Doriano Strologo
Before the 1960s, contraception was “absolutely synonymous with men”, says Richard Anderson, co-director of the Centre for Reproductive Health at the University of Edinburgh. Rubber condoms were invented in 1855 and the first vasectomy was performed in 1897. This changed when ‘the pill’ arrived and contraception became largely the preserve of women. But attitudes are changing and, with the overturning of the constitutional right to an abortion by the United States Supreme Court in June 2022, male contraception is again on the agenda. Research into new methods is progressing and those in the field now think that it is a matter of when, not if, men will finally share responsibility for birth control.
Although the pharmaceutical industry has shown scant interest in developing male or female contraceptives for the past 20 years — contributing only 5% of funding (US$13m) from 2018–2020 — this was not always the case[1]. After the development of the female pill, there was work on male alternatives too.
“There was significant interest in the early years of the century,” says Anderson, but through a series of mergers and acquisitions, those working in the area faded away.
Michael O’Rand, chief executive of male contraceptive start-up Eppin Pharma, says fears about causing permanent changes in men’s fertility kept the pharmaceutical industry away, but medicinal chemist Gunda Georg from the University of Minnesota suspects part of the continuing multifaceted problem is that “the pharmaceutical companies are largely run by men”.
The bar to bring any contraceptive to the market is very high, be it male or female
Medicinal chemist Gunda Georg from the University of Minnesota
Given that contraceptives are used by young, healthy people, one issue has always been a low tolerance of side effects.
“The bar to bring any contraceptive to the market is very high, be it male or female,” says Georg.
For male contraceptives, “safety bars have also not been clearly laid out by the US Food and Drug Administration or other regulatory agencies”, says endocrinologist Arthi Thirumalai from the University of Washington School of Medicine, Seattle.
“And that’s the challenge, from the perspective of developing a product.”
The limited renaissance in male contraceptive development has been led by several small start-ups and academic groups, supported by government funding agencies and non-governmental organisations. The non-profit Male Contraceptive Initiative (MCI) was set up in the United States in 2014 and has provided grant funding for male contraceptive development of more than US$7m since 2017 — small fry compared with industry spending.
However, Heather Vahdat, executive director at MCI, points out that early-stage development of most contraceptives has been managed and funded by non-profits before industry got on board.
The hormonal approach
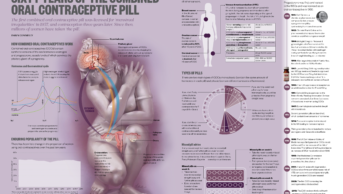
The approach closest to market is one that mirrors the female contraceptive pill in altering sex hormone levels (see Figure). Work on this method started back in the 1950s but a candidate, NES/T — a combination gel of the progestin segesterone acetate (Nestorone) and testosterone, developed by the Population Council and the National Institute of Child Health and Human Development in the United States — has only recently reached advanced clinical trials, which began in 2018[2].
Click here for a full size version of the Figure.
The candidate delivers androgens that block the production of two hormones in the pituitary gland: luteinizing hormone and follicle stimulating hormone, which subsequently inhibits natural testosterone production. “Essentially, if you switch those off, then you switch off spermatogenesis [in the testes],” explains Anderson, who is part of the team running the multi-centre phase II clinical trial. The addition of testosterone to the progestin replaces suppressed natural testosterone in the bloodstream (but not the testes), helping to maintain other functions dependent on the hormone, such as a normal sex drive.
It’s the first time a male hormonal contraceptive has been available by a self-administered method
Richard Anderson, co-director of the Centre for Reproductive Health at the University of Edinburgh
As Nestorone cannot be administered orally, NES/T has been formulated as a gel that is rubbed into the shoulders and upper arms, and absorbed through the skin. “It’s the first time [a male hormonal contraceptive has] been [available by a] self-administered method, rather than you needing to go to the doctor for an injection, which is a really interesting refinement,” says Anderson.
In addition, systemic delivery through the skin has the advantage of lowering the dose needed. The current NES/T clinical trial is being run by the UCLA Medical Centre and University of Washington with investigators in Kansas, Edinburgh, Manchester (UK), Sweden, Kenya, Chile and Italy[2].
Thirumalai is part of the NES/T trial team but is also developing alternative oral formulations in collaboration with colleagues at the University of Washington and the Lundquist Institute in Los Angeles, California. The team has designed two pro-drug candidates, dimethandrolone undecanoate (DMAU) and 11b-methyl-19-nortestosterone-17b-dodecylcarbonate (11b-MNTDC), which are cleaved after ingestion to release the active hormones. “These two drugs actually have this long chain fatty acid attached to them that helps allow them to be dosed once a day,” explains Thirumalai. Both molecules have undergone phase I clinical studies and were shown to be effective as a daily pill[3,4].
As with the female contraceptive pill, there are likely to be some side effects for some individuals from these candidates, but so far not many have been seen in the NES/T study. “Most guys feel absolutely fine,” says Anderson. However, it is too early to say if there are any longer term effects.
“People are very aware of the long-term benefits of female contraceptive methods outside contraception for regulating periods, controlling period pain, reduced anaemia … so we will be intrigued as to whether any of those sort of analogous benefits accrue to male contraception.”
A meta-analysis of all male hormonal contraceptives tested showed that men experience the same side effects some women experience, such as acne, weight gain, changes in mood, changes in sexual function and sex drive, although not the risk of venous thromboembolism seen in women, which is linked with oestrogen effects[5]. In her trials, Thirumalai also found no changes in prostate specific antigen levels in men taking the contraceptive and, generally, the newer regimens being developed seem to have better side effect profiles[4,5].
While the hormonal method has the advantage of many years of research and good evidence that there is no permanent impact on sperm quality, the biggest problem with this approach is the time it takes to work — around 70 days for the spermatogenesis process to shut down completely and another three or four months to restore fertility. “Those two challenges are not going to really disappear,” says Christopher Barratt, a fertility scientist from the University of Dundee, Scotland.
Non-hormonal strategies
Non-hormonal male contraceptives are still some way behind hormonal methods, but they offer the possibility of quicker acting methods of contraception (see Figure, above). There are several targets being investigated, including the retinoic acid receptor alpha (RAR-α), which is involved in sperm formation. Georg and colleagues have demonstrated that knocking out the RAR-α gene makes male mice sterile[6]. The team has now developed a drug that selectively inhibits this receptor. When given orally to male mice for four weeks, the lead compound, YCT529, dramatically reduced sperm counts and was 99% effective in preventing pregnancy, but the mice could father pups again four to six weeks after the compound was withdrawn.
The drug is now being developed by California-based start-up Your Choice Therapeutics, which has started trials in macaque monkeys. “They also observed that sperm counts are going down [in the monkeys] and so that’s really proof of concept in a species that’s much closer to humans … now we’re waiting and hopefully we will see that recovery as well,” says Georg.
Another non-hormonal approach comes from O’Rand, who retired from his academic position at the University of North Carolina at Chapel Hill in 2014 and set up Eppin Pharma to commercialise a male contraceptive based on inhibition of the protein eppin (epididymal protease inhibitor). Eppin coats the surface of sperm throughout the male reproductive tract; blocking it causes immobilisation of the sperm, preventing them from reaching the ovum.
O’Rand’s initial work showed that immunising male monkeys so they created anti-eppin antibodies could make them temporarily infertile[7], but he decided a small molecule drug might provide a more consistent response in users. The team’s lead molecule EP055 has now been successfully tested intravenously on male macaque monkeys and the company is applying for investigational new drug status, with the view to developing an oral dosage form to test in human trials[8].
With this being a novel approach and human trials yet to take place, it is not clear what type of side effects there could be, but O’Rand does not expect to see those associated with hormonal contraceptives, such as loss of libido. “The protein eppin is only found in testes and epididymus and on sperm, so it’s not anywhere else in the body, which means that the chance of off-target effect is quite low. So far, we’ve found that to be true.”
It’s a new concept … if we succeed in getting to the clinic, it’s going to be a first in class for humans
Michael O’Rand, chief executive of male contraceptive start-up Eppin Pharma
“We haven’t run across any hint of any kind of long-term toxicology in our 28-day studies on animals, and are using very high doses.” The drug also seems to act quickly (showing activity in monkeys within a few days) and reversing its effects is also fast. “It’s a new concept … if we succeed in getting to the clinic, it’s going to be a first in class for humans.”
Barrier methods
Not all of the current development of male contraceptives is focused on a pill, though. Several companies are looking at barrier methods that would prevent sperm from entering the urethra by blocking the tubes that carry them (the vas deferens) (see Figure). “It’s like the [coil] for men,” says Kevin Eisenfrats, founder and chief executive of Contraline, based in Virginia, which is developing ADAM, an injectable hydrogel designed to provide men with long-lasting contraception.
Intrauterine devices and other long-acting reversible contraceptives are the fastest growing contraceptive products for women, so should also be attractive to men, Eisenfrats believes. “They’re set and forget … until you want to remove them.”
Our idea was to create something that’s safer and easily injectable and water soluble, and that’s where we came up with using a hydrogel
Kevin Eisenfrats, founder and chief executive of Contraline
The idea is not new, but previous attempts have faltered. “Our idea was to create something that’s safer and easily injectable and water soluble, and that’s where we came up with using a hydrogel.” The biocompatible hydrogel is injected into the vas deferens by a urologist in minutes and, once in place, forms an elastic barrier that can be removed when desired.
The company is not currently disclosing the details of its proprietary removal procedure but says it is “minimally invasive” and that animal studies have shown that, once removed, sperm quality and count return to normal.
First-in-human trials are underway in Australia, with investment from the MCI and others. Another similar product, Vasalgel, developed by the Parsemus Foundation and based on technology developed in India, is also starting clinical trials[9]. Vahdat says: “If everything goes smoothly”, ADAM is likely to be the first new male contraceptive method to reach the market, within five to ten years, “everything else is still probably ten plus years”.
Attitudes shifting
The array of new male contraceptive methods in development indicates male contraception is possible, but will there be a market for these products? In the past, there have been suggestions that men would not take responsibility for birth control and women would not trust them to do so.
“We have made contraceptives inextricably linked with women’s rights and women’s autonomy, and we’ve taken out men’s reproductive involvement completely,” says Vahdat. But, in the past five years, she has seen a shift. “When we talk to men, they will say to us, ‘I wish I had a way that I could protect myself’.”
This business, ‘oh, I wouldn’t trust my man’, is just nonsense
Richard Anderson, co-director of the Centre for Reproductive Health at the University of Edinburgh
From his interactions with couples taking part in the NES/T trial, Anderson also sees equal enthusiasm. “They want to share the burden of contraception and they recognise that it isn’t appropriate for women just to be constantly taking pills or getting implants … and this business, ‘oh, I wouldn’t trust my man’, is just nonsense.”
Several recent surveys back up this view. A 2016 review of multiple attitude surveys carried out over 20 years by the Parsemus Foundation indicated that about 50% of men would be willing to use new male contraceptives — with some variation country to country — and women overwhelmingly responded that they would trust their male partners to use contraception[10]. The MCI has recently commissioned a new global survey of couples, in partnership with the Gates Foundation, to gauge attitudes across eight countries. It is yet to be published but Vahdat hopes the data will help give male contraception “the punch that we need”.
Even with changing attitudes, the pharmaceutical industry still seems reticent to re-enter the arena yet. “The risk for them is just a little too much,” says O’Rand.
“There are not enough assets right now that are far enough along for them to make a big splash … but I think people are starting to see that the market is there,” says Eisenfrats.
And Thiumalai hopes that once NES/T has successfully gone through phase III clinical trials, the bigger players will become interested and it will get the ball rolling for the whole field.
As things stand, Vahdat sees the development of male contraceptives as a great niche for investors looking for disruptive opportunities in the pharmaceutical industry. And for those looking to make a real impact on the world, she says, “I can think of no better impact than reducing the number of unintended pregnancies”.
- 1Total contraceptive funding by funder category. Male Contraceptive Initiative. https://www.malecontraceptive.org/uploads/1/3/1/9/131958006/mci_communications-infographics_whofundscontraceptionrd_landscape-v2.png (accessed 23 Nov 2022).
- 2Study of Daily Application of Nestorone® (NES) and Testosterone (T) Combination Gel for Male Contraception. clinicalstrials.gov. 2018.https://clinicaltrials.gov/ct2/show/NCT03452111 (accessed 23 Nov 2022).
- 3Nguyen BT, Yuen F, Farrant M, et al. Acceptability of the oral hormonal male contraceptive prototype, 11β-methyl-19-nortestosterone dodecylcarbonate (11β-MNTDC), in a 28-day placebo-controlled trial. Contraception. 2021;104:531–7. doi:10.1016/j.contraception.2021.06.009
- 4Thirumalai A, Yuen F, Amory JK, et al. Dimethandrolone Undecanoate, a Novel, Nonaromatizable Androgen, Increases P1NP in Healthy Men Over 28 Days. The Journal of Clinical Endocrinology & Metabolism. 2020;106:e171–81. doi:10.1210/clinem/dgaa761
- 5Fernández-Balsells MM, Murad MH, Lane M, et al. Adverse Effects of Testosterone Therapy in Adult Men: A Systematic Review and Meta-Analysis. The Journal of Clinical Endocrinology & Metabolism. 2010;95:2560–75. doi:10.1210/jc.2009-2575
- 6Noman MAA, Kyzer JL, Chung SSW, et al. Retinoic acid receptor antagonists for male contraception: current status†. Biology of Reproduction. 2020;103:390–9. doi:10.1093/biolre/ioaa122
- 7O’Rand MG, Widgren EE, Sivashanmugam P, et al. Reversible Immunocontraception in Male Monkeys Immunized with Eppin. Science. 2004;306:1189–90. doi:10.1126/science.1099743
- 8O’Rand MG, Hamil KG, Adevai T, et al. Inhibition of sperm motility in male macaques with EP055, a potential non-hormonal male contraceptive. PLoS ONE. 2018;13:e0195953. doi:10.1371/journal.pone.0195953
- 9Brent L. NEXT steps for Vasalgel. Parsemus Foundation. 2022.https://www.parsemus.org/2022/07/next-steps-for-vasalgel/ (accessed 23 Nov 2022).
- 10Male Contraception Attitudes: Summary of Surveys and Research. Parsemus Foundation. 2016.https://www.parsemus.org/wp-content/uploads/2016/03/Male-Contraception-Attitudes-Surveys-and-Research-7-8-16.pdf (accessed 23 Nov 2022).