
Mclean/Shutterstock.com
One in four people will experience a mental health condition during their lifetime, and medicines are commonly prescribed to treat people. The number of prescriptions rose during the COVID-19 pandemic, with annual antidepressant spending in the NHS increasing by £140m[1,2]. Antipsychotic prescribing has steadily increased over the past decade too[3].
Healthcare professionals working in mental health pharmacy regularly see the challenges relating to these medicines.
A major challenge in psychiatry is non-adherence. Adherence to mental health medicines could be as low as 40–50%, despite it being an important factor in improving patient outcomes[4]. Alongside patient preference and clinical parameters, a trial-and-error approach to prescribing psychiatry medicines is often used, which can mean frequent medication switches from those that do not yield the required therapeutic response or lead to adverse drug reactions (ADRs). The high prevalence of ADRs and lack of therapeutic response from psychiatric medicines are both factors driving poor adherence[5,6].
In recent decades, the role of genetics in contributing to inter-individual variability in drug response has been established[7]. Pharmacogenomics is the study of how variation in a person’s genetic sequence can influence their response to medicines. This can be both pharmacodynamically — how the drug exerts its therapeutic action — and pharmacokinetically — how the drug moves through the body[8]. Pharmacogenetic testing can be used to identify genetic variants responsible for this altered response by obtaining a patient sample (e.g., blood or saliva) and genotyping this in the laboratory. Pharmacogenomic-guided prescribing can then improve the precision of prescribing, reducing the reliance on trial-and-error prescribing approaches by using a more tailored method.
The high prevalence of ADRs and lack of therapeutic response from psychiatric medicines are both factors driving poor adherence
In March 2022, the Royal College of Physicians and the British Pharmacological Society published the ‘Personalised prescribing’ report[9]. This outlines the potential opportunities for pharmacogenomic-guided prescribing and how barriers to its uptake, such as funding, service design, education and training, clinical decision support and patient engagement can be addressed. Using pharmacogenomics to reduce ADRs from antipsychotics and antidepressants is highlighted as an opportunity in the report.
This coincides with the production of the first clinical guidelines for the use of pharmacogenetic testing in psychiatry, authorised by the Dutch Psychiatric Association (NVvP)[10]. These guidelines recommend that clinicians should consider genotyping if patients experience inefficacy or side effects with antidepressants and antipsychotics that have pharmacogenetic recommendations, especially if trialled medicines have similar cytochrome P450 (CYP450) metabolism routes. This can help determine if metaboliser status has contributed to inefficacy or side effects, and this pharmacogenetic information can then be used to personalise future prescribing decisions.
The NHS Genomic Medicine Service is embedding genomic medicine at scale into NHS care, including pharmacogenomics and precision medicine[11]. NHS England has convened an expert test evaluation working group for pharmacogenomics, which will use an ethical evidence-based approach to review drug-gene associations and provide advice to support the development of the NHS England test directory[12,13]. As evidence for pharmacogenomics in psychiatry develops, NHS England will continue to consider the cost-benefit balance of expanding the national genomic test directory to include pharmacogenomic tests for psychiatry.
Could pharmacogenomics help improve antipsychotic prescribing?
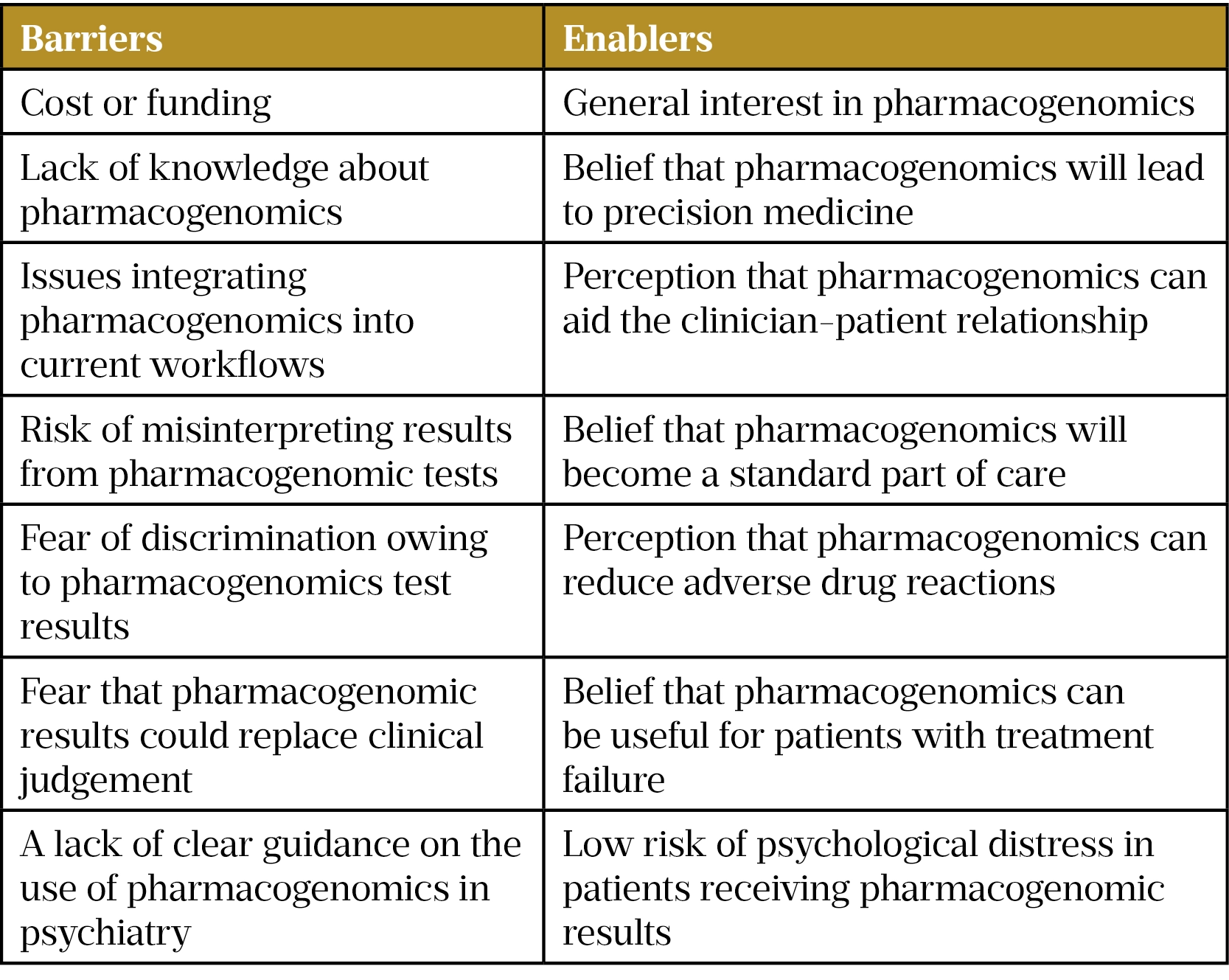
As part of our PhD project at the University of Bradford, sponsored by the Bradford District Care NHS Foundation Trust (BDCT), we have summarised the barriers and enablers (see Table) to implementing pharmacogenetic testing in mental health settings[14]. Throughout the remainder of the project, using qualitative and quantitative methods, we will further explore issues relating to the implementation of pharmacogenetic testing in mental health settings. The project will focus specifically on how pharmacogenomics can be used to support antipsychotic prescribing in first episode psychosis (FEP). It is hoped we can gather perspectives towards using pharmacogenomics in FEP and demonstrate how its use can reduce the reliance on trial-and-error antipsychotic prescribing approaches.
Many people working in mental health will understand the difficulty that healthcare professionals face when trying to communicate the importance of antipsychotic adherence. Clinicians appreciate the benefit that antipsychotics can provide in terms of quality of life and social functioning, but also understand why individuals have reservations about taking antipsychotics, as explained above. As a research team this is where we believe pharmacogenomics can help.
Why focus on first episode psychosis?
Antipsychotics play a critical role in symptom management both during and following FEP. But antipsychotic-naïve individuals have a higher risk of ADRs: a major contributor to antipsychotic discontinuation[15,16]. Yet, adherence can have a positive impact on illness progression and longer-term outcomes, such as relapse, hospitalisation and suicide rates in those who experience psychosis[17–20].
Initial experiences of antipsychotics have been found to contribute to long-term attitudes towards antipsychotics[21]. This means that experiencing ADRs or a lack of efficacy can cause individuals to develop negative perceptions towards antipsychotics, impacting on their willingness to adhere to them in the future[22].
By using a pharmacogenomic approach to antipsychotic prescribing, adherence may improve by reducing the prevalence of ADRs and increasing the likelihood of therapeutic response. With this, the benefits of antipsychotic adherence can be realised and longer-term attitudes towards antipsychotics will likely improve. The time it takes to find the most appropriate antipsychotic for an individual may also be reduced. By tailoring antipsychotic selection and dose to individuals, we hope the difficult conversations around antipsychotic adherence could be made a little easier.
The project so far
A cross-sectional analysis of antipsychotic prescribing in the BDCT Early Intervention in Psychosis (EIP) service found that three in every four antipsychotics prescribed had a pharmacogenetic association, according to the PharmGKB database on drug-gene pairs.
In the future, we believe that pharmacogenomics will be a routine clinical factor that is assessed when making prescribing decisions
This represents an urgent clinical need for NHS mental health settings, in particular EIP services, to follow other clinical specialties in the NHS by integrating pharmacogenetic testing services into treatment pathways. This will allow prescribing of antipsychotics, including aripiprazole, risperidone, olanzapine, quetiapine, haloperidol, clozapine and zuclopenthixol, with pharmacogenetic associations to be tailored to individuals, based on their genetic profile[23–25]. This may include altering the dose of an antipsychotic or avoiding specific antipsychotics owing to the risk of inefficacy or ADRs.
In the future, we believe that pharmacogenomics will be a routine clinical factor that is assessed when making prescribing decisions. Just as prescribers consider renal, hepatic or cardiac function, blood results, allergies, sex, age or ethnicity, pharmacogenetic data can also be embedded into patient records and clinical support systems, and used as an additional piece of clinical information to support prescribers in practising evidence-based medicine.
This could help to address the day-to-day challenges that patients and healthcare professionals face in mental health settings across the NHS. Pharmacogenomics could also reduce costs to the NHS by preventing hospitalisations owing to ADRs and decreasing the time taken trying to find a tolerable and efficacious antipsychotic when using a trial-and-error approach.
By improving medicines optimisation for our mental health populations, pharmacogenomics can increase the safety and effectiveness of psychiatric medicines.
- 1Adult Psychiatric Morbidity Survey: Survey of Mental Health and Wellbeing, England, 2014. NHS Digital. 2016.https://digital.nhs.uk/data-and-information/publications/statistical/adult-psychiatric-morbidity-survey/adult-psychiatric-morbidity-survey-survey-of-mental-health-and-wellbeing-england-2014 (accessed 3 Aug 2022).
- 2Rabeea SA, Merchant HA, Khan MU, et al. Surging trends in prescriptions and costs of antidepressants in England amid COVID-19. DARU J Pharm Sci. 2021;29:217–21. doi:10.1007/s40199-021-00390-z
- 3Buhagiar K, Ghafouri M, Dey M. Oral antipsychotic prescribing and association with neighbourhood-level socioeconomic status: analysis of time trend of routine primary care data in England, 2011–2016. Soc Psychiatry Psychiatr Epidemiol. 2019;55:165–73. doi:10.1007/s00127-019-01793-9
- 4Semahegn A, Torpey K, Manu A, et al. Psychotropic medication non-adherence and its associated factors among patients with major psychiatric disorders: a systematic review and meta-analysis. Syst Rev. 2020;9. doi:10.1186/s13643-020-1274-3
- 5Lally J, Ajnakina O, Stubbs B, et al. Remission and recovery from first-episode psychosis in adults: systematic review and meta-analysis of long-term outcome studies. Br J Psychiatry. 2017;211:350–8. doi:10.1192/bjp.bp.117.201475
- 6Keyloun KR, Hansen RN, Hepp Z, et al. Adherence and Persistence Across Antidepressant Therapeutic Classes: A Retrospective Claims Analysis Among Insured US Patients with Major Depressive Disorder (MDD). CNS Drugs. 2017;31:421–32. doi:10.1007/s40263-017-0417-0
- 7Roden DM, Wilke RA, Kroemer HK, et al. Pharmacogenomics. Circulation. 2011;123:1661–70. doi:10.1161/circulationaha.109.914820
- 8Roden DM. Pharmacogenomics: Challenges and Opportunities. Ann Intern Med. 2006;145:749. doi:10.7326/0003-4819-145-10-200611210-00007
- 9Personalised prescribing: using pharmacogenomics to improve patient outcomes. Royal College of Physicians. 2022.https://www.rcp.ac.uk/projects/outputs/personalised-prescribing-using-pharmacogenomics-improve-patient-outcomes (accessed 3 Aug 2022).
- 10van Westrhenen R, van Schaik RHN, van Gelder T, et al. Policy and Practice Review: A First Guideline on the Use of Pharmacogenetics in Clinical Psychiatric Practice. Front. Pharmacol. 2021;12. doi:10.3389/fphar.2021.640032
- 11Everything you need to know about the NHS genomic medicine service. Pharmaceutical Journal. 2022. doi:10.1211/pj.2020.20207495
- 12Terms of Reference Genomics Test Evaluation Working Groups. NHS England. 2021.https://www.england.nhs.uk/commissioning/wp-content/uploads/sites/12/2021/01/DRAFT-Terms-of-Reference-Test-Evaluation-Working-Groups-v0.3.pdf (accessed 3 Aug 2022).
- 13National Genomic Test Directory. NHS England. 2022.https://www.england.nhs.uk/publication/national-genomic-test-directories/ (accessed 3 Aug 2022).
- 14Jameson A, Fylan B, Bristow GC, et al. What Are the Barriers and Enablers to the Implementation of Pharmacogenetic Testing in Mental Health Care Settings? Front. Genet. 2021;12. doi:10.3389/fgene.2021.740216
- 15Lieberman JA, Dixon LB, Goldman HH. Early Detection and Intervention in Schizophrenia. JAMA. 2013;310:689. doi:10.1001/jama.2013.8804
- 16Gómez-Revuelta M, Pelayo-Terán JM, Juncal-Ruiz M, et al. Antipsychotic Treatment Effectiveness in First Episode of Psychosis: PAFIP 3-Year Follow-Up Randomized Clinical Trials Comparing Haloperidol, Olanzapine, Risperidone, Aripiprazole, Quetiapine, and Ziprasidone. International Journal of Neuropsychopharmacology. 2020;23:217–29. doi:10.1093/ijnp/pyaa004
- 17Alvarez-Jimenez M, Priede A, Hetrick SE, et al. Risk factors for relapse following treatment for first episode psychosis: A systematic review and meta-analysis of longitudinal studies. Schizophrenia Research. 2012;139:116–28. doi:10.1016/j.schres.2012.05.007
- 18Thompson A, Winsper C, Marwaha S, et al. Maintenance antipsychotic treatment versus discontinuation strategies following remission from first episode psychosis: systematic review. BJPsych open. 2018;4:215–25. doi:10.1192/bjo.2018.17
- 19Tiihonen J, Tanskanen A, Taipale H. 20-Year Nationwide Follow-Up Study on Discontinuation of Antipsychotic Treatment in First-Episode Schizophrenia. AJP. 2018;175:765–73. doi:10.1176/appi.ajp.2018.17091001
- 20Fedyszyn IE, Robinson J, Harris MG, et al. Suicidal behaviours during treatment for first-episode psychosis: towards a comprehensive approach to service-based prevention. Early Intervention in Psychiatry. 2013;8:387–95. doi:10.1111/eip.12084
- 21Yeisen RAH, Bjornestad J, Joa I, et al. Experiences of antipsychotic use in patients with early psychosis: a two-year follow-up study. BMC Psychiatry. 2017;17. doi:10.1186/s12888-017-1425-9
- 22Higashi K, Medic G, Littlewood KJ, et al. Medication adherence in schizophrenia: factors influencing adherence and consequences of nonadherence, a systematic literature review. Therapeutic Advances in Psychopharmacology. 2013;3:200–18. doi:10.1177/2045125312474019
- 23Allen JD, Bishop JR. A systematic review of genome-wide association studies of antipsychotic response. Pharmacogenomics. 2019;20:291–306. doi:10.2217/pgs-2018-0163
- 24Arranz MJ, Salazar J, Hernández MH. Pharmacogenetics of antipsychotics: Clinical utility and implementation. Behavioural Brain Research. 2021;401:113058. doi:10.1016/j.bbr.2020.113058
- 25van Westrhenen R, Aitchison KJ, Ingelman-Sundberg M, et al. Pharmacogenomics of Antidepressant and Antipsychotic Treatment: How Far Have We Got and Where Are We Going? Front. Psychiatry. 2020;11. doi:10.3389/fpsyt.2020.00094
1 comment
You must be logged in to post a comment.

Great to see development of MH genomics.