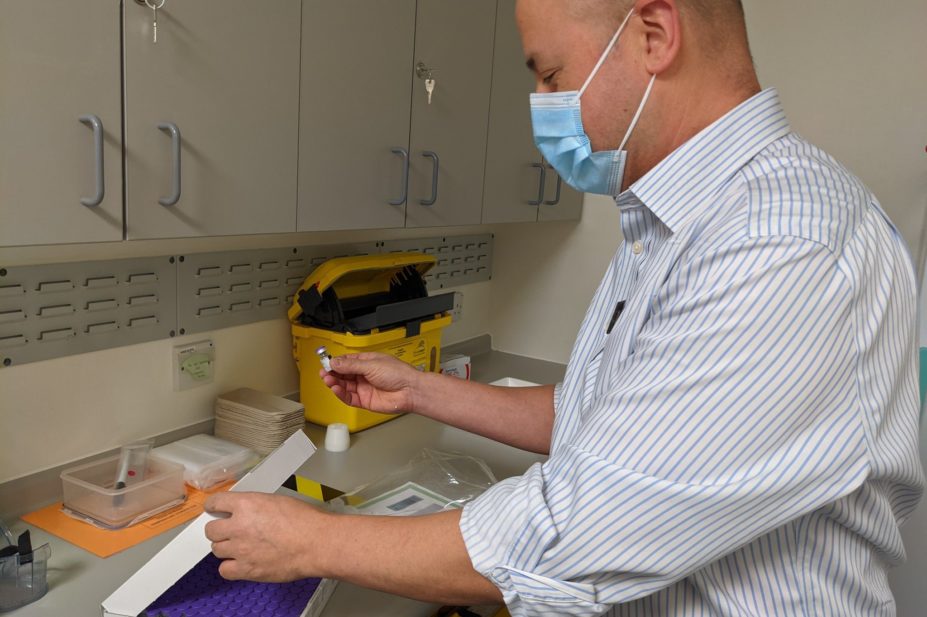
Source: Mark Easter
By now, most people will know Margaret Keenan as the first person in the world to receive the COVID-19 vaccine outside of a clinical trial. This historic event occurred at University Hospitals Coventry & Warwickshire (UHCW) and it was incredible to be a part of it.
I’m divisional clinical director for clinical support services at UHCW, a large teaching hospital with 155 staff working in the pharmacy team. I am also responsible for 20 other services, including critical care, theatres, therapy, outpatients and complex discharge, which have been essential in the hospital’s COVID-19 response.
Like for so many others, 2020 has been a particularly interesting year for me.
The rollout of a vaccine as vital as the COVID-19 vaccine required careful planning. I participated in numerous virtual meetings with my counterparts across the country on logistics of transportation, storage, legal framework and administration of the vaccine.
The trust was initially chosen to be a vaccine ‘hub’ to receive and store the vaccine, and distribute it throughout our health economy — the two neighbouring hospitals and the mental health trust. It was mid-October 2020 when we learned that it would be the Pfizer/BioNTech vaccine coming to UHCW. One of the few things we knew about the vaccine, before it was authorised by the Medicines and Healthcare products Regulatory Agency (MHRA), was that it was a peculiarly fragile product that required storage in a highly specialised freezer, which could maintain the vaccines at -70°C.
In late November 2020, it transpired that there is a limit to the number of times the frozen vaccine can be moved — just four times. That meant that UHCW would now be a hospital hub focused on vaccination, rather than supply, and we had to adapt our plans.
When the MHRA authorised the vaccine on 3 December 2020 — just four days before the first one was administered — we received a technical datasheet, telling us exactly what we needed to do with the medicine. At the same time, Keith Ridge, chief pharmaceutical officer for England, made it clear that chief pharmacists were to be responsible for all aspects of governance. This was incredibly helpful given the novel and fragile nature of the vaccine, and the clear necessity for pharmacy oversight. Together with the medicines safety officer (pharmacist Nixy Samuel), quality controller (Matt Berwick), aseptic service lead (Steve Almond) and aseptic lead technician (Nicole Hadland), I had full oversight of the vaccine receipt, storage, distribution and preparation.
The next day, I received the national protocol for the vaccine, and the team (together with the nurse practice lead) spent the weekend developing standard operating procedures (SOPs) that we could use on the ground. We had to be ready to respond to the latest information available, including changes to the vaccination priority list.
On Monday 7 December 2020, the nursing and pharmacy teams walked the areas in the hospital earmarked for the vaccination centre and we went through the SOPs with them in detail. At this point, because of the challenging process of vaccine preparation, we agreed that during the first week of vaccinations, the pharmacy team would be based in the vaccination centre and conduct most of the drawing up of the vaccine — a task that has now been passed on to the nursing teams.
The anticipation grew on that Monday. The vaccine vials arrived and the trust was then told that it would be administering the very first COVID-19 vaccine in the world. When I got to work at 05:00 on Tuesday morning, the world’s media had starting arriving on site — there were already numerous TV crews outside the hospital, building the excitement and pressure that lasted for several days.
For the team, there was a mixture of nerves and pressure to get this done well in front of the world’s eyes, and immense pride at seeing Margaret, our patient, get that first dose at 06:31 on 8 December 2020, quickly followed by the second patient, Mr William Shakespeare of Warwickshire, at 06:45.
That momentous occasion had such a buzz, but we immediately switched to ‘business as usual’: we still had 100 patients to vaccinate on that first day.
Since then, we have increased our daily vaccination capacity from 100 patients to around 900 and, as the vaccination numbers grow, we are looking to use pharmacy and nursing staff, along with allied health professionals, to run the vaccination centre.
Our learning from this whole process has been shared locally, nationally and internationally — as far afield as Australia and the United States. It was especially rewarding to share our experiences with a network of Italian hospital professionals, with whom we had kept in touch regularly since the onset of the pandemic, and who had supported us with advice on critical care in those early days.
It has been a proud achievement for both me and my team, and for pharmacy teams nationwide, whatever their sector.
Mark Easter, director of pharmacy and divisional clinical director of clinical support services, University Hospitals Coventry & Warwickshire
2 comments
You must be logged in to post a comment.

Photo shows Technical Services lead Steve Almond, not Mark Easter as per the sub title!
Hi Mark,
The text underneath the image is the image credit. I have amended this to clarify.
Best wishes,
Sophie, senior subeditor