PAUL BRIAN/DREAMSTIME.COM
This content was published in 2013. We do not recommend that you take any clinical decisions based on this information without first ensuring you have checked the latest guidance.
In developed countries, the incidence of preterm births is 5–9 per cent of all live births.1 Numbers have increased in recent years owing, in part, to the higher rate of multiple births and assisted births (ie, use of forceps or vacuum) and advances in obstetric interventions.2
Induction of labour due to maternal or fetal problems (eg, pre-eclampsia or eclampsia, and intrauterine growth restriction, respectively) accounts for 12 to 15 per cent of preterm births. However, labour can also be spontaneous, and is particularly associated with premature rupture of membranes.
Premature birth is the leading cause of perinatal death and disability. Common risk factors include:1,2
- Maternal smoking (doubles the likelihood of a delivery before 32 weeks)
- Poor nutrition
- Young maternal age
- Physiological stress
- Multifetal pregnancy
Pharmacists can play a useful part advising pregnant women on making healthy choices. For example, meta-analysis of randomised controlled trials has shown that antenatal smoking cessation programmes can lower the incidence of preterm birth.2
Nutrition and lifestyle advice such as maintaining ideal weight by eating a healthy balanced diet and regular exercise is important. In particular, lack of micronutrients, such as magnesium, zinc, calcium and iron, are associated with intrauterine growth retardation.
Understanding the terms
If somebody says their baby was born prematurely, what does this mean in terms of that baby’s development and likely problems? Associated disabilities include deafness, cerebral palsy and behavioural problems. A preterm or premature birth is defined as a baby born before 37 weeks’ gestational age, as opposed to a full-term birth, when a baby is born between 37 and 40 weeks’ gestational age. Panel 1 explains gestational age and other common terms. The terminology of prematurity can be confusing but understanding it is essential to care because doses of many medicines commonly used in neonates depend on these terms. If a baby was born at 32 weeks 12 days ago, then gestational age is 32 weeks, postnatal age is 12 days and corrected gestational age is 33 (weeks) + 5 (days).
Panel 1: Other useful definitions
Neonate A baby aged from birth up to four weeks of life
Gestational (or menstrual) age Age of a fetus in completed weeks counted from the first day of the mother’s last menstruation
Postnatal (or chronological) age Time in number of days, weeks or months from birth
Corrected gestational age The gestational age at birth plus the number of days after delivery (postnatal age), expressed as weeks plus up to six days.
Most mortality and morbidity affects “very premature” neonates (ie, those born before 32 weeks’ gestation) and especially “extremely premature” neonates (those born before 28 weeks of gestation). There are also categories for birth weight:
- Low birth weight (LBW) < 2,500g
- Very low birth weight (VLBW) <1,500g
- Extremely low birth weight (ELBW) <1,000g
These birth weight and gestational age categories are not interchangeable but generally the level of additional care needed by a neonate is inversely related to gestational age and birth weight. Additional care can be defined as follows:
- Special care (eg, gastric tube feeding, temperature maintenance and monitoring for apnoea)
- High dependency care (eg, continuous monitoring, supplemental oxygen and parenteral nutrition)
- Intensive care (eg, mechanical ventilation, exogenous surfactant and other organ support such as the use of inotropes)3
Reflect
- How is the age of a premature baby expressed for pharmaceutical purposes?
- What are the common complications of premature birth?
- What prescriptions might you expect for a premature baby in the community?
Before reading on, think about how this article may help you to do your job better.
Neonatal pharmacokinetics
Although a baby born at 24 weeks is about three months early, all his or her organs will be present.
Their maturity, however, and the production and effectiveness of their biological compounds may be impaired. Neonatal pharmacokinetics varies greatly depending on (ie, level of organ maturity) but with knowledge of fetal development, it is possible to predict some parameters and to modify drug treatment accordingly. Panel 2 discusses this.
Panel 2: Neonatal pharmacokinetics considerations
Absorption
Gastric emptying and gastrointestinal transit is delayed in the premature newborn.4 There is also reduced gastric acid secretion in the first few months of life4 — gastrointestinal pH is close to neutral for at least two weeks in a premature neonate compared with three days in a term neonate, with normal acidic pH reached at around two years of age. This can have an impact where drugs need an acidic environment for activation or where they are in an ionic or anionic state to aid absorption.
Preterm neonates have scarce skeletal muscle mass, making the intramuscular route not only painful but also inappropriate for most treatments due to poor absorption. The rectal route offers unpredictable absorption because of a reduced surface area5 and absorption depends on drug volume and the proximity of the rectal veins. For topical products, it is important to note that neonatal skin is thin and poorly keratinised, which may lead to excessive absorption. Great care must be taken with any product put on the skin of a premature baby, especially in the first few weeks of life.
Distribution
Volume of body fluids, tissue composition, penetration of biological membrane and organ and tissue perfusion all affect drug distribution.4 Neonates have a higher proportion of total body water compared with infants. This can impact on highly water soluble drugs, such as aminoglycosides, increasing their volume of distribution so that bigger doses are needed. Fat content is only 1/5 of body weight and scarce in newborns.4 It is partly due to such differences that the relative doses of some medicines are higher or lower in premature babies than in term babies.
The neonatal blood-brain barrier is a lot more permeable because it is not fully formed so certain drugs can cross it, leading to toxic effects. For example, opiates and benzodiazepines are more likely to cause respiratory depression.
The extent of protein and tissue binding of drugs also affects distribution. High levels of circulating bilirubin may compete for the same binding sites, increasing the free portion of drugs. The opposite can also happen — bilirubin can be displaced from albumin binding sites by certain drugs (eg, furosemide, indometacin, phenytoin, ceftriaxone) causing hyperbilirubinaemia (see main text).
Metabolism
Drug elimination that depends on hepatic biotransformation is generally slow in the first few weeks of life and increases at varying and unpredictable rates. Hepatic phase 1 reactions (cytochrome), such as oxidation, hydrolysis and reduction, develop gradually and reach a metabolic capacity similar to that of adults at six months of age.5 Hepatic phase 2 reactions, such as glucuronidation and acetylation, increase significantly over the first two to three months of life and reach full maturity by three to five years of age. Examples of drugs with metabolism issues in all neonates, particularly in premature ones, include:
- Paracetamol Glucuronidation is reduced in neonates (sulphation is not) so great care must be taken with dosing.
- Chloramphenicol Glucuronidation (the main pathway for this drug) is reduced. If the drug accumulates and crosses the blood–brain barrier, grey baby syndrome results.
- Benzodiazepines The hydroxylation pathway is immature, significantly extending half-lives, so much lower doses need to be used.
Elimination
Glomerular filtration rate and nephron development depends on gestational age. After 26 weeks, all the nephrons are present but are smaller.4 GFR increases rapidly over the first week of life and reaches adult values by about three months.4 Renal tubular secretion and reabsorption appears to mature much more slowly, reaching adult values over six months.4 For comparison, GFR in a preterm neonate over 1kg is 2–3ml/min whereas it is 40ml/min in a term neonate. Dosing of drugs that are renally excreted (eg, aminoglycosides) must take these factors into account. Dosing frequency of up to 36- to 48-hourly may be required in premature infants to avoid accumulation related toxicity.
Common complications
Common complications in preterm babies include respiratory distress syndrome, feed intolerance, gastrooesophageal reflux, hypothermia and poor glucose control. Once these complications have been resolved, the baby is able to maintain his or her own temperature and is growing on enteral feeds, parents feel competent to deal with their baby’s needs, and there are no other concerns, plans for discharge will be started. This will rarely be before 34 weeks corrected gestational age.
Respiratory distress syndrome
Respiratory distress syndrome (RDS) is one of the main clinical problems faced by premature neonates, occurring within four hours of birth.5 It is primarily caused by a deficiency of pulmonary surfactant, a complex mixture of phospholipids and proteins which reduces alveolar surface tension and maintains alveolar stability.5,6 Most alveolar surfactant is produced after about 30–32 weeks’ gestation so babies born before then will probably develop RDS.
In neonates born over 12 weeks early (ie, less than 28 weeks’ gestation), few alveoli are developed and there is insufficient surfactant synthesis to ensure alveolar stability and optimal gas exchange. This leads to alveolar collapse at expiration, and reduced oxygen in blood. The diaphragm and intercostal muscles are not fully developed and, as a consequence, respiratory efforts are not enough to sustain life.6
Presenting features of RDS include tachypnoea (respiratory rate >60bpm), intercostal, subcostal and sternal recession, expiratory grunting, cyanosis and diminished breath sounds. Untreated infants can become fatigued, apnoeic and hypoxic. They may progress to respiratory failure and need assisted ventilation.6
Over the past 20 to 30 years, two major advances in perinatal management — the use of antenatal corticosteroids and exogenous surfactant replacement — have greatly improved clinical outcomes for preterm infants with RDS.5 Corticosteroids that cross the placenta (eg, betamethasone or dexamethasone) given to women at risk of preterm delivery (12mg intramuscularly 24 hours apart for betamethasone or equivalent), support fetal surfactant production by accelerating the maturation of type II pneumocytes and lung maturation. Beneficial effects for preterm infants include a 40 per cent reduced risk of death, RDS and intraventricular haemorrhage.
Exogenous surfactant, given via an endotracheal tube, for the treatment or prophylaxis of RDS is associated with a 40 per cent reduction in mortality and a 30 to 65 per cent reduction in the risk of pneumothorax. In neonates at risk of RDS, surfactant replacement is most effective given at delivery (ie, prophylaxis rather than rescue).5,6
RDS may evolve into chronic lung disease and the baby may be discharged with oxygen therapy. Children with severe chronic lung disease may also be prone to respiratory syncytial virus (RSV) infections. The UK Joint Committee on Vaccination and Immunisation advises that the RSV-monoclonal antibody palivizumab should be offered prophylactically to preterm babies with severe chronic lung disease (defined as oxygen-dependent for at least 28 days from birth) and premature babies with haemodynamically significant acynotic congenital heart disease, during the RSV season (October to February in the UK) and on a case-by-case basis for babies with rare conditions such as severe congenital immunodeficiency.7
Panel 3 discusses routine childhood immunisation.
Panel 3: Advice on immunisation
The overall aim of the routine childhood immunisation programme in the UK is to protect all children against diphtheria, tetanus, pertussis (whooping cough), haemophilus influenza type b (Hib), polio, meningococcus C, measles, mumps, rubella and pneumococcus.8
The first set of immunisations is given at two months of age.8 In premature babies, this is two months postnatal age.
Paracetamol is licensed for use post immunisation, but should only be used if the baby is really irritable due to fever. It is not generally recommended for pain and fever prophylaxis preimmunisation because, in theory, it may dampen the body’s immune response to the vaccine so that full immunity may not be achieved.
Neonatal apnoea
Neonatal apnoea is a pause in breathing lasting more than 20 seconds, accompanied by cyanosis and bradycardia. It is as if the baby forgets to breathe. Gentle stimulation, by touch or movement, may restart breathing.
Apnoea and bradycardia are common in VLBW neonates until they reach 32 weeks corrected gestational age. They result from immaturity of central respiratory regulation and of protective mechanisms that maintain airway patency. Causes include infection, hypoxia, anaemia, metabolic disturbances, convulsions, aspiration from gastrooesophageal reflux and heart failure.
Management of apnoea is by continuous monitoring with alarms, nasal continuous positive airway pressure (CPAP) or mechanical ventilation support, treating any underlying causes and medication.
Methylxanthines such as theophylline and caffeine (the active metabolite of theophylline) are used to stimulate the respiratory centre. Caffeine has a long half-life in neonates, allowing for once-daily dosing. It also does not require any therapeutic drug monitoring, unlike theophylline, because it has a wide therapeutic index. Side effects include tachycardia and seizures but these are more associated with toxic concentrations of theophylline. Gastro-oesophageal reflux may be worsened.
Caffeine (base) is administered intravenously or orally at a loading dose of 10mg/kg followed by maintenance dose of 5mg/kg daily. The maintenance dose can be increased to twice a day, which may be needed after three or four weeks of life as metabolism increases. It is important to note, when prescribing caffeine, that every 1mg of caffeine base is equivalent to 2mg of caffeine citrate and the form required by the prescriber should be clear.
Treatment is usually stopped at 32–34 weeks corrected gestational age because it is thought that the respiratory centre should have developed sufficiently to support breathing. Only rarely will a baby be discharged on caffeine treatment.
Jaundice
All neonates are jaundiced to some extent at birth. Bilirubin, a byproduct of haemoglobin breakdown, is normally conjugated by the liver from a fat soluble to a more water soluble form to be excreted via bile and urine. In premature neonates, rapid haemolysis of maternal haemoglobin, and reduced liver and renal function lead to high levels of bilirubin. Unconjugated (fat soluble) bilirubin can cross the blood brain barrier and deposit in the brain, leading to bilirubin neurotoxicity (kernicterus), which can be fatal.9
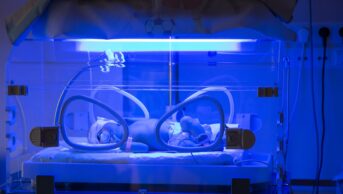
The National Institute for Health and Clinical Excellence released guidance in May 2010 on the management of neonatal jaundice. It recommends single or multiple phototherapy as first-line treatment — exposing the neonate to blue light (wavelength 450nm) has been shown to convert unconjugated bilirubin to water soluble form.9
If phototherapy is ineffective, exchange transfusion can be performed.
Any mother presenting at a pharmacy with a baby with yellow tinted eyes or nails, or both, should be referred to a GP or hospital for bilirubin monitoring but in premature infants, jaundice is usually dealt with before discharge.
Sepsis
Neonatal sepsis can be divided into early and late onset infection. Early onset infection is acquired during labour or delivery and presents in the first 48–72 hours after birth. Late onset infection is usually acquired in hospital and becomes clinically evident more than 72 hours after birth.
Early onset systemic infection includes bacteraemia, pneumonia, meningitis and urinary tract infection. It is rare and should be treated immediately.10 The principal pathogens responsible are group B streptococci and Escherichia coli.10 Most infants will present with signs of sepsis, such as respiratory distress or fever, in the first 12 hours after birth.
Empirical antibiotic treatment is given to all preterm neonates with signs consistent with sepsis or specific risk factors for systemic infection. Intravenous benzylpenicillin plus an aminoglycoside, such as gentamicin, is the commonly used first-line regimen. If listeriosis is suspected (eg, stained meconium or discoloured amniotic fluid), amoxicillin is often used instead of penicillin. Antibiotics are usually stopped after 48 hours in an asymptomatic infant where cultures do not confirm infection.10
Late onset infection is more common, occurring in about 20 per cent of VLBW neonates. Most infections are caused by Gram positive organisms such as Staphylococcus aureus. Coagulase negative staphylococci (CONS) account for half of these.10
Antibiotic treatment should be started as soon as possible and stopped when cultures are confirmed as negative — usually after 48 hours. The first-line treatment for suspected nosocomial sepsis should include an antistaphylococcal antibiotic, such as flucloxacillin, and an aminoglycoside. Flucloxacillin may be switched to vancomycin if CONS is suspected or if there is poor response to first-line antibiotics.10
Seizures
Hypoxic ischaemic encephalopathy due to asphyxia at birth tends to be the most common cause of neonatal seizures.4 Other causes include metabolic disturbance, cerebral infarction, cerebral trauma, infection, hypoglycaemia and opioid withdrawal.4 A cause can be identified in most cases and electroencephalography can confirm this. Management involves treating the underlying cause and pharmacological intervention.
Phenobarbital is normally used as the first-line anticonvulsant,4 leading to 70 per cent seizure control. Phenytoin may be used as a second-line agent. Other drugs that may be used are clonazepam, rectal paraldehyde or diazepam. If a metabolic cause is suspected, pyridoxine and biotin can be tried for a week.
Seizures can be self-limiting or can develop into epilepsy, requiring continued anticonvulsant therapy after discharge.
Gastro-oesophageal reflux
Gastro-oesophageal reflux (GOR) is defined as the nonforceful regurgitation of milk and other gastric contents into the oesophagus.11 It is important to differentiate between GOR and vomiting, which is an active process requiring forceful contraction of the diaphragm and abdominal muscles. GOR encompasses non-erosive reflux and erosive oesophagitis.
Uncomplicated GOR is common in infancy, with at least one episode a day in half of all babies aged up to three months. It normally reduces in the first month to year of life as the oesophageal sphincter gets stronger.11 Peak “problematic” regurgitation occurs by six months and by 12–18 months reflux spontaneously resolves.
Risk factors for GOR include prematurity, congenital oesophageal anomalies, gastric distension, cerebral palsy or other severe neurodevelopmental problems, chronic relaxation or immaturity of the lower oesophageal sphincter.11 Longterm complications include dysphagia, recurrent vomiting, haematemesis, feeding and behavioural problems, chronic cough, aspiration pneumonia and chronic oesophagitis.
GOR is probably the most common problem that community pharmacists will encounter in premature babies. The first step of managing GOR should always be to reassure parents that symptoms often resolve on their own accord. Lifestyle advice, such as avoiding overfeeding (increasing feed frequency and reducing volume) and changing the baby’s position during and after feeding, should be offered.
Feeds may also be thickened with Gaviscon Infant powder (unlicensed use) or Thick and Easy so they act as a barrier against stomach acid coming into contact with the oesophagus. H2 receptor antagonists (eg, ranitidine), dopamine antagonists (eg, domperidone) and proton pump inhibitors (eg, omeprazole) can be used either on their own or in combination as add on treatment for unresolved reflux. Giving doses less than 10mg of omeprazole can be difficult because the tablet does not disperse uniformly in water. Many unlicensed liquids formulated in sodium bicarbonate are available but the bicarbonate can neutralise the acidic environment needed for activation of omeprazole, so their effectiveness is questionable.
Nutrition
Ensuring optimal nutrition is by far the most important intervention that can be made to ensure survival of a premature baby. Breast milk is the most complete form of nutrition in terms of providing appropriate calories and nutrients for growth and development. Breastfeeding has a protective effect on the gut and reduces the risk of necrotising enterocolitis. A mother’s antibodies are also passed on to her baby through breast milk.
However, due to prematurity of the gut or other gastrointestinal complications, a neonate may not be able to tolerate milk and parenteral nutrition may be needed. Parenteral nutrition composition aims to mimic breast milk with macronutrients (glucose, lipid and nitrogen), micronutrients (sodium, potassium, calcium, phosphate and magnesium), vitamins and trace elements.
Central venous access is preferred because there is a risk of extravasation and thrombophlebitis with peripheral venous access.
In very premature neonates, breast milk, even at large volumes, may not be enough to sustain growth and preterm specific formula milks or breast milk fortifiers may be used.
Once off parenteral nutrition, neonates may need additional vitamins and minerals. Oral phosphate and sodium supplements, available from many specials manufacturers (they are unlicensed), are often given to reduce renal losses and promote bone growth.
Most premature neonates will be discharged on a supplement such as Abidec or Dalivit). They also get folic acid and oral iron (to prevent anaemia).
Other issues to consider
Most of the medicines used in neonates are not licensed in that population or are used off label. Pharmacists have an invaluable role in advising on appropriate use of such medicines.
Often the low birth weights of preterm neonates make drug dosage calculation and administration more of a challenge.
Pharmacists have an important role in procuring formulations that are suitable for use in the neonatal population. Liquid formulations often have excipients in them that may not be suitable and this is an area where pharmacists can have a huge impact. For example, commercially available phenobarbital elixir contains 38 per cent ethanol.12 Ethanol readily crosses the blood-brain barrier and can cause adverse effects. An alcohol-free preparation should be used in neonates. Propylene glycol is a solvent with anti-microbial properties used in many paediatric formulations.12 It has been associated with cardiovascular, hepatic, respiratory and CNS adverse events, especially in neonates. Benzyl alcohol, another commonly used preservative should not be used in neonates, because of saturation of its metabolic pathway, leading to a toxic condition known as gasping syndrome.13
Neonatal pharmacists in particular can advise on the right formulations to use and recommend an appropriate dilution for administration. The neonatal pharmacist can also play a vital role in providing accurate and up to date information on whether medicines that the mother may be taking is expressed in breast milk and how this may impact on the neonate.
Another important pharmacy role is the provision of a centralised intravenous additives service (CIVAS) — individually made (patient specific) intravenous medicines prepared in a dedicated aseptic area to reduce the risk of errors that could occur when intravenous medicines are prepared at a ward level by nurses. Use of such products should also reduce infection risk.
Key points
- Premature babies are prone to conditions such as seizures and gastro-oesophageal reflux as well as long-term disabilities.
- Different terms (ie, gestational age, corrected gestational age) may be used for prescribing and need to be understood.
- The suitability of formulations and doses should be checked, consulting a neonatal pharmacist where necessary.
Practice points
Reading is only one way to undertake CPD and the regulator will expect to see various approaches in a pharmacist’s CPD portfolio.
- Look at signposting options for parents of premature babies.
- Ensure that products supplied for premature babies do not contain unsuitable excipients.
- Find out about the milk formulas suitable for premature babies.
Consider making this activity one of your nine CPD entries this year.
References not currently available online.