Shutterstock.com
Rhinitis, the inflammation of the mucous membranes inside the nasal cavity, can be caused by an infection or allergic response (e.g. to cats, dogs, house dust mite faeces or pollen), or can be non-allergic in nature — triggered by perfumes, irritants, medication or changes in the weather[1]. Rhinitis usually co-exists with sinusitis (inflammation of the sinuses) and is therefore collectively referred to as rhinosinusitis[2]. It is a common condition that has a profound impact on healthcare costs and loss of productivity[2].
Acute rhinosinusitis (ARS) has a one-year prevalence of 6–15% and often occurs after a viral common cold or flu[3]. Although usually a self-limiting disease, serious complications leading to life-threatening situations may occur[4]. In addition, ARS is one of the most common reasons for antibiotic prescriptions; therefore, ensuring the condition is correctly identified is essential to prevent antibiotic resistance[5].
Chronic rhinosinusitis (CRS) is a significant health problem affecting 5–12% of the general population[6]. Considering whether the disease is unilateral or bilateral is important, as well as the nature of the inflammatory response[7]. In 2020, the condition became divided into primary CRS (where the disorder is limited to the airway or respiratory system) and secondary CRS (where the disorder is secondary to an established systemic disease or pathological condition, e.g. vasculitis, immune disorders)[8]. Clinical diagnosis involves endoscopic examination and/or a computerised tomography scan in secondary care.
Allergic rhinitis predominantly affects children and young adults for variable durations, and may be seasonal (e.g. hayfever) or perennial (e.g. allergy to house dust mite or pet). As its diagnosis and management has been previously covered in detail (see British National Formulary and summarised visually here), this will not be further covered in this article.
Many patients can self-manage their condition, especially if their symptoms are considered mild or are relieved by over-the-counter (OTC) medication. However, pharmacists need to be able to identify the symptoms of rhinosinusitis and know when to recommend appropriate treatment or referral[7].
This article provides a framework for the diagnosis and appropriate management of patients presenting to pharmacy and primary care.
Symptoms
Patients may present at the pharmacy with the following symptoms indicative of rhinosinusitis:
- Pain, swelling and tenderness around the cheeks, eyes or forehead;
- A blocked nose;
- A reduced sense of smell;
- Green or yellow mucus from the nose;
- A sinus headache is often described; however, headache is not a feature of CRS;
- A high temperature;
- Toothache — owing to the roots of the upper molars lying within or near the maxillary sinuses, which (when these sinuses are inflamed) is interpreted by the brain as coming from the nearby teeth;
- Bad breath — caused by the bacterial sinus infection giving off odours[9,10].
Loss of smell occurs in 20–60% of COVID-19 cases and may be the presenting symptom before other symptoms, such as cough and fever, occur[11]. Patients presenting with sudden onset loss of smell should be considered COVID-19 positive and are advised to follow current guidance on COVID-19. It is important that pharmacists and pharmacy staff ask follow-up questions (see Table) that further examine the patient’s presenting complaint to rule out other possible diagnoses and recommend appropriate treatment once a firm diagnosis has been made[7,12,13].
Patients may complain of facial swelling, typically on their cheeks and around their eyes. Any patients with a visible swelling without a reasonable explanation should be referred[6]. For example, if swelling occurs over the cheek, this may be of dental origin or could be caused by a malignancy[14]. An acute swelling of the forehead could lead to Pott’s puffy tumour — an osteomyelitis of the frontal bone — a rare complication of acute rhinosinusitis[15]. Sometimes chronic swellings occur owing to benign conditions; for example, a mucocele can cause swelling around the eye, and nasal polyps may cause widening of the bridge of the nose[16,17].
Patients presenting with any of the following symptoms should be referred to their GP for potential escalation to an ear, nose and throat specialist or emergency department:
- Unilateral symptoms;
- Bleeding;
- Crusting — this is caused by abnormal mucus production or excessive accumulation. The mucus is thick and does not drain normally. It can be associated with systemic diseases, such as granulomatosis with polyangiitis;
- Red and swollen eyelids;
- Eye/s that appears to be pushed forwards;
- Reduced or double vision;
- A severe unilateral or bilateral frontal headache;
- Swelling of the forehead;
- Drowsiness, neck stiffness and dislike of bright light;
- Cacosmia — the sensation of a bad smell when it does not exist can be caused by temporal lobe epilepsy, brain lesions and psychoses[18–20].
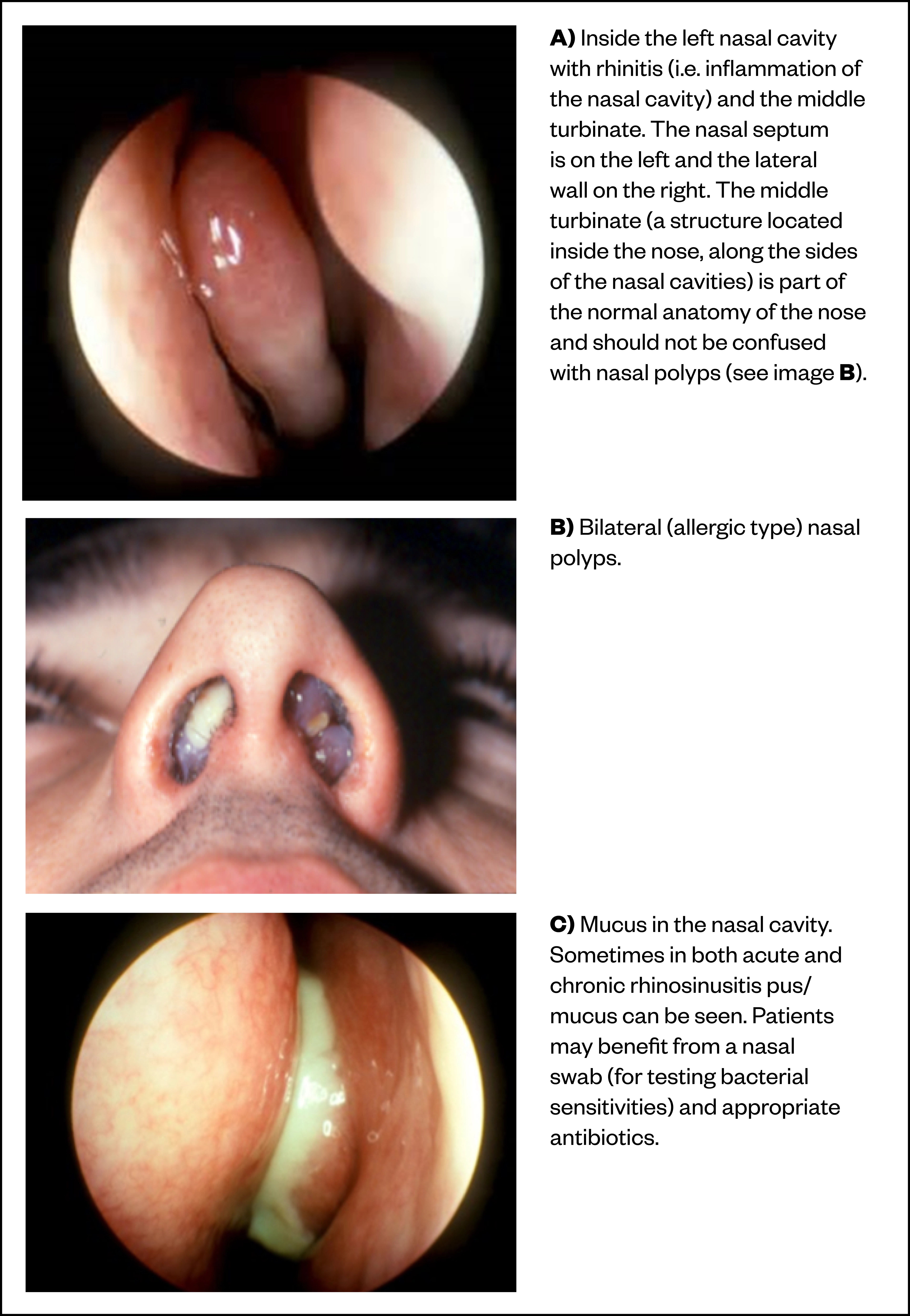
Visual examination inside the patients’ nose may highlight abnormalities or help provide clarification of diagnosis (see Photoguide).
Andrew Pfleiderer
Diagnosis
A clinical diagnosis of rhinosinusitis is made if the following two symptoms are present:
- A feeling of nasal blockage or discharge;
- Facial pain/pressure;
- Loss of sense of smell in adults or a cough in children[7].
If symptoms have been present for less than 12 weeks, the disease is termed ARS. It becomes CRS after 12 weeks[7].
Differentiating rhinosinusitis
Rhinosinusitis is classified into several conditions, based not only on the duration of symptoms but also the presence of other symptoms, including nasal obstruction, rhinorrhoea, sneezing, nasal pruritus and conjunctivitis[20].
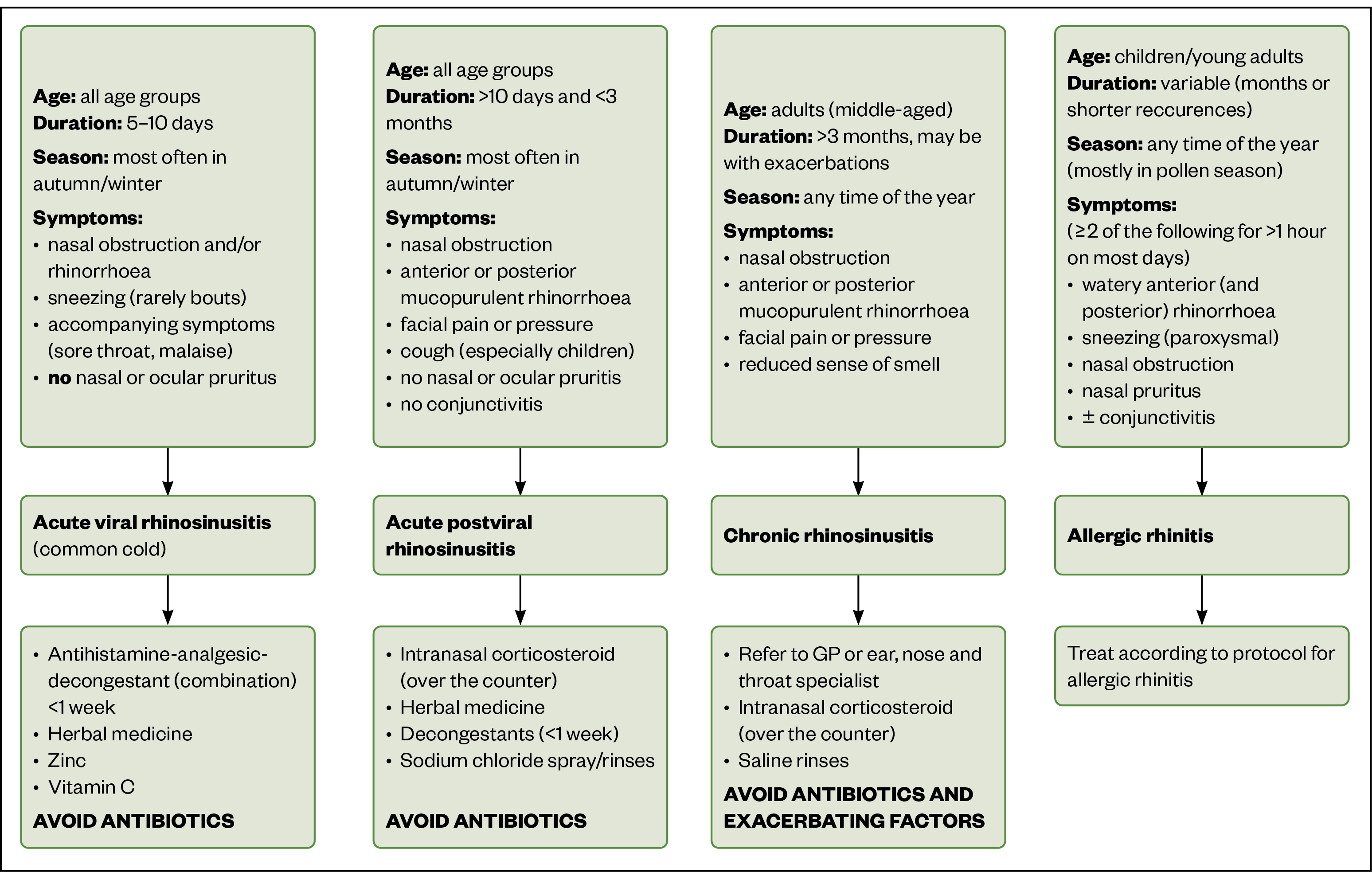
ARS mostly occurs in the autumn and winter, and is subdivided into:
- Acute viral rhinosinusitis (common cold) — affects all age groups with nasal obstruction and/or rhinorrhoea, sneezing, sore throat and malaise, and typically lasts for five to ten days;
- Acute post viral rhinosinusitis — occurs when symptoms persist for more than 10 days, but less than 12 weeks. It may be accompanied by symptoms including facial pain/pressure, reduced sense of smell and a cough;
- Acute bacterial rhinosinusitis (ABRS) can be differentiated by the following signs and symptoms:
- discoloured discharge;
- ‘double-sickening’ — when a deterioration occurs after an initially mild illness;
- elevated erythrocyte sedimentation rate or C-reactive protein (although this result will not be present in the community setting);
- fever (>38°C in adults);
- severe local pain (predominantly unilateral)[7].
CRS occurs when symptoms (such as those in post-viral ARS) are present for more than 12 weeks[6]. Patients may have periods in which their symptoms are exacerbated during this time. In addition, it is more likely to affect adults of working age (18–65 years) and is non-seasonal[2].
Management
The following algorithm (see Figure) can be used to aid diagnosis and guide recommended treatments.
Reproduced with permission from the ‘European position paper on rhinosinusitis and nasal polyps 2020’
Acute viral rhinosinusitis
There are a wide range of over-the-counter (OTC) products available for the symptomatic relief of acute viral rhinosinusitis, including several combination preparations (often containing an analgesic, decongestant and an antihistamine). It is recommended to use separate products that treat the patient’s symptoms and avoid taking unnecessary medication. Some patients need to avoid medicines that will exacerbate comorbidities, including hypertension and glaucoma, or that interact with other medicines, such as monoamine oxidase inhibitors (MAOI) and selective serotonin re-uptake inhibitors[21].
Analgesics
Paracetamol and ibuprofen are useful for managing fever and pain associated with the common cold. However, combination products containing codeine are not advised owing to a lack of codeine efficacy at OTC doses. Use may lead to opioid dependence, addiction and side effects, such as constipation[22]. Pharmacists should counsel against prolonged use of analgesics; for example, advise patients to limit the consumption of simple analgesics to fewer than 15 days per month and, for caffeine- or opioid-containing analgesics, to less than 10 days per month, to avoid medication overuse headache and to ensure patients are only taking one paracetamol-containing product[23].
Nasal decongestants
Decongestants (e.g. oxymetazoline, phenylephrine and pseudoephedrine) stimulate the alpha-adrenergic receptors and act as vasoconstrictors, providing symptomatic relief from nasal blockage[24]. However, their use should be limited to five days to prevent rebound congestion[25]. Use should be avoided in patients with high blood pressure (especially if taking MAOIs), diabetes, hyperthyroidism, enlarged prostates, glaucoma or liver, kidney, heart or circulatory problems[26]. Decongestants should also be avoided in pregnancy and if breastfeeding[26].
Antihistamines
Owing to their ability to cross the blood-brain barrier and central action, first generation (also referred to as sedating) antihistamines, such as brompheniramine, chlorpheniramine and clemastine, are more effective than second-generation antihistamines. However, this benefit is only seen on day one and two of viral ARS[7]. When used in combination with a decongestant, beneficial effects are seen but should be avoided in patients with narrow-angle glaucoma or benign prostatic hypertrophy[27].
Zinc and vitamin C
Zinc (acetate or gluconate) commenced within 24 hours of symptom onset at >75mg per day can lead to a three-fold increase in the rate of recovery from the common cold[28]. Vitamin C also appears to reduce the duration and severity of common cold symptoms and a trial of vitamin C may be worthwhile[29].
Saline douching
There are a range of OTC hypotonic, isotonic and hypertonic saline solutions available to help relieve acute rhinosinusitis symptoms. Delivered via nebulisation or nasal irrigation, they reduce viscosity of mucus and remove it, along with allergens, irritants and bacteria from the nose and sinuses[7]. However, the benefits are more evident in children than adults[30]. Pharmacy teams should be familiar with these products and counsel patients appropriately on their use according to manufacturers’ instructions.
Steam inhalation
The use of steam inhalation (with or without menthol) can be a suitable treatment for congestion associated with rhinosinusitis. However, its use may be detrimental; there is a suggestion that the heat may cause further swelling to the lining of the nose, resulting in further obstruction and thereby preventing drainage[31].
Other therapies
Some herbal medicines, such as the herbal drug BNO 1016 (Sinupret; Bionorica), may have a significant impact on symptoms of the common cold and post-viral ARS, without important adverse effects. There is, however, little to no evidence to support the use of other remedies, such as some herbal medicines (e.g. echinacea), homeopathic products or intranasal steroids to treat colds, post-viral ARS or other acute respiration infections[7,32,33].
Post-viral ARS
Intranasal steroids
Intranasal corticosteroids are recommended to help reduce inflammation at the site of swelling in post-viral ARS[7]. There are several OTC nasal sprays available and pharmacists should be aware of their use and side effects.
Newer intranasal corticosteroids (e.g. fluticasone furoate, fluticasone propionate and mometasone furoate) only require once-daily administration. These drugs are also less likely to cause side effects, owing to their reduced bioavailability compared to older steroids (e.g. beclomethasone dipropionate)[7].
Pharmacists should consult the summary of product characteristics for individual products before recommending their use to pregnant or breastfeeding women[34,35]. Likewise, age restrictions for individual products should also be checked before making recommendations[29].
Following use, some patients may experience irritation, dryness or nose bleeds from the tip of the spray or the steroid itself[36]. Bleeding is usually resolved by modifying the technique; for example, directing the spray away from the nasal septum. Referral may be required if bleeding persists despite these measures.
Pharmacy teams should explain to patients that intranasal steroids have a delayed onset of action (e.g. 12 hours) and maximal efficacy may take days to weeks[37]. Regular use is therefore required to control symptoms[38]. There is also a need to instruct patients on the use of their nasal sprays (and corticosteroid drops, if prescribed) to ensure they are administered effectively[39–43].
Herbal remedies
Some patients take myrtol or pelargonium extracts for post-viral ARS, with some studies suggesting these may be effective; however, important adverse effects or interactions can occur[7]. Pharmacists should not recommend these products to patients, but if a patient chooses to use them, ensure there is no interaction with any prescribed medicine and that they understand the product is only a temporary measure.
Homeopathic products
Pharmacy teams are not encouraged to promote the use of homeopathic products, but some patients may choose to use them. One study found that Sinfrontal (Dr. Gustav Klein GmbH & Co. KG) significantly reduced symptoms and led to radiographic improvement in patients with post-viral ARS[44].
Acute bacterial rhinosinusitis
Adults with ABRS will benefit from an antibiotic prescribed by their GP[7]. There is very low-quality evidence that antihistamines are a useful adjuvant. Patients can obtain symptomatic relief by taking simple, analgesics such as paracetamol and ibuprofen.
Chronic rhinosinusitis
CRS without any red flag symptoms can be managed with OTC saline spray, rinses or intranasal corticosteroid sprays[7].
Patients who ask for antihistamines for CRS or ABRS should be advised that there is no evidence to support their use, and that side effects (e.g. dry mouth, urinary retention, blurred vision and sedation) can occur[26].
It is important to provide patients with advice on risk factors for exacerbation of CRS, which include high body mass index, asthma, hay fever and history of sinus surgery (this most likely correlates to the severity of the CRS warranting surgical intervention rather than to surgery itself)[45].
Antibiotics are not required unless there is an acute exacerbation of the CRS owing to a secondary bacterial infection following a viral upper respiratory tract infection[46].
Management of nasal and sinus symptoms in the COVID-19 era
The British Rhinological Society has made the following recommendations:
- Systemic corticosteroids should not be prescribed in patients with sudden loss of smell in the first two weeks as the spontaneous recovery rate is good, and the side effects and delay in clearing the virus outweigh the benefit of their use[47]. Fluticasone or betamethasone drops can be considered in patients with anosmia for more than two weeks. Topical nasal corticosteroid sprays could be considered in patients with associated nasal obstruction[47].
- Patients who develop anosmia/smell disorders may benefit from smell training, which is performed daily for at least two to three months[48]. They can also be directed to Fifth Sense and Abscent, which are charities providing useful online advice for patients with these symptoms[49,50].
Summary
Rhinosinusitis is a common condition that affects patients’ quality of life, reduces work productivity and results in significant healthcare costs. Information on the range of symptoms and their duration is required to determine the type of rhinosinusitis a patient is experiencing, enabling pharmacists and their teams to give appropriate evidence-based advice, as well as identify patients with symptoms requiring urgent/emergency referral to other healthcare professionals.
This article has been reviewed by the expert author to ensure it is relevant and up to date, following its original publication in July 2021.
- 1Hellings PW, Klimek L, Cingi C, et al. Non-allergic rhinitis: Position paper of the European Academy of Allergy and Clinical Immunology. Allergy. 2017;72:1657–65.
- 2Blackwell D, Collins J, Coles R. Summary health statistics for U.S. adults: National Health Interview Survey, 1997. Vital Health Stat 10. 2002;1–109.
- 3Piatt JH. Intracranial suppuration complicating sinusitis among children: an epidemiological and clinical study. PED. 2011;7:567–74.
- 4Tan R, Cvetkovski B, Kritikos V, et al. Identifying the hidden burden of allergic rhinitis (AR) in community pharmacy: a global phenomenon. asthma res and pract. 2017;3. doi: 10.1186/s40733-017-0036-z
- 5Mayrhuber EA-S, Peersman W, van de Kraats N, et al. “With fever it’s the real flu I would say”: laypersons’ perception of common cold and influenza and their differences – a qualitative study in Austria, Belgium and Croatia. BMC Infect Dis. 2018;18. doi: 10.1186/s12879-018-3568-9
- 6Fokkens W, Lund V, Mullol J, et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2012. Rhinol Suppl. 2012;23:3 p preceding table of contents, 1–298.
- 7Fokkens WJ, Lund VJ, Hopkins C, et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2020. Rhin. 2020;0:1–464.
- 8Grayson J, Hopkins C, Mori E. Contemporary classification of chronic rhinosinusitis: moving beyond CRSwNP and CRSsNP. JAMA Otolaryngol Head Neck Surg. 2020;In press.
- 9Eross E, Dodick D, Eross M. The Sinus, Allergy and Migraine Study (SAMS). Headache. 2007;47:213–24.
- 10Sinusitis (sinus infection). NHS. 2021. https://www.nhs.uk/conditions/sinusitis-sinus-infection/ (accessed July 2021)
- 11Kaye R, Chang CWD, Kazahaya K, et al. COVID-19 Anosmia Reporting Tool: Initial Findings. Otolaryngol Head Neck Surg. 2020;163:132–4.
- 12Carter A, Dattani N, Hannan SA. Chronic rhinosinusitis. BMJ. 2019;l131.
- 13RACHELEFSKY G, GOLDBERG M, KATZ R, et al. Sinus disease in children with respiratory allergy. Journal of Allergy and Clinical Immunology. 1978;61:310–4.
- 14Agrawal A, Singh V, Kumar P, et al. Unilateral swelling of cheek. Natl J Maxillofac Surg. 2017;8:157.
- 15Sharma P, Sharma S, Gupta N, et al. Pott Puffy Tumor. Baylor University Medical Center Proceedings. 2017;30:179–81.
- 16Capra GG, Carbone PN, Mullin DP. Paranasal Sinus Mucocele. Head and Neck Pathol. 2012;6:369–72.
- 17Schoenenberger U, Tasman AJ. Adult-Onset Woakes’ Syndrome: Report of a Rare Case. Case Reports in Otolaryngology. 2015;2015:1–4.
- 18Swain S, Ray R. Wegener’s Granulomatosis of Nose: A Case Report. Indian J Otolaryngol Head Neck Surg. 2011;63:402–4.
- 19Leopold D. Distortion of Olfactory Perception: Diagnosis and Treatment. Chemical Senses. 2002;27:611–5.
- 20Fauci A, Braunwald E, Kasper D, et al. Harrison’s principles of internal medicine. 17th ed. New York: McGraw-Hill Professional 2008.
- 21Rodman M. OTC interactions. Cough, cold, and allergy preparations. RN. 1993;56:38–41.
- 22Robinson J. Is a complete ban on OTC opioids the solution? Pharmaceutical Journal. 2019. https://pharmaceutical-journal.com/article/feature/is-a-complete-ban-on-otc-opioids-the-solution (accessed July 2021)
- 23Diener H-C, Dichgans J, Scholz E, et al. Analgesic-induced chronic headache: long-term results of withdrawal therapy. J Neurol. 1989;236:9–14.
- 24Johnson D, Hricik J. The pharmacology of alpha-adrenergic decongestants. Pharmacotherapy. 1993;13:110S-115S; discussion 143S-146S.
- 25Graf P. Rhinitis Medicamentosa. Treatments in Respiratory Medicine. 2005;4:21–9.
- 26Nose — rhinitis. British National Formulary. 2021. https://bnf.nice.org.uk/treatment-summary/nose.html (accessed July 2021)
- 27Smith SM, Schroeder K, Fahey T. Over-the-counter (OTC) medications for acute cough in children and adults in community settings. Cochrane Database of Systematic Reviews. Published Online First: 24 November 2014. doi: 10.1002/14651858.cd001831.pub5
- 28Hemilä H, Fitzgerald JT, Petrus EJ, et al. Zinc Acetate Lozenges May Improve the Recovery Rate of Common Cold Patients: An Individual Patient Data Meta-Analysis. Open Forum Infectious Diseases. 2017;4. doi: 10.1093/ofid/ofx059
- 29Hemilä H, Chalker E. Vitamin C for preventing and treating the common cold. Cochrane Database of Systematic Reviews. Published Online First: 31 January 2013. doi: 10.1002/14651858.cd000980.pub4
- 30King D, Mitchell B, Williams CP, et al. Saline nasal irrigation for acute upper respiratory tract infections. Cochrane Database of Systematic Reviews. Published Online First: 20 April 2015. doi: 10.1002/14651858.cd006821.pub3
- 31Singh M, Singh M, Jaiswal N, et al. Heated, humidified air for the common cold. Cochrane Database of Systematic Reviews. Published Online First: 29 August 2017. doi: 10.1002/14651858.cd001728.pub6
- 32Karsch-Völk M, Barrett B, Kiefer D, et al. Echinacea for preventing and treating the common cold. Cochrane Database of Systematic Reviews. Published Online First: 20 February 2014. doi: 10.1002/14651858.cd000530.pub3
- 33Hawke K, van Driel ML, Buffington BJ, et al. Homeopathic medicinal products for preventing and treating acute respiratory tract infections in children. Cochrane Database of Systematic Reviews. Published Online First: 9 April 2018. doi: 10.1002/14651858.cd005974.pub4
- 34Alhussien AH, Alhedaithy RA, Alsaleh SA. Safety of intranasal corticosteroid sprays during pregnancy: an updated review. Eur Arch Otorhinolaryngol. 2017;275:325–33.
- 35Corticosteroids (intranasal). British National Formulary. 2021. https://bnf.nice.org.uk/drug-class/corticosteroids-intranasal.html (accessed July 2021)
- 36Ganesh V, Banigo A, McMurran AEL, et al. Does intranasal steroid spray technique affect side effects and compliance? Results of a patient survey. J Laryngol Otol. 2017;131:991–6.
- 37van Cauwenberge P, Bachert C, Passalacqua G, et al. Consensus statement * on the treatment of allergic rhinitis. Allergy. 2000;55:116–34.
- 38Bridgeman MB. Overcoming barriers to intranasal corticosteroid use in patients with uncontrolled allergic rhinitis. IPRP. 2017;Volume 6:109–19.
- 39Menditto E, Costa E, Midão L, et al. Adherence to treatment in allergic rhinitis using mobile technology. The MASK Study. Clin Exp Allergy. 2019;49:442–60.
- 40Scadding GK, Kariyawasam HH, Scadding G, et al. BSACI guideline for the diagnosis and management of allergic and non-allergic rhinitis (Revised Edition 2017; First edition 2007). Clin Exp Allergy. 2017;47:856–89.
- 41Tan NC-W, Drilling AJ, Jardeleza C, et al. Is nasal steroid spray bottle contamination a potential issue in chronic rhinosinusitis? J Laryngol Otol. 2013;128:S28–33.
- 42Merkus P, Ebbens F, Muller B, et al. The ‘best method’ of topical nasal drug delivery: comparison of seven techniques. Rhinology. 2006;44:102–7.
- 43Harvey RJ, Snidvongs K, Kalish LH, et al. Corticosteroid nasal irrigations are more effective than simple sprays in a randomized double-blinded placebo-controlled trial for chronic rhinosinusitis after sinus surgery. Int Forum Allergy Rhinol. 2018;8:461–70.
- 44Zabolotnyi DI, Kneis KC, Richardson A, et al. Efficacy of a Complex Homeopathic Medication (Sinfrontal) in Patients with Acute Maxillary Sinusitis: A Prospective, Randomized, Double-Blind, Placebo-Controlled, Multicenter Clinical Trial. EXPLORE. 2007;3:98–109.
- 45Kuiper JR, Hirsch AG, Bandeen‐Roche K, et al. Prevalence, severity, and risk factors for acute exacerbations of nasal and sinus symptoms by chronic rhinosinusitis status. Allergy. 2018;73:1244–53.
- 46Wu D, Bleier BS, Wei Y. Current Understanding of the Acute Exacerbation of Chronic Rhinosinusitis. Front Cell Infect Microbiol. 2019;9. doi: 10.3389/fcimb.2019.00415
- 47Walker A, Pottinger G, Scott A, et al. Anosmia and loss of smell in the era of covid-19. BMJ. 2020;m2808.
- 48Anosmia/smell disorders. ENT UK. 2023. https://www.entuk.org/patients/conditions/81/anosmia_smell_disorders_new%20(accessed%2019%20Nov%202023 (accessed January 2024)
- 49SmellAbility Toolbox. Fifth Sense. 2024. https://www.fifthsense.org.uk/smell-training/ (accessed January 2024)
- 50Welcome to smell training. Abscent. 2024. https://abscent.org/learn-us/smell-training (accessed January 2024)