
nobeastsofierce / Shutterstock.com
- Describe the current CAR-T treatment landscape and potential future therapeutic areas;
- Describe the differences between the acute toxicities and delayed effects of CAR-T cell therapy in terms of presentation and pharmacological management;
- Describe the importance of shared care between cell therapy treatment centres and local teams;
- Outline the referral triggers and escalation pathways in place for management of late toxicities identified by non-specialist teams.
Chimeric antigen receptor (CAR) T-cell therapy is an advanced form of adoptive cellular immunotherapy in which autologous (i.e. the patient’s own) T lymphocytes are genetically engineered to express synthetic receptors, targeting specific tumour antigens1. These autologous T-cells are modified ex vivo and reinfused to mediate targeted cytotoxicity against malignant cells – a treatment strategy that has demonstrated improved progression-free survival, overall survival and cancer remission rates for multiple haematological malignancies1. Other T-lymphocyte-based therapies have shown potential for improved outcomes in solid tumour malignancy1. Building on this, clinical trials are now exploring their application in non-malignant diseases, notably autoimmune conditions2.
Despite the positive outcomes of CAR T-cell therapy, it comes with the caveat of significant toxicities, such as neurotoxicity, infections and cytokine release syndrome (CRS), which are described in more detail below.
This article aims to equip non-specialist healthcare professionals (HCPs) with practical knowledge on recognising and responding to toxicities following CAR T-cell therapy, with a particular focus on delayed effects, shared-care responsibilities, long-term follow up and escalation pathways. It should be read alongside other advanced therapy medicinal product (ATMP) articles from The Pharmaceutical Journal, see Box 13–5.
Box 1: Previous advanced therapy medicinal product articles from The Pharmaceutical Journal
Current and emerging CAR T-cell therapies
The NHS funds several CAR T-cell therapies, all of which target CD19, see Box 26–12.
Box 2: CAR T-cell therapies available through the NHS (accurate at time of publication)
- Tisagenlecleucel (Kymriah);
- Paediatric and young adult (≤25 years) B-cell acute lymphoblastic leukaemia (B-ALL);
- Axicabtagene ciloleucel (Yescarta);
- Adult large B-cell lymphoma (LBCL);
- Brexucabtagene autoleucel (Tecartus);
- Adult mantle cell lymphoma (MCL);
- B-ALL in adults aged ≥26 years;
- Lisocabtagene maraleucel (Breyanzi)
- Adult LBCL;
- Obecabtagene autoleucel (Aucatzyl).
- B-ALL in adults aged ≥26 years.
Many other CAR-T products are in development, with more than 1,500 active clinical trials registered globally1. Efforts focus on improving efficacy, safety and manufacturing logistics1.
Why non-specialist teams need awareness of CAR-T products
Non-specialist teams in smaller district general hospitals and primary care settings should be aware of CAR T-cell therapy and its longer-term toxicity risks13. As more patients return to local services after receiving treatment at specialist cell therapy treatment centres, they may present with delayed or chronic complications that require prompt recognition and management outside tertiary settings13.
These complications can include, but are not limited to, prolonged cytopenias (i.e. decreased blood cell counts), late infections and delayed neurotoxicity, which may manifest months to years after infusion and can be mistaken for other conditions if clinicians are unaware of the patient’s CAR T-cell history. Timely recognition and intervention for immune-related adverse events, including CRS, neurotoxicity, hemophagocytic lymphohistiocytosis (i.e. lymphocytes and macrophages attack the body’s own tissues and organs), and infection risk, can reduce morbidity and mortality14.
Recent relaxation of the US Food and Drug Administration’s (FDA’s) risk evaluation and mitigation strategies (REMS) requirements for CAR T-cell therapies, together with updates to CAR-T manufacturer guidance, supporting more flexible outpatient monitoring pathways, reflects a broader international shift toward decentralised follow-up care15,16, as well as underscoring the need for all clinicians involved in longitudinal patient management to be familiar with CAR-T-related toxicity management13.
Acute toxicities of CAR T-cell therapy
CAR T-cell therapy is associated with a range of serious and potentially life-threatening toxicities that require prompt recognition and accurate clinical assessment, as delays or suboptimal management can significantly increase the risk of harm13,14.
In clinical practice, adverse events associated with CAR T-cell therapies are often categorised as acute or delayed, based on their timing relative to infusion, although this temporal framework does not always reflect the underlying pathophysiology, see Figure 114,17. In addition, lymphodepleting chemotherapy (LDC), most commonly fludarabine and cyclophosphamide administered in the days preceding infusion to optimise CAR T-cell expansion and persistence, contributes its own spectrum of adverse effects, such as neutropenia, thrombocytopenia and increased infection risk, which may overlap with or exacerbate CAR T-cell-related toxicities14,18–20.
Figure 1: Approximate timeline of selected CAR T-cell therapy-related adverse events
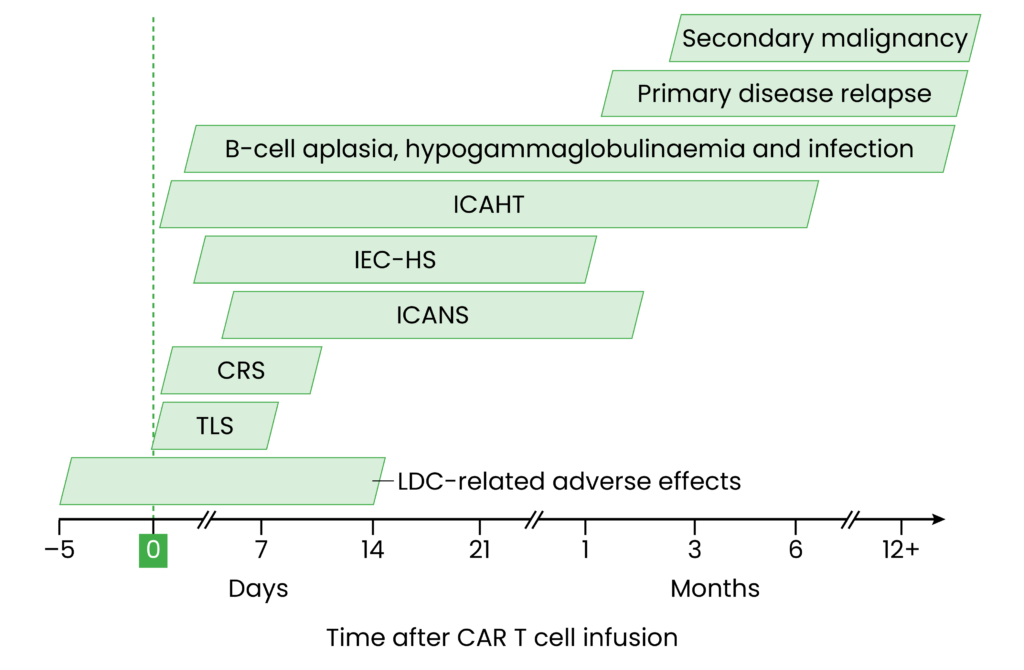
CRS: cytokine release syndrome; ICAHT: immune effector cell-associated haematotoxicity; ICANS: immune effector cell-associated neurotoxicity syndrome; IEC-HS: immune effector cell-associated haemophagocytic lymphohistiocytosis-like syndrome; LDC: lymphodepleting chemotherapy; TLS: tumour lysis syndrome
Figure adapted from: Brudno JN, Kochenderfer JN. Nat Rev Clin Oncol. 2024; 21(7): 501-21. and Jain T., et al. Blood. 2023; 141(2): 2460-68.
Acute CAR T-cell-related toxicities typically emerge within the first month following infusion. Their incidence and severity vary by product, underlying disease and individual patient factors. These systemic immune-mediated complications arise from CAR T-cell activation, leading to sustained cytokine release and immune dysregulation13,14,17. In addition, on-tumour effects, such as tumour lysis syndrome (TLS), may also occur during this period18. While recognition and management primarily sit with specialist teams, broader awareness among non-specialist HCPs is essential to support continuity of care, particularly when patients present outside designated cell therapy treatment centres — for example, in accident and emergency departments, GP practices or community pharmacies.
Cytokine release syndrome
CRS is the most common early toxicity of CAR T-cell therapy, occurring in 70–90% of recipients21. It reflects a progressive systemic inflammatory response driven by rapid CAR T-cell activation and proliferation, resulting in a large increase in the release of pro-inflammatory cytokines, particularly interleukin-6 (IL-6). This cytokine surge activates bystander immune and non-immune cells, further amplifying inflammation21–23.
CRS typically presents within two to three days of infusion, with severity ranging from mild flu-like symptoms to life-threatening multiorgan failure. Hallmark features – such as fever, hypotension and hypoxia – guide clinical assessment and grading13,21–23.
First-line treatment for moderate-to-severe CRS is the IL-6 receptor antagonist, tocilizumab, which is nationally commissioned for this indication, with funding approved for up to four doses per patient18. Corticosteroids and other anti-cytokine therapies (e.g. anakinra, siltuximab) are reserved for cases refractory to tocilizumab or for higher-grade CRS, although clinical trial data is lacking13,18,21. Supportive care, including antipyretics, broad-spectrum antibiotics and intravenous fluids is essential18,21. Around one-quarter of patients require intensive care for severe CRS, including vasopressor support and mechanical ventilation24.
Immune effector cell-associated neurotoxicity syndrome
Immune effector cell-associated neurotoxicity syndrome (ICANS) is a distinct complication of CAR T-cell therapy, occurring in 20–40% of recipients25. Its multifactorial pathophysiology involves blood-brain barrier disruption, cytokine-driven endothelial activation and immune cell trafficking into the central nervous system (CNS), resulting in neuroinflammation, coagulopathy and neuronal dysfunction13,23,25.
ICANS typically presents within four to ten days of infusion, often overlapping with or following CRS. Clinical features range from encephalopathy, aphasia and cognitive impairment to motor weakness, seizures, and, in rare cases, cerebral oedema and death13,23,25.
Although most cases occur acutely, a minority of patients develop delayed-onset ICANS, which emerges after the resolution of CRS and within the first month of CAR-T infusion. Symptoms mirror those of acute ICANS and may fluctuate or recur after apparent improvement or persist as subacute cognitive dysfunction26,27. Episodes are usually self-limiting, with symptoms lasting 5–17 days26.
ICANS is a major cause of morbidity in CAR T-cell therapy and requires prompt recognition and intervention. Management relies on corticosteroids titrated to severity, alongside supportive care including levetiracetam for seizure prophylaxis13,18,23,25. Between 17–35% of recipients require intensive care support.
With timely intervention, ICANS is reversible, although severe cases may lead to prolonged deficits, or rarely, fatal outcomes. Long-term follow-up is therefore warranted25. In addition, manufacturers advise that all patients undergoing CAR T-cell therapy should refrain from driving or operating heavy machinery for at least eight weeks post-infusion, regardless of whether neurotoxicity occurs16.
Immune effector cell-associated haemophagocytic lymphohistiocytosis-like syndrome
Immune effector cell-associated hemophagocytic lymphohistiocytosis-like syndrome (IEC-HS) is a rare but potentially fatal hyperinflammatory toxicity associated with CAR T-cell therapy, occurring in up to 3.5% of recipients13. It is driven by excessive activation of T-cells and macrophages, leading to a cytokine storm and multiorgan dysfunction21,28.
Diagnosis is challenging, as IEC-HS overlaps clinically and temporally with CRS and ICANS13,28. Clinical and laboratory features resemble classical hemophagocytic lymphohistiocytosis (HLH), including persistent fever, cytopenia, hyperferritinaemia, hypofibrinogenaemia, transaminitis, coagulopathy and evidence of hemophagocytosis13,21,28.
Prompt recognition and treatment are essential, as IEC-HS can progress to multiorgan failure and death13,21,28. First-line therapy consists of corticosteroids, with cytokine-targeted agents, such as anakinra (IL-1 blockade) and ruxolitinib (JAK inhibition), considered in refractory cases13,21,28.
Sepsis
Sepsis is a determinant of hospitalisation and non-relapse mortality in CAR T-cell recipients29,30. It most commonly occurs within the first month following therapy, with the highest risk during the initial 10–14 days post-infusion, coinciding with neutropenia and acute immune effector cell (IEC)-associated toxicities1,22,29. Reported incidence ranges from 5–32%, with bacterial infection predominating1,29. Although sepsis represents the principal early infectious presentation, other early infections, including herpesvirus reactivation may also occur31.
Clinical presentation may be indistinguishable from CRS and other IEC-associated toxicities, making diagnosis challenging. Careful clinical assessment, microbiological evaluation and prompt initiation of empirical broad-spectrum antibiotics are essential to reduce morbidity and mortality18,21,31.
Delayed effects of CAR T-cell therapy
The principal late complications of CAR-T therapy are outlined below. Compared with early toxicities, late effects are less well characterised and inconsistently reported32,33.
Prolonged neurologic symptoms following ICANS
While ICANS is typically self-limited, around 27% of patients who develop ICANS experience prolonged neurologic deficits that persist beyond four weeks26. Clinical manifestations include impairments in short-term and long-term memory, aphasia and tremor26,27.
Risk factors for prolonged neurological symptoms are not fully established, although high-grade (grade ≥3) acute neurotoxicity has been consistently associated with incidence, affecting 42% of this subgroup, compared with only 7% of those with low-grade ICANS26.
Management of prolonged symptoms follows standard ICANS treatment algorithms, with supportive care and ongoing monitoring recommended26,27.
Non-ICANS neurological complications
Although no BCMA-directed CAR T-cell therapies are available within the NHS, it is important to note that they are associated with distinct non-ICANS delayed neurological complications, including IEC-Parkinsonism and cranial nerve palsies. These complications are considered a class effect of BCMA-targeting, with the mechanism thought to involve on-target CAR T-cell activity within the CNS, although this is not yet fully established34.
Immune effector cell-associated haematotoxicity and infection risk
Cytopenias, particularly neutropenia, are the most common high-grade adverse events following CAR T-cell therapy. Although the underlying pathophysiology remains unclear, the pattern suggests a class-wide effect, characterised by profound and persistent cytopenia that may last for months to years after infusion, often with periods of intermittent count recovery31,35. Prolonged grade 3–4 cytopenias occur in 30–40% of patients beyond Day +30 and there are varied reports in 3–22% of patients beyond Day +90 after CAR-T infusion36. This clinical phenotype is recognised as immune effector cell-associated haematotoxicity (ICAHT), a distinct entity with its own consensus-driven grading system and a corresponding severity-based management algorithm31,35,37.
The risk of immunosuppression following CAR T-cell therapy is multifactorial and can predispose patients to severe infectious complications and inferior survival, with infections being the leading cause of non-relapse mortality31,35. Although the most severe infections occur within the first 28 days, late infections, particularly upper respiratory viral infections, are frequently observed months to years after CAR-T therapy. While ongoing care is typically coordinated between the cell therapy treatment centre and referring haematology teams, patients may present acutely to local services, making awareness of their prolonged immunocompromised status essential for non-specialist clinicians. Late infections can result in repeated hospitalisation, increased healthcare contact and poorer quality of life. Although uncommon, life-threatening invasive fungal infections have also been reported32. The use of granulocyte-colony stimulating factor (G-CSF) and anti-infective prophylaxis is therefore recommended, with duration typically guided by the extent of immune reconstitution. Management of suspected or confirmed infections should follow institutional guidelines18,31, see Box 326,31.
Box 3: Post-CAR T-cell immunosuppression risk factors
- Prolonged neutropenia;
- Long-term CD4 T-cell lymphopenia;
- B-cell aplasia with hypogammaglobulinaemia;
- Grade ≥3 cytokine release syndrome / immune effector cell-associated neurotoxicity syndrome;
- Extended use of immunosuppressive agents (e.g. high-dose corticosteroids).
B-cell aplasia and hypogammaglobulinaemia are recognised ‘on-target/off-tumour’ complications of CAR T-cell therapy directed against B-lineage antigens, occurring in 44–53% of patients — increasing susceptibility to infection and necessitating coordinated long-term follow-up between treating and referral centres31,36. Recovery patterns are variable but often prolonged, frequently extending beyond six months and requiring immunoglobulin replacement therapy (IGRT)18,31,37, which is nationally commissioned for patients with secondary immunodeficiency following CAR-T therapy38. Vaccination forms a vital part of long-term immune reconstitution, as discussed below.
Primary disease relapse and secondary malignancies
Primary disease relapse remains the most common cause of treatment failure after CAR-T therapy, occurring in about 40–60% of patients within 1–2 years, with rates varying by disease, CAR construct and patient-specific factors. Relapse may arise from antigen loss, inadequate CAR-T expansion or persistence, or aggressive underlying disease biology. Patients who relapse often require further immunosuppressive therapy, contributing to cumulative marrow toxicity and an increased risk of infection1,36. Ongoing surveillance is therefore essential to detect relapse early and guide subsequent management39.
Secondary malignancies have a cumulative incidence of up to 6.5% at 3 years40. Most are myeloid neoplasms, with smaller proportions of solid tumours; secondary T-cell malignancies are rare, occurring in about 0.1% of patients41,42. Risk appears related to older age, multiple prior cytotoxic therapies, clonal haematopoiesis and longer follow-up, and does not appear higher than in comparable populations receiving other standard therapies. Cases are typically detected through routine surveillance27,41,42. Although isolated reports of CAR transgene-positive T-cell cancers have been described, causality remains unproven. Nonetheless, regulatory agencies have advised lifelong monitoring42–44.
Supportive care and patient empowerment
Supportive care measures, including individualised risk assessment, infection prophylaxis and immunoglobulin replacement are essential for mitigating long-term risks45.
Vaccination guidance
UK vaccination guidelines recommend that all routine immunisations be repeated after CAR T-cell therapy, regardless of prior vaccination status46. Influenza and COVID-19 vaccines may be administered from three months post-infusion, while other non-live (inactivated) vaccines should generally be deferred until at least six months after therapy for optimal immunogenicity46. Guidance suggests an optimal window of 6–18 months owing to prolonged B-cell aplasia and hypogammaglobulinemia, which can impair vaccine responses46.
Patients with persistent hypogammaglobulinemia or ongoing B-cell–depleting therapies may mount reduced responses, but vaccination remains recommended to lower the risk of severe infection46.
Live and live-attenuated vaccines are contraindicated for at least two years following CAR-T therapy and should only be considered if there is no ongoing immunosuppression or B-cell dysfunction46.
These recommendations reflect the high risk of infection and loss of pre-existing immunity after CAR-T therapy and the need to optimise protection despite potentially reduced vaccine efficacy46.
Red-flag symptoms and skilled-task restriction counselling
Severe toxicities, particularly within the first eight weeks post-infusion, require vigilant monitoring and clear patient education about early and late effects. Red-flag symptoms requiring urgent medical attention during this period are summarised below, see Box 414.
Box 4: Red-flag symptoms requiring urgent medical attention
- Suggestive of cytokine release syndrome: fever, hypotension, tachycardia or hypoxia;
- Suggestive of immune effector cell-associated neurotoxicity syndrome: new or worsening confusion, aphasia, seizures, motor weakness, reduced alertness, cognitive and/or language disturbances;
- Suggestive of cytopenias or disseminated intravascular coagulation: unexplained bleeding, bruising or petechiae;
- Suggestive of infection: fever, rigors, persistent cough or dyspnoea, chest pain, urinary symptoms, gastrointestinal symptoms, skin changes or unexplained clinical deterioration.
Skilled-task restrictions, such as driving or operating heavy machinery, require careful consideration in all CAR T-cell therapy recipients. In the UK, manufacturer summary of product characteristics (SmPC) advise that patients avoid skilled tasks for at least eight weeks post-infusion, regardless of whether neurotoxicity occurs, or until any neurologic adverse events have fully resolved18. In practice, consensus guidance emphasises that restrictions should be individualised, particularly in the presence of neurotoxicity or other adverse effects that may impair a patient’s senility to safely perform skilled tasks13.
Patient and caregiver education should include written instructions and emergency contact information, emphasising the importance of promptly reporting symptoms to the cell therapy treatment centre or specialist team rather than attempting to self-manage red-flag concerns13. The optimal duration and criteria for lifting skilled-task restrictions remain under investigation, as late-onset neurotoxicity and other adverse effects may occur beyond the initial monitoring window13.
The use of alert cards or medical bracelets, carried at all times after receiving CAR‑T therapy, helps ensure rapid recognition of a patient’s treatment history by HCPs and supports timely escalation during acute illness. These tools also empower patients to notify HCPs early if symptoms or concerns arise13.
Shared care
Survivorship care plans (SCPs) are recommended to support communication and individualised follow-up between cell therapy treatment centres, referring teams and primary care. SCPs should outline the CAR-T product administered, anticipated toxicities, recommended surveillance intervals and clear triggers for referral13. Patients are followed for up to 15 years as part of post-authorisation safety surveillance (PASS) requirements, including registry participation, to monitor long-term safety and outcomes13,44.
Non-specialist teams play a critical role in long-term monitoring, early recognition of late toxicities and coordination of care. A multidisciplinary approach is essential, with regular updates from the cell therapy treatment centre, defined referral pathways for acute or late complications, as well as shared protocols for surveillance and supportive care13,27,44. Non-specialist clinicians should be prepared to promptly refer patients with hematologic, infectious or neurological complications, and to collaborate closely with specialist teams in the management of complex, late effects13,27.
Referral triggers and escalation pathways
Late complications resulting from CAR-T therapy should be escalated to the patient’s cell therapy treatment centre. In the UK, patients are monitored intensively during the initial post-infusion period, with clear pathways for managing acute toxicities27,33,44. After discharge, patients remain within proximity to the cell therapy treatment centre until at least Day +30 and are advised to return immediately if they develop fever, neurological symptoms or other signs of toxicity47.
Long-term follow-up is delivered through shared-care arrangements between commissioned cell therapy treatment centres and local haematology teams, enabling regular, systematic and longitudinal surveillance27,33,44. Late complications may present with subtle or non-specific symptoms, so a low threshold for seeking specialist advice is advised13,33. All patients should carry a product-specific CAR-T alert card to support rapid recognition and escalation27. Supportive care, including anti-infective prophylaxis, IGRT and vaccination schedules, should be communicated and managed jointly. Local teams should ensure they have access to the cell therapy treatment centre’s 24-hour contact details for urgent advice27,47.
Clear referral triggers and standard escalation pathways between the cell therapy treatment centre, the referring haematology team and non-specialist services are essential to ensure timely communication and multidisciplinary assessment whenever late complications are suspected27,33,41. Most cell therapy treatment centres run dedicated late-effects clinics and use SCPs to outline monitoring schedules and coordination between HCPs27,33. Adverse effects should be reported to the Medicines and Healthcare products Regulatory Agency (MHRA) via the Yellow Card Scheme, and ongoing surveillance contributes to national and international registries, see Box 527,33,41,44.
Box 5: Referral triggers for late complications or treatment failure after CAR T-cell therapy
- New or worsening cytopenias;
- Recurrent or severe infection;
- Clinical or laboratory evidence of relapse;
- Suspicion of secondary malignancy;
- Persistent neurological symptoms;
- Unexplained fatigue.
Pharmacy best practice considerations for CAR-T therapy
- Ensure clear identification of Chimeric antigen receptor (CAR) T-cell recipients within pharmacy systems to prevent inadvertent dispensing of contraindicated medications (e.g. corticosteroids unless specifically indicated for toxicity management)4;
- Maintain readily available stock of tocilizumab (minimum two doses per patient) for rapid intervention in cytokine release syndrome (CRS), in line with institutional protocols and international consensus46,47;
- Anticipate requests for anakinra or siltuximab in refractory CRS or neurotoxicity, and ensure protocols for rapid access, approval and dispensing are in place48;
- Support antimicrobial prophylaxis and immunoglobulin replacement therapy for patients with persistent cytopenias or hypogammaglobulinemia, in collaboration with the clinical team46,48,49;
- Reinforce vaccination guidance of administering inactivated vaccines according to local and national schedules; live vaccines remain contraindicated until immune reconstitution is confirmed46;
- Participate in pharmacovigilance and monitoring for late toxicities, including cytopenias, infections and neurotoxicity, ensuring adverse events are documented and reported according to institutional and national standards48,50;
- Collaborate with the multidisciplinary team to standardise operating procedures for CAR-T therapy, including drug ordering and traceability, handing and storage, dispensing and preparation, administration and outcome monitoring50–52;
- Ensure pharmacy staff are trained in CAR-T toxicity recognition and management, and are familiar with institutional protocols for supportive therapies and emergency interventions51,52. Educational resources include:
- Advanced therapy medicinal product (ATMP) resources authored by the Pharmacy ATMP Network UK (PAN-UK), hosted on the Specialist Pharmacy Service website53;
- BOPA SACT Verification Hub54, including the competency-based CAR-T passport;
- NHS England e-Learning for Healthcare (elfh) ATMP modules on immune effector cell and CAR-T therapy55.
- 1.Brudno JN, Maus MV, Hinrichs CS. CAR T Cells and T-Cell Therapies for Cancer. JAMA. 2024;332(22):1924. doi:10.1001/jama.2024.19462
- 2.Chung JB, Brudno JN, Borie D, Kochenderfer JN. Chimeric antigen receptor T cell therapy for autoimmune disease. Nat Rev Immunol. 2024;24(11):830-845. doi:10.1038/s41577-024-01035-3
- 3.McKenzie C, Chappell J, Guy R. Advanced therapy medicinal products: a comprehensive overview for pharmacy professionals. Pharmaceutical Journal . 2022. https://pharmaceutical-journal.com/article/ld/advanced-therapy-medicinal-products-a-comprehensive-overview-for-pharmacy-professionals
- 4.Palmer R, Evans N. Medication restrictions for patients receiving CAR-T therapy. Pharmaceutical Journal. 2022. https://pharmaceutical-journal.com/article/ld/medication-restrictions-for-patients-receiving-car-t-therapy
- 5.Black A, Gabriel S, Caulfield D. Implementing chimeric antigen receptor T-cell therapy in practice professionals. Pharmaceutical Journal . 2020. https://pharmaceutical-journal.com/article/ld/implementing-chimeric-antigen-receptor-t-cell-therapy-in-practice
- 6.Tisagenlecleucel for treating relapsed or refractory B-cell acute lymphoblastic leukaemia in people 25 years and under. National Institute for Health and Care Excellence . 2024. https://www.nice.org.uk/guidance/ta975
- 7.Axicabtagene ciloleucel for treating relapsed or refractory diffuse large B-cell lymphoma after first-line chemoimmunotherapy. Technology appraisal guidance [TA895]. National Institute for Health and Care Excellence . 2023. https://www.nice.org.uk/guidance/ta895
- 8.Axicabtagene ciloleucel for treating diffuse large B-cell lymphoma and primary mediastinal large B-cell lymphoma after 2 or more systemic therapies. Technology appraisal guidance [TA872]. National Institute for Health and Care Excellence . 2023. https://www.nice.org.uk/guidance/ta872
- 9.Brexucabtagene autoleucel for treating relapsed or refractory mantle cell lymphoma. Technology appraisal guidance [TA677]. National Institute for Health and Care Excellence . 2021. https://www.nice.org.uk/guidance/ta677
- 10.Brexucabtagene autoleucel for treating relapsed or refractory B-cell acute lymphoblastic leukaemia in people 26 years and over. Technology appraisal guidance [TA893]. National Institute for Health and Care Excellence . 2023. https://www.nice.org.uk/guidance/ta893
- 11.Lisocabtagene maraleucel for treating relapsed or refractory large B-cell lymphoma after first-line chemoimmunotherapy when a stem cell transplant is suitable. Technology appraisal guidance [TA1048]. National Institute for Health and Care Excellence . 2025. https://www.nice.org.uk/guidance/ta1048
- 12.Obecabtagene autoleucel for treating relapsed or refractory B-cell precursor acute lymphoblastic leukaemia. Technology appraisal guidance [TA1116]. National Institute for Health and Care Excellence . 2025. https://www.nice.org.uk/guidance/ta1116
- 13.Santomasso BD, Nastoupil LJ, Adkins S, et al. Management of Immune-Related Adverse Events in Patients Treated With Chimeric Antigen Receptor T-Cell Therapy: ASCO Guideline. JCO. 2021;39(35):3978-3992. doi:10.1200/jco.21.01992
- 14.Brudno JN, Kochenderfer JN. Current understanding and management of CAR T cell-associated toxicities. Nat Rev Clin Oncol. 2024;21(7):501-521. doi:10.1038/s41571-024-00903-0
- 15.FDA eliminates risk evaluation and mitigation strategies (REMS) for autologous chimeric antigen receptor CAR T cell immunotherapies. U.S. Food and Drug Administration . 2025. https://www.fda.gov/news-events/press-announcements/fda-eliminates-risk-evaluation-and-mitigation-strategies-rems-autologous-chimeric-antigen-receptor
- 16.Yescarta- Summary of Product Characteristics . Gilead Sciences Ltd. Yescarta . 2025. https://www.medicines.org.uk/emc/product/9439/smpc
- 17.Epperly R, Giordani VM, Mikkilineni L, Shah NN. Early and Late Toxicities of Chimeric Antigen Receptor T-Cells. Hematology/Oncology Clinics of North America. 2023;37(6):1169-1188. doi:10.1016/j.hoc.2023.05.010
- 18.Tam A, Gabriel S. Diagnosis and medical management of acute CAR-T cell side effects in adults: version 2. Specialist Pharmacy Service. 2022. https://www.sps.nhs.uk/articles/diagnosis-and-medical-management-of-acute-car-t-cell-toxicities-in-adults/
- 19.Ruggeri A, Boissel N, Rabian F. 39 – lymphodepleting conditioning regimens. In: Aljurf M, Bonini C, Chabannon C, et al. The EU CAR-T handbook. EBMT. 2024. https://www.ebmt.org/eu-car-t-handbook
- 20.Lickefett B, Chu L, Ortiz-Maldonado V, et al. Lymphodepletion – an essential but undervalued part of the chimeric antigen receptor T-cell therapy cycle. Front Immunol. 2023;14. doi:10.3389/fimmu.2023.1303935
- 21.Ayuketang F, Jager U, Sanderson R. 40 – management of CRS and IEC-associated haemophagocytic lymphohistiocytosis-like syndrome (IEC-HS). In: Aljurf M, Bonini C, Chabannon C, et al. The EU CAR-T handbook. EBMT. 2024. https://www.ebmt.org/eu-car-t-handbook
- 22.Shimabukuro-Vornhagen A, Gödel P, Subklewe M, et al. Cytokine release syndrome. j immunotherapy cancer. 2018;6(1). doi:10.1186/s40425-018-0343-9
- 23.Morris EC, Neelapu SS, Giavridis T, Sadelain M. Cytokine release syndrome and associated neurotoxicity in cancer immunotherapy. Nat Rev Immunol. 2021;22(2):85-96. doi:10.1038/s41577-021-00547-6
- 24.Azoulay É, Castro P, Maamar A, et al. Outcomes in patients treated with chimeric antigen receptor T-cell therapy who were admitted to intensive care (CARTTAS): an international, multicentre, observational cohort study. The Lancet Haematology. 2021;8(5):e355-e364. doi:10.1016/s2352-3026(21)00060-0
- 25.Rees J, Velasco R, Schoberl F. 41 – Management of IEC-associated neurotoxicity syndrome (ICANS) and non-ICANS neurotoxicity. In: Aljurf M, Bonini C, Chabannon C, et al. The EU CAR-T handbook. EMBT. 2024. https://www.ebmt.org/eu-car-t-handbook
- 26.Maillie L, Nasta SD, Svoboda J, et al. Prolonged Neurologic Symptoms Following Immune Effector Cell-Associated Neurotoxicity Syndrome in Patients With Large B-cell Lymphoma Treated With Chimeric Antigen Receptor-Modified T Cell Therapy. Transplantation and Cellular Therapy. 2025;31(4):228-235. doi:10.1016/j.jtct.2025.01.884
- 27.Bishop MR. Late complications and long-term care of adult CAR T-cell patients. Hematology. 2024;2024(1):109-115. doi:10.1182/hematology.2024000534
- 28.Hines MR, Knight TE, McNerney KO, et al. Immune Effector Cell-Associated Hemophagocytic Lymphohistiocytosis-Like Syndrome. Transplantation and Cellular Therapy. 2023;29(7):438.e1-438.e16. doi:10.1016/j.jtct.2023.03.006
- 29.Kampouri E, Little JS, Rejeski K, Manuel O, Hammond SP, Hill JA. Infections after chimeric antigen receptor (CAR)‐T‐cell therapy for hematologic malignancies. Transplant Infectious Dis. 2023;25(S1). doi:10.1111/tid.14157
- 30.Rejeski K, Perez A, Iacoboni G, et al. The CAR-HEMATOTOX risk-stratifies patients for severe infections and disease progression after CD19 CAR-T in R/R LBCL. J Immunother Cancer. 2022;10(5):e004475. doi:10.1136/jitc-2021-004475
- 31.Rejeski K. 42 – Management of ICAHT, infectious complications, hypogammaglobinemia and B cell aplasia. In: Aljurf M, Bonini C, Chabannon C, et al. The EU CAR-T handbook. EBMT. 2024. https://www.ebmt.org/eu-car-t-handbook
- 32.Chakraborty R, Hill BT, Majeed A, Majhail NS. Late Effects after Chimeric Antigen Receptor T Cell Therapy for Lymphoid Malignancies. Transplantation and Cellular Therapy. 2021;27(3):222-229. doi:10.1016/j.jtct.2020.10.002
- 33.Camacho-Arteaga L, Iacoboni G, Kwon M. Late adverse events after chimeric antigen receptor T-cell therapy for patients with aggressive B-cell non-Hodgkin lymphoma. Front Oncol. 2025;14. doi:10.1001/jamanetworkopen.2024.61683
- 34.Graham CE, Velasco R, Alarcon Tomas A, et al. Non-ICANS neurological complications after CAR T-cell therapies: recommendations from the EBMT Practice Harmonisation and Guidelines Committee. The Lancet Oncology. 2025;26(4):e203-e213. doi:10.1016/s1470-2045(24)00715-0
- 35.McGann M, Gautama B. Management of cytopenia post CAR T-cell therapy. Pharmacy Practice in Focus: Oncology. 2024. https://www.pharmacytimes.com/view/management-of-cytopenia-post-car-t-cell-therapy
- 36.Cordeiro A, Durisek G, Batista M. Late event after anti-CD19 CAR T-cell therapy for relapsed/refractory B-cell non-Hodgkin lymphoma. JAMA Netw Open. Published online 2024. doi:10.3389/fonc.2024.1404351
- 37.Rejeski K, Subklewe M, Aljurf M, et al. Immune effector cell–associated hematotoxicity: EHA/EBMT consensus grading and best practice recommendations. Blood. 2023;142(10):865-877. doi:10.1182/blood.2023020578
- 38.Clinical commissioning policy for the use of therapeutic immunoglobulin (Ig) England (2025): version 2.0. NHS England . 2025. https://www.england.nhs.uk/publication/commissioning-criteria-policy-for-the-use-of-therapeutic-immunoglobulin-ig-in-england/
- 39.Tomas A, Jager U, Bristol G. 45 – Post CAR-T cell therapy (consolidation and relapse): lymphoma. In: Aljurf M, Bonini C, Chabannon C, et al. The EU CAR-T handbook. EBMT. 2024. https://www.ebmt.org/eu-car-t-handbook
- 40.Hamilton MP, Sugio T, Noordenbos T, et al. Risk of Second Tumors and T-Cell Lymphoma after CAR T-Cell Therapy. N Engl J Med. 2024;390(22):2047-2060. doi:10.1056/nejmoa2401361
- 41.Tix T, Alhomoud M, Shouval R, et al. Second Primary Malignancies after CAR T-Cell Therapy: A Systematic Review and Meta-analysis of 5,517 Lymphoma and Myeloma Patients. Clinical Cancer Research. 2024;30(20):4690-4700. doi:10.1158/1078-0432.ccr-24-1798
- 42.Berg P, Bakker C, Sander M, et al. Secondary malignancy of T-cell origin after CAR T-cell therapy: EMA’s conclusions from the evaluation of 38 suspected cases. Gene Ther. Published online December 22, 2025. doi:10.1038/s41434-025-00586-x
- 43.CD19- or BCMA-directed CAR T-cell therapies: risk of secondary malignancy of T-cell origin. Medicines and Healthcare products Regulatory Agency . 2024. https://www.gov.uk/drug-safety-update/letters-and-medicine-recalls-sent-to-healthcare-professionals-in-july-2024
- 44.Hayden P, Bucklein V, Peric Z. 48 – Long-term follow-up and late effects. In: Aljurf M, Bonini C, Chabannon C, et al. The EU CAR-T handbook. EBMT. 2024. https://www.ebmt.org/eu-car-t-handbook
- 45.Renninger J, Kurz L, Stein H. Mitigation and Management of Common Toxicities Associated with the Administration of CAR-T Therapies in Oncology Patients. Drug Saf. 2025;48(7):719-737. doi:10.1007/s40264-025-01538-5
- 46.Miller P, Patel S, Skinner R, et al. Joint consensus statement on the vaccination of adult and paediatric haematopoietic stem cell transplant recipients: Prepared on behalf of the British society of blood and marrow transplantation and cellular therapy (BSBMTCT), the Children’s cancer and Leukaemia Group (CCLG), and British Infection Association (BIA). Journal of Infection. 2023;86(1):1-8. doi:10.1016/j.jinf.2022.11.005
- 47.CAR-T therapy. London: NHS England. NHS England. 2025. https://www.england.nhs.uk/commissioning/spec-services/advanced-therapy-medicinal-products/car-t-therapy
- 48.Penack O, Peczynski C, Boreland W, et al. Management of complications of chimeric antigen receptor T-cell therapy: a report by the European Society of Blood and Marrow Transplantation. haematol. Published online May 30, 2024. doi:10.3324/haematol.2023.284810
- 49.Hill JA, Seo SK. How I prevent infections in patients receiving CD19-targeted chimeric antigen receptor T cells for B-cell malignancies. Blood. 2020;136(8):925-935. doi:10.1182/blood.2019004000
- 50.Marzal-Alfaro MB, Escudero-Vilaplana V, Revuelta-Herrero JL, Collado-Borrell R, Herranz-Alonso A, Sanjurjo-Saez M. Chimeric Antigen Receptor T Cell Therapy Management and Safety: A Practical Tool From a Multidisciplinary Team Perspective. Front Oncol. 2021;11. doi:10.3389/fonc.2021.636068
- 51.Ahmed SO, El Fakih R, Kharfan-Dabaja MA, et al. Setting up a Chimeric Antigen Receptor T Cell Therapy Program: A Framework for Delivery from the Worldwide Network for Blood & Marrow Transplantation. Transplantation and Cellular Therapy. 2025;31(8):533-543. doi:10.1016/j.jtct.2025.05.012
- 52.Nezvalova-Henriksen K, Langebrake C, Bauters T, et al. Implementation and operational management of marketed chimeric antigen receptor T cell (CAR-T Cell) therapy—a guidance by the GoCART Coalition Pharmacist Working Group. Bone Marrow Transplant. 2023;58(10):1069-1074. doi:10.1038/s41409-023-02072-7
- 53.Advanced therapy medicinal products . Pharmacy ATMP Network UK (PAN-UK). 2026. https://www.sps.nhs.uk/home/guidance/advanced-therapy-medicinal-products/
- 54.BOPA SACT verification HUB (including the passport). British Oncology Pharmacy Association. 2026. https://www.bopa.org.uk/verificationhub/
- 55.About the advanced therapy medicinal products programme. NHS England elearning for healthcare . 2025. https://portal.e-lfh.org.uk/


