Key points:
- Attention deficit hyperactivity disorder (ADHD) is a condition with significant academic, occupational, social and emotional impairment, which often starts in childhood and continues through adolescence and adulthood.
- Medication treatment is very effective in addressing symptoms of ADHD, when it is carefully titrated to the optimal dose and side effects are effectively addressed.
- Generally stimulant medications (methylphenidate and amphetamines) are more effective than non-stimulants, and are therefore used first unless there is a strong contraindication.
- Improvement in function often requires additional psychosocial treatments (e.g. organisational, time management and social skills, and emotional regulation) because ‘pills don’t teach skills’.
- ADHD is a highly comorbid condition and therefore comorbidities need to be addressed in addition to ADHD for optimal clinical outcomes.
Introduction

Source: Shutterstock.com
Inattention and hyperactive/impulsive symptoms are commonly seen in practice, with up to 6% of children meeting criteria for attention deficit hyperactivity disorder (ADHD)[1]
. Up to 78% of them continue to exhibit symptoms at ten years of follow-up, and inattentive symptoms are more likely to persist into adulthood[2],
[3],
[4]
. In addition, intellectual disabilities have been associated with a longer and more persistent course of ADHD[5]
.
ADHD is a neurodevelopmental disorder characterised by developmentally inappropriate levels of inattention, hyperactivity and impulsivity in various combinations across home, school, work and social settings[6]
. The diagnosis of childhood ADHD requires the presence of six or more symptoms of inattention and/or six or more symptoms of hyperactivity/impulsivity. Similarly, adult ADHD requires the presence of five or more symptoms of inattention and/or five or more symptoms of hyperactivity/impulsivity. ADHD also requires an onset of symptoms before the age of 12, impairment in at least two settings, and symptoms should not be better accounted for by another mental disorder[6]
. In addition to detailed assessment of children and adolescents, behaviour and developmental information needs to be obtained from other sources such as the family and school[6]
. Rating scales, such as the Conners’ rating scales, are useful to support clinical evaluation and monitor progress[7]
. Importantly, a diagnosis of ADHD should be carried out by a competent professional (a specialist such as a psychiatrist or paediatrician, and when available and clinically acceptable, diagnosis and management aided by trained specialist nurses is valuable)[8]
.
ADHD accounts for 30–50% of mental health referrals among children. The combined subtype and a higher prevalence of ADHD is seen among boys[9]
, however, the inattention and hyperactive/impulsive behaviour could also be owing to various other reasons such as mood, anxiety, learning and sleep problems[9],
[10]
. Comorbidities are highly prevalent in ADHD and can include intellectual disability, autism and specific learning disabilities[10]
. Certain genetic conditions have been reported to lead to the increased prevalence of ADHD (e.g. fragile X syndrome)[11]
. ADHD is associated with impairment in various domains, including academic and/or occupational health (including injuries) and relationships, and it is associated with traffic violations and motor vehicle accidents, low self-esteem, legal difficulties, risky sexual behaviour, smoking and substance abuse[12],
[13]
. Adults with ADHD are more likely to be arrested, divorced, have a sexually transmitted disease, and be unemployed[14]
. Around 40–60% of parents with ADHD will have a child with the disorder, and 25% of children with ADHD will have a parent with the disorder. There are added challenges of raising a child with ADHD when the parent has the disorder[15]
. A summary of problems associated with ADHD as a disorder across a person’s lifespan is given in figure 1.
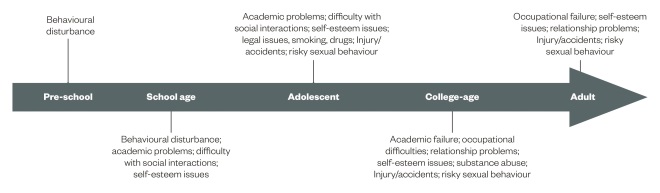
Figure 1: Developmental impact of ADHD
A summary of problems associated with ADHD as a disorder across a person’s lifespan.
Sources
This article focuses on medication treatments for ADHD. An initial search was performed using Medline with keywords referring to ADHD and available medication treatments for children, adolescents and adults. Articles were selected based on their pertinence to medication treatments in ADHD, and supplemented by references from published guidelines and landmark studies (e.g. the multimodal treatment of ADHD study, known as MTA). The retained articles within the domain of medication treatments in ADHD were those pertaining to clinical effects, benefits, limitations, titration procedures, side effects, treatment algorithms, psychological issues in pharmacological management, comorbidities and future developments. The reviewed guidelines included the Canadian ADHD Resource Alliance (CADDRA), the National Institute for Health and Care Excellence (NICE) and the American Academy of Child and Adolescent Psychiatry Practice Parameters (AACAP).
Treatments
The MTA study was among the large-scale studies set up to evaluate different treatment strategies in childhood ADHD[16],
[17]
. It was a multi-site, randomised controlled trial (RCT) of four groups including medication, intensive behavioural treatment, a combination of medication and intensive behavioural treatment, and community treatment[16],
[17]
. It included 579 children with combined ADHD, aged 7.0–9.9 years with a wide range of comorbid conditions[17]
. After 14 months, the study suggested that medication provided in the study but not in the community, with or without behavioural treatment, was the most effective treatment for core ADHD symptoms[16]
. The medication treatment in the community was much less effective since it lacked careful titration, monitoring and follow-up[16]
. While parents preferred behavioural treatment with or without medication, the combination of medication and behavioural treatment was found to be more effective than behavioural treatment alone for problems associated with ADHD, such as social and academic functioning and parent-child relationships[16]
. Medication treatment required close monitoring and adequate dosing to achieve optimal outcomes[16]
.
Medication treatments are important in the management of ADHD symptoms among children, adolescents and adults with ADHD, although non-medication treatments are also often required to improve functioning. Among adults, the non-medication interventions include psychoeducation, behavioural intervention and goal setting, and cognitive behavioural therapy[18],
[19]
. The available medication treatments include stimulant and non-stimulant medications[20]
. The treatment guidelines for ADHD currently available have significant overlap among their treatment recommendations[21]
. However, there are some notable differences; the NICE guidelines recommend medication only in the most severe cases, while CADDRA guidelines recommend medication in almost all cases[21]
. For adults with ADHD, the NICE guidelines recommend that medications could be first-line treatments and they don’t need to be reserved for the most severe cases[22]
. This review will provide an overview of medication treatments, the available evidence, and factors pertinent to the selection of appropriate treatment. The article also follows the Canadian approach, which is holistic, personalised and sees medication as a part of the treatment plan (multimodal approach)[21]
. A summary of the available pharmacological treatments available in Canada for ADHD, as stated in the CADDRA guidelines, is provided in supplementary table 1.
Neurobiology of ADHD and mechanism of action of ADHD medications
ADHD is a highly heritable disorder with heritability of up to 0.76% of the variance in ADHD[23]
. Dysfunction in the cortico-striatal-thalamic-cortical loops have been associated with ADHD. Catecholamine (noradrenaline and dopamine) dysfunction has also been implicated in ADHD, and the most commonly used medications in ADHD act on this system. Delayed brain growth, smaller basal ganglia, cerebellum and frontal lobes on MRI scans, and hypoperfusion in the areas corrected by stimulant medications, have been noted in children with ADHD in neuroimaging studies[24],
[25]
. In addition, various genes (e.g. encoding the dopamine receptor subtypes) have been implicated and are being actively studied in the pathogenesis of ADHD and treatment response to medication[26]
.
Stimulant medications
In use since the 1880s (amphetamine was synthesized in 1887), stimulant medications principally act on dopamine and noradrenaline reuptake inhibition, and presynaptic release. Many studies have documented their safety and efficacy and numerous RCTs have specifically demonstrated their efficacy in ADHD[27],
[28],
[29]
. While methylphenidate is a dopamine reuptake blocker, dexamphetamine brings about dopamine reuptake blockade and presynaptic release. A high response rate of 70–85% has been noted with methylphenidate and amphetamine formulations. The response rate is lower for atomoxetine (60–65%) and guanfacine (30–40%). However, subjects without ADHD also show some improvement in attention with stimulant medications, and therefore, response rate cannot be used to confirm or refute a diagnosis of ADHD[30]
. Release of dopamine and noradrenaline in the subcortical limbic areas has been considered to be responsible for the abuse potential of some stimulant medications. Different medications are available in generic and branded drugs and include immediate release (IR), extended release (ER), slow release (SR), and long acting (LA) formulations.
Methylphenidate formulations
This includes Ritalin (and also Medikinet in the UK), which is available as IR, SR and LA forms in some countries. Ritalin tablets have a short duration of action (3–4 hrs) and Ritalin SR has a 5–6 hour duration of action. However, Ritalin SR has a wax matrix that results in variable release and inconsistent effect[21]
. Biphentin (Equasym XL and Matoride XL, UK equivalents) is a long-acting methylphenidate product purported by the product monograph to last 10–12 hours, but which usually lasts about 8–10 hours in practice. Ritalin LA (Medikinet XL, UK equivalent) provides biphasic delivery of methylphenidate using immediate and delayed-release beads in a capsule (50% immediate, 50% delayed). Concerta (Concerta XL in the UK) delivers methylphenidate using an immediate-release coating and a slow/time-release osmotic mechanism; the OROS delivery system (22% immediate, 78% delayed). The starting doses and titration of various formulations in children, adolescents and adults is further summarised in supplementary table 1.
Focalin and Focalin XR, and the patch (Daytrana), are available in the United States but not in Canada or the UK[21]
. The total daily dose of the transdermal patch is dependent on the patch size and wear duration. Novo-methylphenidate ER-C is a generic methylphenidate-based product available in Canada and it is marketed to be equivalent to Concerta[21]
. However, it peaks and dissipates at a different time point and is therefore not clinically equivalent.
Clinically important side effects include decreased appetite, weight reduction, initial insomnia, somnolence, headache and rebound irritability. Further details on specific side effect profiles of individual methylphenidate formulations published in the CADDRA guidelines is provided in supplementary table 2. The starting doses for Ritalin (for UK Medikinet dose regimens, consult the summary of product characteristics) is 5mg twice or three times daily, and the dose titration, according to the product monograph, is 5–10mg at weekly intervals. The starting doses for Biphentin (for UK Equasym XL and Matoride XL dose regimens, consult the summary of product characteristics) is 10–20mg once daily given in the morning and the dose titration, as per the product monograph, is 10mg at weekly intervals. The starting doses for Concerta (for UK Concerta XL dose regimens, consult the summary of product characteristics) is 18mg once daily given in the morning and the dose titration, as per the product monograph, is 9–18mg at weekly intervals. The starting doses and titration of various formulations for children, adolescents and adults stated in the CADDRA guidelines is further summarised in supplementary table 1.
Amphetamine formulations
Dexamfetamine (Dexedrine in North America, Amfexa in the UK) tablets have a short duration of action (3–4 hours). The pills can be crushed and sprinkled over food; however, it is associated with a greater risk of abuse. Unlike dexamfetamine, Adderall XR and Vyvanse (lisdexamfetamine, Elvanse, UK equivalent) are long-acting and have a 10–13 hour duration of action. Adderall XR delivers mixed salts using immediate and timed-release beads (50% immediate, 50% delayed). Lisdexamfetamine (Elvanse) is a pharmacologically-inactive prodrug composed of a lysine-amphetamine combination that requires an enzyme in the red blood cells to separate the lysine from the amphetamine and activate the amphetamine. Release of the active ingredient in lisdexamfetamine does not rely on gastrointestinal factors, such as gastrointestinal transit time or gastric pH, and it can also be dissolved in water if a child has difficulties swallowing pills. The starting doses and titration of Dexedrine pills and spansules, and lisdexamfetamine, in children, adolescents and adults, is further summarised in supplementary table 1. In addition, the abuse potential is significantly lowered when long-acting stimulants are used and it is particularly low with lisdexamfetamine[21]
.
As with the methylphenidate formulations, clinically important side effects include decreased appetite, weight reduction, initial insomnia, somnolence, headache and rebound irritability. Further details on specific side effect profiles of individual amphetamine formulations published in the CADDRA guidelines is provided in supplementary table 2. The starting doses for dexamfetamine is 2.5–5.0mg twice daily and the dose titration, according to the product monograph, is 2.5–5.0mg at weekly intervals (for UK dose regimens, consult the summary of product characteristics). The starting doses for Adderall XR is 5–10mg once daily given in the morning and the dose titration, as per the product monograph, is 5–10mg at weekly intervals (for UK dose regimens, consult the summary of product characteristics). The starting doses for lisdexamfetamine (Elvanse) is 10–30mg once daily given in the morning and the dose titration, as per the product monograph is by clinical discretion at weekly intervals (for UK dose regimens, consult the summary of product characteristics). The starting doses and titration of various formulations for children, adolescents and adults as stated in the CADDRA guidelines, is further summarised in supplementary table 1.
Non-stimulant medications
Atomoxetine (Strattera)
Atomoxetine is a potent noradrenaline reuptake inhibitor that is highly selective and inhibits the presynaptic noradrenaline transporter. It was initially studied as a potential treatment for depression but was found to be ineffective. An eight-week randomised prospective controlled trial involving subjects with moderate to severely-impairing ADHD treated with atomoxetine reported a response rate of 65% and significant improvement in ADHD symptoms, and measures of social and family functioning[31]
. More than 12 double-blind, placebo-controlled trials support the efficacy of atomoxetine in children, adolescents and adults[32],
[33]
. Children and adolescents have a medium effect size with atomoxetine, while adults have a small to moderate effect size, when compared to the large effect sizes for stimulants[33],
[34]
. Common side effects include dizziness, drowsiness, dyspepsia, sexual side effects and decreased appetite. Less common but not rare (>2%) side effects include nausea, urinary hesitancy, QTc prolongation in cardiac conduction, depression, tremor, early morning awakening, and pruritus (generalised itching). In addition, atomoxetine is a CYP2D6 substrate and needs to be used cautiously when some other medicines are used (e.g. paroxetine, fluoxetine, and quinidine).
AACAP’s dose range guidelines start at 0.5mg/kg/day for 10–14 days, followed by 0.8mg/kg/day for 10–14 days and then the target dose of 1.2mg/kg/day, with maximum doses of 1.4mg/kg/day or 100mg (whichever is less). This careful titration is recommended because 10% of the general population are slow metabolisers of the cytochrome system, which is involved in metabolism of atomoxetine. Therefore, if a large response is seen with 0.5mg/kg/day, the patient might be a slow metaboliser and the dose should not be increased to 1.2mg/kg/day. However, note that this recommended dosing regimen may not be the convention for some prescribers and could differ based on the location of practice. For UK dose regimens, consult the summary of product characteristics. The pills need to be swallowed whole and can be administered once or twice daily; the typical duration of action is 6–10 hours (but could be up to 24 hours). In addition, a period of 6–8 weeks is required to notice the full effect of the medication. The starting doses and titration of various formulations for children, adolescents and adults is summarised in supplementary table 1.
Assessment of liver function is useful but not necessary prior to starting atomoxetine, and monitoring for hepatotoxicity is not routinely required[35]
. A black box warning has been issued by the FDA regarding an increased risk of suicidal thinking among adolescent patients[36]
and height, weight, pulse and blood pressure need to be monitored as small decreases in growth (up to 0.5 cms/year) and small increases in heart rate and pulse have been noted. There is also possible sexual dysfunction among adult patients. However, there is a markedly reduced potential for abuse, as there is no activation of dopamine in the nucleus accumbens with atomoxetine. To sum up, the advantages of atomoxetine compared to stimulants include more continuous coverage during the day, less potential for drug abuse, little or no long-term growth suppression in children followed up to five years, less likely to be used for weight loss in an individual with an eating disorder, lowers risk of worsening tics, and it might be associated with less stigma for some patients[37],
[38],
[39]
. However, the disadvantages are that it has a smaller effect size, a longer titration period that requires waiting for weeks or months for a full response, drug interactions with CYP2D6 inhibitors, and concern about suicidal thoughts[40]
. Atomoxetine may therefore become a drug of choice when there are significant concerns of stimulant diversion or the presence of adverse dopaminergic side effects (e.g. tics or stereotypies) with stimulants.
Guanfacine extended-release (Intuniv)
Intuniv XR was released in the United States in November 2009. It is an alpha-2a agonist, non-stimulant medication with a half-life of 17 hours[41]
. The pill needs to be swallowed whole and the AACAP guidelines is 1–4mg/day (0.05–0.08mg/kg/day up to a maximum of 0.12mg/kg/day). Intuniv can be started at 1mg/day, then increased by 1mg weekly up to 4mg per day. For UK dose regimens, consult the summary of product characteristics. Common side effects include drowsiness, dyspepsia and fatigue, and blood pressure and heart rate should be monitored for hypotension and bradycardia. The patient should be warned against sudden discontinuation, which increases risk for hypertensive crisis. It is also a CYP3A4/5 substrate and must be used cautiously when other medicines are used owing to potential additive effects and drug interactions. It is approved as an adjunct therapy to stimulants and is much less effective when directly compared with stimulants[42]
. The starting doses and titration of Intuniv XR in children, adolescents and adults are further summarised in supplementary table 1.
Clonidine extended-release (Kapvay, Jenloga)
This medication was released in the United States in 2010 and is not yet currently available in the UK. It is an alpha-2a agonist, non-stimulant, and a non-schedule II medication. It has a half-life of 12 hours (6 hours to Cmax) with a dose range of 0.1–0.4mg total daily dose[43]
. Starting doses for clonidine could be 0.1mg each evening, followed by a gradual increase of 0.1mg weekly (if needed, up to 0.4mg total daily dose, given divided, twice daily). Note that the medication should not be cut in half. Common side effects include drowsiness, dyspepsia, fatigue and headache, and blood pressure and heart rate should be monitored for hypotension and bradycardia. The AACAP advises that it must be used cautiously when other medicines are used because of potential additive effects on cardiac conduction via QT prolongation and drug interactions[21]
. The patient should be warned against sudden discontinuation, which increases risk for hypertensive crisis.
It is approved as an adjunct therapy to stimulants and is much less effective when compared directly with stimulants (both for inattentive and hyperactive symptoms). In addition, it is more sedating when compared to Intuniv XR[41]
.
Clinical effects, benefits and side effects
The main clinical benefits of stimulant medications include improved focus, concentration and attention span, and reduced hyperactivity, impulsivity and fidgeting. With regard to short-term benefits, around 70% of children and adults will respond to any given stimulant, compared to 4–30% who respond to placebo[44]
. Around 90% will respond to either methylphenidate or amphetamine and the overall effect size for stimulants, as a class, is large[45]
. With regard to long-term benefits, numerous studies of up to two years in duration, as well as clinical experience, show sustained improvement with ongoing medication[28],
[46]
. Sustained improvement in academic performance has been noted with ongoing treatment over five years and much of the benefit of stimulant medication disappears after the medication is discontinued[47]
. Also, the use of stimulant medications for ADHD during childhood may not have an impact on adult substance abuse[48]
. While some authors have found evidence to suggest that treatment of children and adolescents with stimulant medications reduces their risk of adult substance abuse[49]
, others have found that it neither increases nor decreases the level of adult substance abuse[50],
[51]
.
Important contraindications to using stimulant medications include treatment with MAO inhibitors, or two weeks following MAO discontinuation, untreated hyperthyroidism, glaucoma, moderate to severe hypertension, pre-existing severe gastrointestinal narrowing, advanced arteriosclerosis, and known hypersensitivity or allergy to products[21]
. In addition, stimulant medications can increase the levels of phenytoin, carbamazepine, phenobarbital and valproic acid (which are amenable to serum monitoring), while these medications could also lower stimulant levels in the blood as they are universal enzyme inducers[21]
. Finally, peak and trough effects and end-of-dose rebound effects need to be kept in mind[21]
. A comparison of the long-acting formulations is provided in supplementary table 3.
Common side effects
The side effect profiles of stimulants are well known and are generally manageable[32],
[46]
. A summary of the side effects associated with individual medications is provided in supplementary table 2. Side effects include appetite suppression, weight loss, irritability, anxiety, sadness, tics, sensitivity to crying, sleep problems, stomach ache, headache, dysphoria, ‘zoned-out’ effects and blood pressure changes[52]
. However, it is important to note that many side effects improve with time and are most pronounced at the beginning of treatment[53]
. Amphetamine formulations may produce more sleep and appetite problems, especially at higher doses. Benefits of long-acting stimulants include relieving symptoms for most of the day, improved compliance, decreased abuse potential, and a smoother and more consistent coverage[21]
.
Managing common side effects
Sleep problems
The most common sleep problem comorbid with ADHD is delayed sleep phase syndrome (DSPS); however, children with ADHD are also at increased risk for other sleep disorders. Stimulant medications can worsen already existing sleep difficulties (such as DSPS) and, therefore, stimulants should be preferably prescribed early in the day; small doses can be added later in the day to prevent rebound. Some children benefit from an early evening dose of methylphenidate to reduce hyperactivity in the run up to bedtime. While there are some published reports suggesting that methylphenidate may promote sleep if given at night for some patients, more systematic studies are necessary to modify current clinical practice. Sleep hygiene approaches are helpful in the management of sleep problems that are seen in ADHD and its associated medication treatments[21]
. If sleep hygiene proves inadequate, adding melatonin one hour prior to bedtime has been shown to be useful[21]
.
Appetite suppression and management
Growth restriction is covered under ‘Growth suppression’. Parents need to monitor height and weight, ensure availability of nutritious food, and encourage healthy eating habits[21]
. Encouraging increased food intake when the medication is not operative (e.g. breakfast time before medication is taken and before bedtime when it has worn off) helps to maintain a 24-hour calorie intake.
Headaches
A tension-type headache can occur, most commonly in the first 2–3 hours after medication intake. It is often accompanied by nausea, gastric irritation or hunger. Symptoms usually respond to mild analgesics[21]
.
Serious side effects
Cardiac events
Patients who have been treated with stimulants and have had serious cardiac events have been primarily those with pre-existing cardiac conduction defects and structural cardiac abnormalities. In addition, analysis of patient-year exposure data on children with ADHD suggests that the rate of sudden death is similar to the general population[54]
. Patients with ADHD should undergo a careful physical examination and a personal and family history examination that could indicate an increased risk for sudden death. This includes history of congenital heart abnormality, sudden tachycardia, fainting, marked shortness of breath on exertion, and family history of sudden cardiac death prior to initiating stimulants and atomoxetine[54]
. However, while routine electrocardiogram (ECG) is not recommended when initiating treatment, the presence of suspected heart disease or identified risk factors for sudden death suggests that a consultation with a cardiologist is recommended[54]
. With continued treatment, changes in personal or family cardiac history needs to be monitored, along with a regular assessment of blood pressure and heart rate[55]
. Tachycardia can be an uncommon but troublesome side effect that often leads to further investigation and drug discontinuation.
Growth suppression
Evidence is mixed but most data suggest small but significant decreased growth rates, particularly during initial therapy and when higher doses of stimulants are continuously given over long periods of time[56]
. Any growth that has been suppressed is likely to be regained in the late teens or during drug holidays. Children especially at risk are those who are continuously medicated during childhood. Adult growth suppression of around 2cm has been noted depending on the extent of continuous use[56]
. Therefore, plotting heights every three months is necessary to monitor growth and provide summer drug holidays if required, or if possible.
Tics and ADHD
Mild or moderate tics occur in a significant number of patients with or without ADHD pharmacotherapy, and 5–20% of schoolchildren will experience a simple, or complex, tic in their lifetime[28]
. A review of 122 children treated with stimulant medication found 9% developed transient tics and <1% developed chronic tics[57]
. Tics are usually transient and very rarely do patients develop a chronic tic disorder. The benefits of improved ADHD symptoms need to be carefully considered against the downside of tic exacerbation. If tics do occur, or are worsened, the dose could be decreased, or a switch to another stimulant could be tried and an additional adjunctive drug, such as clonidine or guanfacine, to treat the tics could be initiated. In addition, non-stimulant medications, such as atomoxetine, could be tried. It is important to note that the cause of tics when a person is on stimulant medication is unclear and other possibilities should also be explored.
Rare side effects
Hallucinations and psychosis, or mania-type symptoms, are rarely seen with stimulants; occurring at a rate of 1.45 per 100 person years[58]
. Typically, visual hallucinations of snakes, insects or worms have been reported[58]
. If there is no history of psychosis prior to starting stimulants, psychotic symptoms usually stop when the stimulant is discontinued[58]
. There is emerging evidence that careful reintroduction of stimulants might be another option after discontinuation[59]
.
Titration procedures
Clinical assessment and monitoring always includes feedback from teachers. To determine optimal dose, it is an appropriate strategy to ‘start low, go slow’, and ‘keep going until you can determine the optimal risk/benefit ratio’. Identify a regular activity of the patient that requires concentration during a time the medication is active and then monitor the effect when the medication is titrated. The optimal dose is the dose above which no further improvement is noted and where side effects are manageable. It is important to measure outcomes; ADHD rating scales could be used with the patient as a psychoeducational tool as well as patients themselves finding observational anchors they can use to self monitor progress. To optimise outcomes, the formulation needs to be chosen with the needs of the patient and family in mind. Therefore, it is important to explore what the patient needs and whether the psycho-stimulant is required early in the morning for just school, or whether it is also required for homework and peer activities. Additionally, parents and teenagers can have preferences for specific formulations and drug insurance coverage can limit decisions. Parents also need to be trained to observe the efficacy and side effects throughout the day and into the evening when their children are on medications for ADHD. A summary of dose titration among children, adolescents and adults is provided in supplementary table 1.
How to initiate dosing
Dosing is generally not initiated by weight, unless patients weigh less than 25kg (0.3–1mg/kg/day for methylphenidate, 0.15–0.5mg/kg/day for amphetamine). Careful titration needs to be done until a plateau is seen in terms of efficacy, or significant side effects begin. A typical example is to start at 5mg methylphenidate or 2.5mg amphetamine. The doses can then be increased every 1–2 weeks by 5mg for methylphenidate, or 2.5mg for amphetamine, in accordance with parent and teacher reports of efficacy and side effects. This needs to be mutually decided upon by doctor and family, in consultation with weekly or bi-weekly reports, and gradually titrated upwards while checking for side effects and efficacy. Once stimulant titration is optimised, repeat prescriptions could be supplied by the family doctor, who needs to continue monitoring efficacy and side effects, as well as blood pressure, pulse, height and weight.
Medication use in adult ADHD
Paediatric and adolescent guidelines can be used to start treatment. Most adults will tolerate larger doses than the typical doses used in paediatrics. Dosing of adult ADHD does not usually need to exceed FDA maximums for paediatric dosing, though some exceptions exist for adults requiring higher doses as summarised in supplementary table 1. As in children and adolescents, medication choice is based on the duration of action, comorbidities, other symptom targets, patient preference, family history, patient medication history, and risk of abuse. The NICE guidelines suggest using methylphenidate as the first-line therapy followed by atomoxetine[22]
.
Suggested pharmacological algorithm for ADHD
Guidelines regarding medication use differ. For example, CADDRA and the American Academy of Child and Adolescent Psychiatry suggest that the first-line medications are the stimulants[21],
[28]
, with methylphenidate or amphetamine and its formulations being the first choice. The second-line decisions are the non-stimulants (e.g. atomoxetine and alpha-2 agonists such as guanfacine) and the third-line treatments include bupropion and tricyclic antidepressants. The CADDRA guidelines suggest long-acting formulations of stimulants as first-line therapy (e.g. Adderall XR, Biphentin, Concerta and Vyvanse), followed by atomoxetine and guanfacine; the latter often being used as an adjunct[21]
. This is followed by other short-acting medications (e.g. Dexedrine and Ritalin)[21]
.
The NICE guidelines suggest medication as first-line therapy in only severe cases and as second-line therapy when psychological approaches have not yielded therapeutic benefits. The recommended medication treatments include methylphenidate followed by atomoxetine, and if both of these are not effective at maximal doses, amphetamines could be tried[22]
.
Common errors in dosing stimulants
Failure to increase dosing slowly until no further improvement is noted, or side effects are manageable, is a commonly observed error (the MTA study showed that lower dosing in the community sample was less effective). However, beginning with a dose that is too high is another common error. ‘Start low and go especially slow’ is often a good approach with patients who are developmentally delayed. It is common clinical experience to observe that, for some patients, the duration of effect is either longer or shorter than that documented in the literature[21]
.
Failure to assess the duration of action can lead to underdosing or overdosing (may need to calculate the sum of dosages with IR formulations). Patients feeling irritable, ‘too wired’ or ‘too serious’, is a sign of overdose. However, if symptoms become marked at the end of the day, they could be a sign of rebound. Importantly, failure to use another psychostimulant if the first or second stimulant fails, and not exploring and adequately addressing side effects, can lead to suboptimal outcomes. In addition, an untreated comorbid psychiatric disorder (e.g. a mood or anxiety disorder) can decrease the response to stimulants or reduce the ability to tolerate the medication. While in children ADHD often gets diagnosed and treated without complete evaluation of comorbidities, among adults the difficulty in treating substance, mood or anxiety disorders can lead to ADHD being overlooked. Finally, failure to use input from the school and family to assess treatment response and side effects can also lead to suboptimal outcomes.
Additional factors in medication treatment in adolescents and adults
It is important for adolescents to be seen alone, with special emphasis placed on forming an effective alliance with their healthcare professional, and with treatment tailored to their concerns. The main issues that should be reviewed include: side effects; signs of overmedication and undermedication; substance abuse or drug misuse; intermittent use of medications; drug-drug interactions; driving risk; and sexual risk. It is important to value adolescent perspectives pertaining to their medication use and decisions, and allow them more personal control over their treatment. In addition, prescription of stimulant medications for shorter durations of time (e.g. 28 days) with regular follow-up can reduce diversion or misuse.
Switching medications
Switching from a stimulant to another stimulant
A summary of pertinent issues as identified in the CADDRA guidelines is provided in supplementary table 4.
Switching from a stimulant to atomoxetine or guanfacine
Decrease the stimulant doses gradually, then start the non-stimulant at a low dose and increase gradually. Clinicians also use a cross-over approach with the simultaneous decrease of one medication and increase of the other. Careful monitoring and assessing of side effects in this process is important. The addition of atomoxetine to stimulants in non-responders does not require special procedures.
Switch from atomoxetine or guanfacine to stimulant
Approach is similar that described in ‘Switch from stimulant to atomoxetine or guanfacine’ above. Clinicians also use a cross-over approach.
Medication management of comorbidity
A summary of the approach for the management of comorbidity in ADHD proposed in the CADDRA guidelines is provided in table 1. In treating ADHD and various comorbid conditions, side effects and risks of the combinations of medications for ADHD, and their comorbid conditions, needs to be taken into consideration.
| Note: Drug combinations and antipsychotics use described in this table is off-label use and reserved for complex cases. Source: reproduced with permission from CADDRA[21] | |
| Psychiatric problems | Clinical aspects to take into account in the treatment process when comorbid with ADHD |
| Mood disorders | |
| Major depression | Treat the most impairing disorder first. Moderate to severe depression should be treated first and suicide must be assessed in all cases. Dysthymia and mild depression may benefit from ADHD treatment first. Stimulants can be combined with the majority of antidepressants when monitored. Also consider cognitive behavioural therapy (CBT). In adults, bupropion and desipramine may reduce ADHD symptoms, but with an effect size significantly lower than psychostimulants. |
| Bipolar disorder | Treat bipolar disorder. Treatment of ADHD can be offered when bipolar disorder is stabilised. Refer to specialist. |
| Anxiety disorders | |
| Generalised anxiety disorder | Treat the most impairing disorder first. Some patients may show worsening of anxiety and some may show improvement in their symptoms. ADHD treatments can be less tolerated in some individuals in this population. Note possible pharmacological interactions with medications metabolised through CYP2D6 system. Start low, go slow but titrate up to therapeutic dose. If not tolerated, switch to another medication, such as atomoxetine. Also consider CBT. If atomoxetine is much less effective, can refer to specialist for augmentation with stimulants. |
| Panic disorder | |
| Social phobia | |
| Obsessive compulsive disorder (OCD) | |
| Post-traumatic stress disorder (PTSD) | |
| Autism spectrum disorder (ASD) | ADHD treatments can be less tolerated in some individuals in the population but could be very helpful in the general management. Start low, go slow, but titrate up to therapeutic dose. If not tolerated, switch to another medication. Refer to specialist for specific interventions for ASD. |
| Psychotic disorders | Treat psychotic disorder first (refer to a specialist, e.g. a psychologist — treatment of ADHD can trigger a psychotic relapse in a predisposed patient). Stable patients who are in remission may benefit from ADHD treatment. |
| Oppositional disorder and conduct disorder | Treat both conditions. Oppositional disorder needs psychosocial interventions. Moderate and severe cases may require combinations of psychostimulants and an alpha-2 agonist such as clonidine, or guanfacine. Conduct disorder needs psychosocial interventions and may involve legal issues. Pharmacological treatment of ADHD may help better modulate reactive-impulsive behaviours. Adding an antipsychotic may improve the symptoms of conduct disorder, according to some cases cited in the literature. |
| Borderline personality disorder | Reducing impulsivity and increasing attention when treating comorbid ADHD may help the patient with a personality disorder to better participate in their psychological treatments. |
| Antisocial personality disorder | Treating patients with antisocial personality disorder and ADHD requires more complex and comprehensive interventions. |
| Medical problems | Clinical aspects to take into account in the treatment process when comorbid with ADHD |
| Epilepsy | Treat epilepsy first, then ADHD. New onset seizure should be managed with antiepileptic medication. Level of antiepileptic medications may increase with methylphenidate as a result of enzyme inhibition. |
| Tics | ADHD medications do not cause tics but some may increase or reduce tics, and uncover pre-existing tics. However, the presence of tics is not a contraindication for ADHD medication. Atomoxetine, clonidine and guanfacine have shown promise in this population. Addition of antipsychotic may be required in severe cases. |
| Sleep-related disorders | Treat primary disorder first. |
| Sleep apnoea | Psychostimulants can reduce residual sleepiness and improve daily function in sleep apnoea and narcolepsy with or without ADHD. |
| Cardiovascular problems | Physical exam before treatment (blood pressure [BP], pulse, cardiac auscultation). Echocardiogram (ECG) and cardiac consult if positive cardiac history or structural heart disease. Measure BP and pulse and monitor vital signs and cardiac side effects during treatment. |
| Obesity | Discuss healthy eating and sleep habits and increase exercise. ADHD treatment may improve a patient’s capacity to implement lifestyle changes. |
| Other problems | Clinical aspects to take into account in the treatment process when comorbid with ADHD |
| Learning disorders | Treat specific learning disorders. ADHD treatments can improve attention, allowing improvement in learning skills. School adaptations, study and organisational skills should be considered and offered when needed. |
| Speech disorders | Treat specific speech disorders. Refer to special education teacher, psychologist and/or speech and language therapist for specific interventions. |
| Developmental coordination disorder | Treat coordination disorders. Refer to occupational therapist and/or physiotherapist for specific interventions. |
| Low IQ | Treat ADHD and adapt non-pharmacological approaches to the patient’s IQ level. |
| High IQ | Treat ADHD and adapt curriculum to the patient’s IQ level. |
Psychological issues in pharmacological management
A large proportion of medication prescriptions are not fulfilled nor taken properly. It is important to be aware of some of the common issues that hinder compliance and address them during clinical assessment and follow-up. Stigmatisation is often an issue both for parents and children, and this needs to be addressed when initiating treatment or discussing treatment options. The meaning and significance of a drug can affect the way patients view the drug, the prescriber and themselves[60]
. It is also important to pay attention to ongoing family stressors and examine the possibility of parental psychopathology.
Inadequate parental surveillance of adherence could limit compliance. Ambivalence of both parent and child, or teenager, regarding the need for medication could reflect various underlying issues, including power struggles within the family. There could also be a misunderstanding of doses, serum levels and onset of effects. Medication side effects need to be discussed in detail and dose adjustments need to be made in accordance with efficacy and side effects reported by patients and families. In addition, information obtained by patients and families from alternative sources, including the internet, could be cause for concern. Finally, among teenagers and adults, substance abuse and diversion from stimulant medications is a concern that needs sensitive assessment.
Studies suggest that ADHD medications, including stimulants, do not exacerbate substance use disorder (SUD). While individuals with ADHD do less well in SUD treatment, ADHD medications have not proven beneficial in reducing substance abuse, and stimulants remain the most effective ADHD medications, even with ongoing SUD. However, the main risks that need to be considered are the illicit diversion from stimulants to controlled substances, and overuse if a patient is abusing their own stimulant medication[61]
. In the presence of ADHD and SUD medication, decisions require individualised risk-benefit assessment. Extended release stimulants are less likely to be abused and non-stimulant medications remain an option for treatment. Importantly, the concurrent SUD itself requires treatment, often before the ADHD[61],
[62]
.
Medication treatment is only a part of the overall treatment plan in ADHD. Medication alone is often not adequate enough to treat patients with ADHD because it does not teach skills such as organisational, social and emotional regulation, and study skills. Therefore, a variety of psychosocial treatments are also required in addition to the medication treatments. Even though medication treatments are very helpful, many patients do not continue to take the medication and a careful exploration of various issues, including effectiveness and side effects, need to be regularly assessed. Exploration of compliance is particularly important as children enter adolescence and young adulthood where there is a higher likelihood for non-compliance.
Note that if the patient has difficulty swallowing the medication, some medications (e.g. Equasym XL and Adderall XR) can be sprinkled on soft food and some (e.g. Elvanse) can be dissolved in water.
Future developments, emerging ideas and trends
Recent emphasis has been on developing longer-acting versions of stimulant and other medication treatments with the advent of various innovative modes of drug delivery. This includes medications such as the extended-release versions and Biphentin. The following substances are being explored for other conditions and may prove helpful in ADHD but their utility is speculative at this point in time. Several novel potential mechanisms are being explored to improve cognitive parameters in ADHD and in other disorders such as schizophrenia and dementia, where cognitive dysfunction is a core feature. The role of synaptosomal-associated protein 25 (SNAP 25) with atomoxetine is currently being examined[63]
and enhancing prefrontal histamine by blocking its presynaptic H3 autoreceptor is also being tested with H3 receptor antagonists[64]
. Another option is to increase prefrontal acetylcholine by targeting the nicotinic cholinergic receptors, and several a7-nicotinic receptor agonists are being investigated[65]
. While there are some interesting results, development of tolerance and comorbid smoking are potential impediments. Multifunctional agents (e.g. RG3487, which is both an a7-nicotinic receptor partial agonist and 5-HT3 receptor antagonist) are being studied[66]
. Vortioxetine, which currently has approval for treatment of major depressive disorder, is a multifunctional antidepressant that appears to ameliorate some of the cognitive dysfunction seen in depression and has potential to be studied further in ADHD[67]
. Other pro-cognitive mechanisms include ampakines that increase glutamate transmission at AMPA receptors, 5-HT6 receptor antagonists and PDE4 (phosphodiesterase) inhibitors[68],
[69],
[70]
.
Conclusion
In summary, ADHD is a common, usually lifelong condition. Effective medication treatment exists but requires careful titration to the optimal dose, appropriate side effect management, and ongoing monitoring and dose adjustment. Psychosocial treatments are also often needed to ensure optimal functioning.
Venkat Bhat is a resident in psychiatry and Lily Hechtman is professor of psychiatry and pediatrics, director of research, division of child psychiatry, McGill University, Montreal Children’s Hospital, Canada. Correspondence to:
lhechtman@hotmail.com
Financial and competing interests disclosure:
Venkat Bhat has no relevant affiliations or financial involvement with any organisation or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. Lily Hechtman has received research support, served on advisory boards, and been a speaker for Eli Lilly, GlaxoSmithKline, Ortho Janssen, Purdue and Shire. No writing assistance was utilised in the production of this manuscript.
Reading this article counts towards your CPD
You can use the following forms to record your learning and action points from this article from Pharmaceutical Journal Publications.
Your CPD module results are stored against your account here at The Pharmaceutical Journal. You must be registered and logged into the site to do this. To review your module results, go to the ‘My Account’ tab and then ‘My CPD’.
Any training, learning or development activities that you undertake for CPD can also be recorded as evidence as part of your RPS Faculty practice-based portfolio when preparing for Faculty membership. To start your RPS Faculty journey today, access the portfolio and tools at www.rpharms.com/Faculty
If your learning was planned in advance, please click:
If your learning was spontaneous, please click:
References
[1] Polanczyk G, de Lima MS, Horta BL et al. The worldwide prevalence of ADHD: a systematic review and metaregression analysis. The American Journal of Psychiatry 2007;164:942–948. doi: 10.1176/ajp.2007.164.6.942
[2] Biederman J, Petty CR, Evans M et al. How persistent is ADHD? A controlled 10-year follow-up study of boys with ADHD. Psychiatry research 2010;177:299–304. doi: 10.1016/j.psychres.2009.12.010
[3] Wilens TE, Biederman J & Spencer TJ. Attention deficit/hyperactivity disorder across the lifespan. Annual review of medicine 2002;53:113–131. doi: 10.1146/annurev.med.53.082901.103945
[4] Faraone SV, Biederman J & Mick E. The age-dependent decline of attention deficit hyperactivity disorder: a meta-analysis of follow-up studies. Psychological medicine 2006;36:159–165. doi: 10.1017/S003329170500471X
[5] Xenitidis K, Paliokosta E, Rose E et al. ADHD symptom presentation and trajectory in adults with borderline and mild intellectual disability. Journal of Intellectual Disability Research 2010;54:668–677. doi: 10.1111/j.1365-2788.2010.01270.x
[6] American Psychiatric Association. Diagnostic and statistical manual of mental disorders, 5th edition. Arlington: American Psychiatric Publishing 2013.
[7] Conners CK. Conners 3rd Edition. Toronto: MHS 2008.
[8] Kendall T, Taylor E, Perez A et al. Guidelines: diagnosis and management of attention-deficit/hyperactivity disorder in children, young people, and adults: summary of NICE guidance. The BMJ: British Medical Journal 2008;337:a1239. doi: 10.1136/bmj.a1239
[9] Murphy K & Barkley RA. Attention deficit hyperactivity disorder adults: comorbidities and adaptive impairments. Comprehensive psychiatry 1996;37:393–401. doi: 10.1016/S0010-440X(96)90022-X
[10] Rowland AS, Lesesne CA & Abramowitz AJ. The epidemiology of attentionâ€deficit/hyperactivity disorder (ADHD): a public health view. Mental retardation and developmental disabilities research reviews 2002;8:162–170. doi: 10.1002/mrdd.10036
[11] Seager MC & O’Brien G. Attention deficit hyperactivity disorder: review of ADHD in learning disability: the Diagnostic Criteria for Psychiatric Disorders for Use with Adults with Learning Disabilities/Mental Retardation [DCâ€LD] criteria for diagnosis. Journal of Intellectual Disability Research 2003;47:26–31. doi: 10.1046/j.1365-2788.47.s1.30.x
[12] Torgersen T, Gjervan B & Rasmussen K. ADHD in adults: a study of clinical characteristics, impairment and comorbidity. Nordic journal of psychiatry 2006;60:38–43. doi: 10.1080/08039480500520665
[13] Spencer TJ, Biederman J & Mick E. Attention-deficit/hyperactivity disorder: diagnosis, lifespan, comorbidities, and neurobiology. Academic Pediatrics 2007;7:73-81. PMID: 17261486
[14] Mannuzza S, Klein RG & Moulton JL. Lifetime criminality among boys with attention deficit hyperactivity disorder: a prospective follow-up study into adulthood using official arrest records. Psychiatry research 2008;160:237–246. doi: 10.1016/j.psychres.2007.11.003
[15] Sonuga-Barke EJ, Daley D & Thompson M. Does maternal ADHD reduce the effectiveness of parent training for preschool children’s ADHD? Journal of the American Academy of Child & Adolescent Psychiatry 2002;41:696–702. doi: 10.1097/00004583-200206000-00009
[16] Jensen PS, Hinshaw SP, Swanson JM et al. Findings from the NIMH Multimodal Treatment Study of ADHD (MTA): implications and applications for primary care providers. Journal of Developmental & Behavioral Pediatrics 2001;22:60–73. PMID: 11265923
[17] Arnold LE, Abikoff HB, Cantwell DP et al. NIMH collaborative multimodal treatment study of children with ADHD (MTA): Design, methodology, and protocol evolution. Journal of attention disorders 1997;2:141–158. doi: 10.1177/108705479700200301
[18] CADDRA. Canadian Attention Deficit Hyperactivity Disorder Resource Alliance (CADDRA): Canadian ADHD Practice Guidelines, 1st edition. Toronto, Canada: CADDRA 2006.
[19] Knouse Le & Safren SA. Current status of cognitive behavioral therapy for adult attention-deficit hyperactivity disorder. Psychiatric Clinics of North America 2010;33:497–509. doi: 10.1016/j.psc.2010.04.001
[20] Pringsheim T, Hirsch L, Gardner D et al. The Pharmacological Management of Oppositional Behaviour, Conduct Problems, and Aggression in Children and Adolescents With Attention-Deficit Hyperactivity Disorder, Oppositional Defiant Disorder, and Conduct Disorder: A Systematic Review and Meta-Analysis. Part 1: Psychostimulants, Alpha-2 Agonists, and Atomoxetine. Canadian Journal of Psychiatry 2015;60:42–51. PMID: 25886655
[21] Canadian Attention Deficit Hyperactivity Disorder Resource Alliance (CADDRA). Available from: http://www.caddra.ca (accessed on February 2016).
[22] National Collaborating Centre for Mental Health (NCCMH). Attention deficit hyperactivity disorder: the NICE guideline on diagnosis and management of ADHD in children, young people and adults. London, UK: The British Psychological Society and the Royal College of Psychiatrists, London 2009.
[23] Faraone SV & Doyle AE. The nature and heritability of attention-deficit/hyperactivity disorder. Child and adolescent psychiatric clinics of North America 2001;10:299–316. PMID: 11351800
[24] Cherkasova MV & Hechtman L. Neuroimaging in attention-deficit hyperactivity disorder: beyond the frontostriatal circuitry. Canadian journal of psychiatry 2009;54:651–664. PMID: 19835672
[25] Volkow ND, Wang GJ, Kollins SH et al. Evaluating dopamine reward pathway in ADHD: clinical implications. Jama 2009;302:1084–1091. doi: 10.1001/jama.2009.1308
[26] Cortese S. The neurobiology and genetics of Attention-Deficit/Hyperactivity Disorder (ADHD): What every clinician should know. European journal of paediatric neurology 2012;16:422–433. doi: 10.1016/j.epjn.2012.01.009
[27] Davidson MA. ADHD in adults: a review of the literature. Journal of attention disorders 2008;11:628–641. doi: 10.1177/1087054707310878
[28] Pliszka S. Practice parameter for the assessment and treatment of children and adolescents with attention-deficit/hyperactivity disorder. Journal of the American Academy of Child & Adolescent Psychiatry 2007;46:894–921. doi: 10.1097/chi.0b013e318054e724
[29] Seiden LS, Sabol KE & Ricaurte GA. Amphetamine: effects on catecholamine systems and behavior. Annual Review of Pharmacology and Toxicology 1993;33:639–676. doi: 10.1146/annurev.pa.33.040193.003231
[30] Kratochvil CJ, Heiligenstein JH, Dittman R et al. Atomoxetine and methylphenidate treatment in children with ADHD: a prospective, randomized, open-label trial. Journal of the American Academy of Child & Adolescent Psychiatry 2002;41:776–784. doi: 10.1097/00004583-200207000-00008
[31] Michelson D, Faries D, Wernicke J et al. Atomoxetine in the treatment of children and adolescents with attention-deficit/hyperactivity disorder: a randomized, placebo-controlled, dose-response study. Pediatrics 2001;108:e83. PMID: 11694667
[32] Vaughan B, Fegert J & Kratochvil CJ. Update on atomoxetine in the treatment of attention-deficit/hyperactivity disorder. Expert Opinion on Pharmacotherapy 2009;10:669–676. doi: 10.1517/14656560902762873
[33] Michelson D, Adler L, Spencer T et al. Atomoxetine in adults with ADHD: two randomized, placebo-controlled studies. Biological psychiatry 2003;53:112–120. doi: 10.1016/s0006-3223(02)01671-2
[34] Cheng JY, Chen RY, Ko JS et al. Efficacy and safety of atomoxetine for attention-deficit/hyperactivity disorder in children and adolescents—meta-analysis and meta-regression analysis. Psychopharmacology 2007;194:197–209. doi: 10.1007/s00213-007-0840-x
[35] Weiss MD, Virani A, Wasdell M et al. Atomoxetine in clinical practice. Future Neurology 2006;1:249–258. doi: 10.2217/14796708.1.3.249
[36] Garnock-Jones KP & Keating GM. Atomoxetine: a review of its use in attention-deficit hyperactivity disorder in children and adolescents. Pediatric Drugs 2009;11:203–226. doi: 10.2165/00148581-200911030-00005
[37] Spencer TJ, Sallee FR, Gilbert DL et al. Atomoxetine treatment of ADHD in children with comorbid Tourette syndrome. Journal of attention disorders 2008;11:470–481. doi: 10.1177/1087054707306109
[38] Donnelly C, Bangs M, Trzepacz P et al. Safety and tolerability of atomoxetine over 3 to 4 years in children and adolescents with ADHD. Journal of the American Academy of Child & Adolescent Psychiatry 2009;48:176–185. doi: 10.1097/CHI.0b013e318193060e
[39] Bloch MH, Panza KE, Landeros-Weisenberger A et al. Meta-analysis: treatment of attention-deficit/hyperactivity disorder in children with comorbid tic disorders. Journal of the American Academy of Child & Adolescent Psychiatry 2009;48:884–893. doi: 10.1097/CHI.0b013e3181b26e9f
[40] Hammerness P, McCarthy K, Mancuso E et al. Atomoxetine for the treatment of attention-deficit/hyperactivity disorder in children and adolescents: a review. Neuropsychiatric disease and treatment 2009;5:215–226. doi: 10.2147/NDT.S3896
[41] Biederman J, Melmed RD, Patel A et al. A randomized, double-blind, placebo-controlled study of guanfacine extended release in children and adolescents with attention-deficit/hyperactivity disorder. Pediatrics 2008;121:e73–e84. doi: 10.1542/peds.2006-3695
[42] Taylor FB & Russo J. Comparing guanfacine and dextroamphetamine for the treatment of adult attention-deficit/hyperactivity disorder. Journal of clinical psychopharmacology 2001;21:223–228. PMID: 11270920
[43] Connor DF, Fletcher KE & Swanson JM. A meta-analysis of clonidine for symptoms of attention-deficit hyperactivity disorder. Journal of the American Academy of Child & Adolescent Psychiatry 1999;38:1551–1559. doi: 10.1097/00004583-199912000-00017
[44] Greenhill LL, Pliszka S & Dulcan MK. Practice parameter for the use of stimulant medications in the treatment of children, adolescents, and adults. Journal of the American Academy of Child & Adolescent Psychiatry 2002;41:26S–49S. doi: 10.1097/00004583-200202001-00003
[45] Faraone SV & Buitelaar J. Comparing the efficacy of stimulants for ADHD in children and adolescents using meta-analysis. European child & adolescent psychiatry 2010:19;353–364. doi: 10.1007/s00787-009-0054-3
[46] Prince J, Wilens T, Spencer T et al. Pharmacotherapy of ADHD in adults. In: Adler LA, Spencer TJ, Wilens TE (eds) Attention-Deficit Hyperactivity Disorder in Adults and Children. Cambridge, UK: Cambridge University Press 2014:276-297.
[47] Scheffler RM, Brown TT, Fulton BD et al. Positive association between attention-deficit/hyperactivity disorder medication use and academic achievement during elementary school. Pediatrics 2009;123;1273–1279. doi: 10.1542/peds.2008-1597
[48] Volkow ND & Swanson JM. Does childhood treatment of ADHD with stimulant medication affect substance abuse in adulthood? The American journal of psychiatry 2008;165:553–555. doi: 10.1176/appi.ajp.2008.08020237
[49] Wilens TE, Faraone SV, Biederman J et al. Does stimulant therapy of attention-deficit/hyperactivity disorder beget later substance abuse? A meta-analytic review of the literature. Pediatrics 2003;111:179–185. PMID: 12509574
[50] Molina BS & Pelham Jr WE. Childhood predictors of adolescent substance use in a longitudinal study of children with ADHD. Journal of abnormal psychology 2003;112:497–507. doi: 10.1037/0021-843X.112.3.497
[51] Mannuzza S, Klein RG, Truong NL et al. Age of methylphenidate treatment initiation in children with ADHD and later substance abuse: prospective follow-up into adulthood. The American journal of psychiatry 2008;165:604–609. doi: 10.1176/appi.ajp.2008.07091465
[52] Barkley RA, McMurray MB, Edelbrock CS et al. Side effects of metlyiphenidate in children with attention deficit hyperactivity disorder: a systemic, placebo-controlled evaluation. Pediatrics 1990;86:184–192. PMID: 2196520
[53] Efron D, Jarman F, Barker M. Side effects of methylphenidate and dexamphetamine in children with attention deficit hyperactivity disorder: a double-blind, crossover trial. Pediatrics 1997;100:662–666. PMID: 9310521
[54] Warren A, Hamilton RM, Belanger SA et al. Cardiac risk assessment before the use of stimulant medications in children and youth: A joint position statement by the Canadian Paediatric Society, the Canadian Cardiovascular Society, and the Canadian Academy of Child and Adolescent Psychiatry. Canadian Journal of Cardiology 2009;25:625–630. doi: 10.1016/S0828-282X(09) 70157-6
[55] Hammerness PG, Perrin JM, Shelley-Abrahamson R et al. Cardiovascular risk of stimulant treatment in pediatric attention-deficit/hyperactivity disorder: update and clinical recommendations. Journal of the American Academy of Child & Adolescent Psychiatry 2011;50:978–990. doi: 10.1016/j.jaac.2011.07.018
[56] Swanson JM, Elliott GR, Greenhill LL et al. Effects of stimulant medication on growth rates across 3 years in the MTA follow-up. Journal of the American Academy of Child & Adolescent Psychiatry 2007;46:1015–1027. doi: 10.1097/chi.0b013e3180686d7e
[57] Lipkin PH, Goldstein IJ & Adesman AR. Tics and dyskinesias associated with stimulant treatment in attention-deficit hyperactivity disorder. Archives of pediatrics & adolescent medicine 1994;148:859-861. doi: 10.1001/archpedi.1994.02170080089017
[58] Mosholder AD, Gelperin K, Hammad TA et al. Hallucinations and other psychotic symptoms associated with the use of attention-deficit/hyperactivity disorder drugs in children. Pediatrics 2009;123:611–616. doi: 10.1542/peds.2008-0185
[59] Chammas M, Ahronheim GA & Hechtman L. Reintroduction of stimulant treatment for patients with ADHD, after stimulant-related psychosis. Clinical Practice 2014;11:289–294. doi: 10.2217/cpr.14.26
[60] Tasman A & Riba M. Psychological management in psychopharmacologic treatment. In: Lieberman J, Tasman A (eds) Psychiatric drug . Philadelphia PA: WB Saunders 2000:242–249.
[61] Wilens TE, Adler LA, Adams J et al. Misuse and diversion of stimulants prescribed for ADHD: a systematic review of the literature. Journal of the American Academy of Child & Adolescent Psychiatry 2008;47:21–31. doi: 10.1097/chi.0b013e31815a56f1
[62] Kaye S & Darke S. The diversion and misuse of pharmaceutical stimulants: what do we know and why should we care? Addiction 2012;107:467–477. doi: 10.1111/j.1360-0443.2011.03720.x
[63] Froehlich TE, McGough JJ & Stein MA. Progress and promise of attention-deficit hyperactivity disorder pharmacogenetics. CNS drugs 2010;24:99–117. doi: 10.2165/11530290-000000000-00000
[64] Alguacil L & Pérez-GarcÃa C. Histamine H3 receptor: a potential drug target for the treatment of central nervous system disorders. Current Drug Targets-CNS & Neurological Disorders 2003;2:303–313. PMID: 14529362
[65] Wallace T & Bertrand D. Importance of the nicotinic acetylcholine receptor system in the prefrontal cortex. Biochemical pharmacology 2013;85:1713–1720. doi: 10.1016/j.bcp.2013.04.001
[66] Stahl SM. Multifunctional drugs: a novel concept for psychopharmacology. CNS spectrums 2009;14:71–73. PMID: 19238121
[67] Masand P. Managing Cognitive Disturbances in Major Depression: An Unmet Need. September 2014. Available from: http://www.emaxhealth.com/11972/managing-cognitive-disturbances-major-depression-unmet-need (accessed on February 2016).
[68] Damgaard T, Larsen DB, Hansen SL et al. Positive modulation of alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors reverses sub-chronic PCP-induced deficits in the novel object recognition task in rats. Behavioural brain research 2010;207:144–150. doi: 10.1016/j.bbr.2009.09.048
[69] Mitchell ES & Neumaier JF. 5-HT 6 receptors: a novel target for cognitive enhancement. Pharmacology & therapeutics 2005;108:320–333. doi: 10.1016/j.pharmthera.2005.05.001
[70] Richter W, Menniti FS, Zhang H-T et al. PDE4 as a target for cognition enhancement. Expert opinion on therapeutic targets 2013;17:1011–1027. doi: 10.1517/14728222.2013.818656


