Massimo Brega / Look at Sciences / Science Photo Library
Summary
Everyone appreciates the importance of a good night’s sleep, however it is only recently that many of the ill effects of a disturbed sleep-wake cycle and insufficient sleep have been defined, including diverse adverse effects on physical and mental health. Although the boundary between normal and abnormal can be indistinct, sleep disorders are undoubtedly common and frequently undiagnosed.
Sleep disorders can be broadly divided into three areas: hypersomnias (excessive sleep or sleepiness during the day); insomnias (poor quality nocturnal sleep that impacts on daytime performance); and parasomnias (disturbing events occurring from sleep). However, certain primary sleep disorders such as narcolepsy, in which sleep-wake regulation is severely disturbed across the 24-hour period, typically comprise all three elements.
The spectrum of sleep disorders is divided into seven sections, which cover normal variants as well as rare phenomena. These comprise: insomnia, sleep-related breathing disorders, central disorders of hypersomnolence, circadian rhythm sleep-wake disorders, sleep-related movement disorders, other sleep disorders and isolated symptoms and normal variants.
Everyone appreciates the importance of a good night’s sleep, however it is only recently that many of the ill effects of a disturbed sleep-wake cycle and insufficient sleep have been defined, including diverse adverse effects on physical and mental health. Although the boundary between normal and abnormal can be indistinct, sleep disorders are undoubtedly common and frequently undiagnosed. This partly reflects a lack of education about sleep medicine for health professionals but also patchy availability of specialist resources devoted to sleep disorders. A common perception is that sleep is an unwelcome intrusion into more useful wakefulness and few appreciate that it is a true drive state. Indeed, akin to hunger and thirst, sleepiness can only be properly satiated by sleep itself.
Sleep disorders can be broadly divided into three areas: hypersomnias (excessive sleep or sleepiness during the day); insomnias (poor quality nocturnal sleep that impacts on daytime performance); and parasomnias (disturbing events occurring from sleep). However, certain primary sleep disorders, such as narcolepsy, in which sleep-wake regulation is severely disturbed across the 24-hour period, typically comprise all three elements. Furthermore, some people have a circadian dysrhythmia or abnormal ‘biological clock’, sleeping and waking normally, but at times out of sync with the day/night cycle where they live[1]
.
Excessive daytime sleepiness (EDS) affects around 5% of an urban population in the United States[2]
and should be carefully distinguished from those who report simple fatigue or poor exercise tolerance. Parasomnias are extremely common in the younger population and an Italian study showed that sleepwalking occurred regularly in at least 15% of children[3]
. It is usually a benign phenomenon that rarely presents to health services, although sleepwalking may persist or recur in around 2% of adults[4]
and is often of considerable concern to the individual and bed partner.
The total amount of sleep required for optimal function varies between individuals, however the majority of adults require seven hours of sleep, with children needing more[5]
. Fragmentation and decreasing sleep continuity are more common with increasing age[6]
, but subtle changes in deep (slow wave) sleep can be seen in healthy young adults with subsequent progressive deterioration as a normal feature of ageing[7]
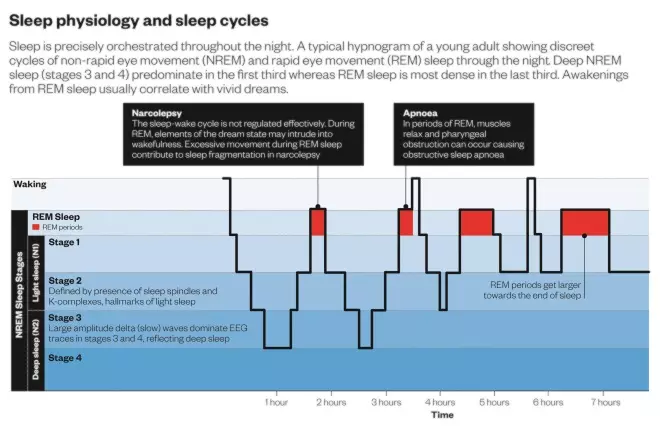
. Excessive nocturnal leg movements are also increasingly identified during overnight testing as patients age, and may interfere with sleep quality (see ‘Sleep physiology and sleep cycles’).
Chronic lack of good quality sleep can contribute to the development of health problems including hypertension, diabetes and obesity, as well as adversely affecting the immune system[8]
. Furthermore, a number of conditions, including generalised pain syndromes and mood disorders such as depression, interact with sleep reciprocally so sleep deprivation may actively fuel the underlying problem[9]
. In addition, although sleepiness may seem the most obvious manifestation of insufficient sleep, it often masquerades as poor memory and concentration, ‘brain fog’, or even dementia in the elderly[10]
. After reduced overnight sleep, characteristic functions attributed to the frontal lobes, including sense of humour, decision making and even emotional intelligence, are particularly impaired[11]
. Attention problems and irritability in young children is frequently a direct result of lack of sleep, yet may not be recognised as such.
Sleep problems associated with shift work are universal and also increase with age[12]
. The relative lack of good quality ‘rebound’ sleep following a night shift leads to a cycle of sleep deprivation and occasional dependence on hypnotic agents. Of concern, long-term night shift work is associated with serious health outcomes, including cancer[12]
.
Spectrum of sleep disorders
The International Classification of Sleep Disorders (ICSD), published by the American Academy of Sleep Medicine, describes the whole spectrum of sleep disorders and their definitions[13]
. The ICSD guidance concerning diagnostic procedures has not been fully accepted by countries outside the United States. In the UK, many sleep laboratories have started to use the new parameters for scoring sleep stages. The spectrum of sleep disorders is divided into seven sections, which cover normal variants as well as rare phenomena. These comprise: insomnia, sleep-related breathing disorders, central disorders of hypersomnolence, circadian rhythm sleep-wake disorders, sleep-related movement disorders, other sleep disorders and isolated symptoms and normal variants.
This article will focus primarily on insomnia, sleep-related breathing disorders and central disorders of hypersomnolence.
Insomnia
Depending on the precise definition and diagnostic criteria used, 4–22% of the population in a large US survey[14]
reported significant insomnia, a proportion that increases with age[15]
.
Chronic insomnia can be defined as an inability to achieve or maintain sleep satisfactorily on the majority of nights over a period of at least three months, despite adequate opportunity, with subsequent adverse consequences on daily functioning[13]
. The amount of sleep needed and quality of sleep varies between individuals, therefore no fixed amount of sleep time is suggested in most definitions. Clinically, it is often helpful to distinguish insomnia into primary and secondary or co-morbid forms, in which there are clear or identifiable factors that are toxic to sleep. Potentially reversible examples of co-morbid insomnia include excessive noise in the environment, pain syndromes and acid reflux at night. Most subjects with insomnia will be unable to nap during the day despite restricted sleep time at night and reports of tiredness. Secondary insomnia is a symptom or side effect of something else or an external factor, such as stress or anxiety. Patients with secondary insomnia will report increased sleepiness and napping throughout the day[13]
.
Psychophysiological insomnia is the most common form of insomnia and is thought to be fuelled by excessive cognitive arousal. Typically, a stressful index event triggers acute problems with achieving sleep which, in someone constitutionally predisposed to insomnia, will then lead to chronic symptoms. Frequently, there are reports of excessive rumination or a ‘racing mind’ in association. Moreover, there are nearly always maladaptive habits that fuel or maintain the insomnia that behavioural therapies could focus on[16]
.
Insomnia is prevalent in the elderly population, with more than 20%[17] experiencing it. A minority of subjects with apparently severe insomnia may actually have sleep-wake misperception or ‘paradoxical’ insomnia which can be demonstrated by objective prolonged monitoring[18]
. Restless leg syndrome may often present in patients of increasing age (see ‘Restless leg syndrome’).
In younger people who appear to display significant sleep onset insomnia, the possibility of delayed sleep phase syndrome (DSPS) should be considered. DSPS typically affects young people in their late teenage years and largely reflects an abnormal biological clock or circadian rhythm, often with a genetic predisposition[19]
. Sleep may not be easily achieved before around 3am but thereafter is of normal length and quality, frequently causing significant problems with waking at a conventional hour. Bad bedtime habits may fuel the problem, but the primary cause reflects an abnormal internal clock mechanism most often caused by defined polymorphisms in genes controlling cellular rhythms[19]
.
Insomnia is also recognised as an independent risk factor for hypertension and depression[20]
.
Restless leg syndrome
Patients with insomnia may also present with restless leg syndrome (RLS), which is a treatable condition. It affects 5–15%[21]
of adults, but has a wide spectrum of severity. RLS is more common in females and rises with increasing age and is associated with iron deficiency and pregnancy[22]
.
A family history of RLS is commonly obtained, especially if age of onset is under 40 years. Diagnosis is symptom-based and requires there to be a sensation of restlessness in the limbs when stationary with temporary relief on movement of the affected limb. The affected limbs can include the arms, particularly in the evening or before bed. Discomfort can be intense and can masquerade as a neuropathic problem. Associated repetitive involuntary limb jerks when drowsy or actually asleep, so-called periodic limb movements (PLMs), may dominate the picture in some people and cause particular distress to bed partners.
Sleep-related breathing disorders
The most common manifestation of sleep-related breathing disorders is socially disruptive snoring, which is reported in more than 30% of the UK population and particularly affects males, the elderly and overweight people[23]
.
Obstructive sleep apnoea (OSA) occurs when snoring is severe enough to intermittently obstruct the upper airway and cause severe degradation of sleep quality with consequent daytime sleepiness and fatigue[24]
,[25]
. The most common reason for referral to a sleep clinic in the UK is to exclude or assess OSA. Around 4% of middle-aged men and 2% of women in the UK are affected by OSA[26]
, with other patients often going undiagnosed, especially if there is no bed partner to corroborate the history. The secondary effects of OSA include deleterious consequences for blood pressure, diabetes risk and cognition[27]
, with evidence indicating neurotoxicity in severe cases, and potential contributions from both chronic sleep deprivation and the effects of recurrent nocturnal hypoxia[28]
.
Abdominal and visceral fat accumulation or central obesity is the highest risk factor for OSA, resulting in increased deposition of soft tissue around the neck[29]
. Pharyngeal obstruction occurs when muscles are relaxed during sleep, especially during the rapid eye movement (REM) stage and when lying supine. In a minority of patients, nasal pathology may be the main site of obstruction, while others may have a congenitally small pharyngeal airway space, often with receding chins as the most common observation. Hypothyroidism and acromegaly may also cause soft tissue accumulation in the neck resulting in OSA[30]
. Sedative medication, including alcohol, can considerably worsen OSA in those people who are predisposed.
Nocturnal hypoventilation occurs when there are significant abnormalities of gas exchange overnight, with patients reporting unrefreshing sleep and subsequent daytime somnolence. Nocturnal hypoventilation can sometimes occur as a result of the mechanical problems of ventilation in the extremely obese, but can be serious in patients with significant diaphragmatic muscle weakness caused by diseases such as muscular dystrophy or motor neuron disease, in whom mechanical or assisted nocturnal ventilation may be appropriate and life-enhancing[31]
,[32]
.
Central disorders of hypersomnolence
Conditions in which cerebral pathology is thought to directly produce excessive daytime drowsiness are collectively termed central disorders of hypersomnolence.
Narcolepsy is the most clearly defined syndrome, which affects around 1 in 2000 people in Caucasian populations[33]
. The common features of narcolepsy reflect an inability to regulate the sleep-wake cycle effectively, with particular problems for the REM sleep stage, such that elements of the dream state may intrude into wakefulness. Aside from an often irresistible need to sleep at intervals throughout the day, other typical symptoms include sleep fragmentation at night with a variety of phenomena such as intrusive dreams, frank hallucinations (usually of a visual nature), sleep paralysis or excessive movement during REM sleep[33]
. The most specific symptom of narcolepsy is cataplexy, in which the voluntary musculature becomes paralysed, in part or totally, when the subject is emotional or anticipating an emotion, particularly laughter. More than 60% of patients with narcolepsy will display cataplexy, although it can be subtle. For some, it is the most disabling symptom, with several attacks of full-blown collapse daily leading to social isolation or even injury. Cataplexy is thought to reflect the inappropriate intrusion of REM sleep paralysis and is usually more obvious when the subject is drowsy[33]
. Consciousness is fully maintained during episodes, which can wax and wane over minutes in severe cases[33]
.
Most narcoleptic subjects will report having slept in unusual or inappropriate places, although, typically, short naps are relatively restoring and often associated with dreaming. With hindsight, a significant majority of patients report excessive sleepiness at school or college, given that the age of peak onset is in early adolescence. As a result, especially if undiagnosed, it is very common for narcolepsy sufferers not to achieve their full potential during educational years[34]
.
It is now firmly established that narcolepsy is usually caused by specific damage in a subset of neurons in the hypothalamus that contain a neuropeptide (hypocretin, also called orexin), which is essential to regulating the sleep-wake cycle[33]
. Most cases arise sporadically and a positive family history is rare. However, occasionally, a clear trigger can be identified in which the immune response is upregulated with presumed collateral damage to hypocretin-containing neurons. Notable examples include reactions to the swine flu vaccine, Pandemrix, which led to a 13-fold increase in the incidence of narcolepsy in the children of several European countries in 2009[35]
.
Rarely, narcolepsy, usually without definite cataplexy, can result from discrete pathology to the hypothalamus, either in the context of neurosurgery or an underlying structural lesion, often in the floor of the third ventricle[36]
. Inflammatory brain disorders, including multiple sclerosis, can sometimes be associated with narcolepsy, presumably because of pathology affecting the hypothalamus or its connections[37]
.
Idiopathic hypersomnolence (IH) is a rare disorder that can mimic the severe daytime sleepiness seen in narcolepsy and is difficult to diagnose with confidence. IH tends to affect younger populations and its pathogenesis is poorly understood[38]
. Typically, a patient will report an excessive amount of good quality overnight sleep with early sleep onset and subsequent extreme difficulty arising at a conventional hour. Despite at least ten hours of nocturnal sleep, they will often nap excessively during the day or display signs of sleepiness including performance of automatic behaviours (e.g. writing nonsense prose down a page in a state between wake and sleep). Usually no REM sleep-related phenomena of note are present in the patient history but accompanying mood disorder is common, most likely a result of the primary sleep disorder, rather than as a causative factor[38]
. Distinguishing IH from patients with a naturally high sleep need, who try unsuccessfully to adopt conventional sleep-wake cycles, can be difficult in the absence of specific diagnostic tests.
Diagnosis
Insomnia
,
in particular chronic insomnia, can be diagnosed in patients presenting to a sleep clinic based on history alone, occasional aided by sleep diaries for a few weeks prior to assessment. If the history suggests possibility factors, such as excessive leg movement, may be fuelling a poor sleep pattern, overnight testing with polysomnography (PSG) may be indicated, particularly if sleep maintenance is the main issue. PSG is considered the gold standard investigation for analysing nocturnal sleep, as it gives an accurate profile of sleep architecture and staging through the night with contemporaneous measurements of breathing parameters and limb movement. It may be helpful to monitor subjects with insomnia for a few weeks with actigraphy in order to characterise their sleep-wake cycle objectively. Several commercial devices resembling wristwatches are available, which are generally worn on the non-dominant hand, detecting even slight movements as a surrogate measure of wakefulness. In practice, actigraphy is mostly used for demonstrating circadian rhythm disorders or paradoxical insomnia[39]
.
Several validated questionnaires have been developed for quantifying levels of insomnia[40]
. Although rarely helpful in a purely clinical setting, one of the most popular is the Pittsburgh Sleep Quality Scale (PSQI), originally used in psychiatric patients with sleep disturbance[41]
. A composite score is obtained from a detailed questionnaire addressing seven domains, including subjective sleep quality, sleep latency, sleep disturbance and use of medication[41]
. The self-reported element of the questionnaire is scored and provides a measure that is more often used in research than in clinical situations.
Sleep-related breathing disorders
are
often suspected in the context of severe snoring and unrefreshing nocturnal sleep, and the most common reason for investigating a sleepy patient is to exclude OSA. Ambulatory pulse oximetry is a simple technique using a finger probe, which repeatedly measures peripheral oxygen desaturation levels overnight along with pulse rate and can be performed in the home setting. In patients with OSA, characteristic dips in oxygen are observed in association with pulse rate rises that often indicate partial arousals from sleep
[42]
(see ‘Sleep physiology and sleep cycles’). If the data are not conclusive of OSA, or if more detail is required, home recordings are undertaken with additional measures. Electrodes that detect lower limb movement are combined with chest leads that record respiratory effort, in order to provide more information on the nature of the breathing disorder and the presence of excessive limb movement during sleep.
Narcolepsy is relatively easy to diagnose with confidence when a patient provides a clear history that includes cataplexy, however, given the importance of an accurate diagnosis, it is usually appropriate to investigate further. Ideally, overnight sleep should be recorded with full PSG, followed by a multiple sleep latency test (MSLT) in which a subject’s propensity to fall asleep is measured at two-hourly intervals. Some authorities prefer to record over two consecutive nights to minimise the disrupting effects of sleeping away from the home environment in a sleep laboratory with multiple sensors. In narcolepsy, it is common to see abnormalities of sleep regulation with chaotic sleep architecture and early onset of REM sleep[33]
. A variety of parasomnias, including sleep talking and excessive limb movement, are also prevalent in this group (see ‘Sleep physiology and sleep cycles’).
Having obtained information on a subject’s typical overnight sleep, a MSLT undertaken the following morning provides a measure of how quickly the patient falls asleep when asked to do so. A patient with narcolepsy will take less than eight minutes and usually only two or three minutes on average, with a particular tendency to enter REM sleep within 15 minutes on at least two of the nap opportunities. Although the MSLT is not generally a good test of ‘sleepiness’, given the wide variation of sleep propensity in the normal population, typically a subject with no sleep disorder will take at least 15 minutes to fall asleep.
The criteria for fulfilling a diagnosis of narcolepsy on a PSG and subsequent MSLT are often criticised for being either too insensitive or sometimes unspecific[43]
. Results are often compromised by variations in the precise protocol for the procedures or if there is excessive anxiety. It can be helpful to measure levels of the neuropeptide hypocretin in cerebrospinal fluid. Very low or undetectable levels (<110 pg/ml) are diagnostic of narcolepsy, usually with cataplexy[44]
.
Idiopathic hypersomnolence is a diagnosis of exclusion, with typical cases showing excessive amounts of good quality overnight sleep including deep NREM sleep. Despite this, the MSLT will usually show a short mean sleep latency the following morning[13]
.
Parasomnias commonly arise from deep NREM sleep and are a variant of sleep walking. If present in adults, the nocturnal disturbances may be violent and potentially dangerous. The usefulness of formal sleep investigations is debated in this situation, given the relatively rare chance of actually capturing events whilst monitored. However, if a secondary sleep disorder, such as sleep apnoea or excessive (periodic) limb movements, are triggering parasomnia activity, excluding and subsequently treating these may be a useful strategy.
Parasomnia resulting from REM sleep that most commonly affects middle-aged or elderly men is being increasingly recognised and confirmed on detailed overnight testing with PSG. In REM sleep behaviour disorder (RBD), the mechanism for peripheral muscle paralysis during REM sleep fails, causing dream enactment and occasional injury. Of interest, this parasomnia may often herald a neurodegenerative condition that subsequently develops over several years in the subject, particularly Parkinson’s disease[45]
.
Paul Reading is a consultant neurologist at The James Cook University Hospital, South Tees Hospitals NHS Trust.
References
[1] Moore RY. Circadian rhythms: basic neurobiology and clinical applications. Annu Rev Med 1997;48:253–266. doi:10.1146/annurev.med.48.1.253
[2] Karacan I, Thornby JI, Anch M et al. Prevalence of sleep disturbance in a primarily urban Florida County. Sci Sci Med 1976;10(5):239–244. doi:10.1016/0037-7856(76)90006-8
[3] Bruni O & Novelli L. Sleep disorders in children. BMJ Clin Evid 2010. PMID: 21418676
[4] Ohayon MM, Guilleminault C & Priest G. Night terrors, sleepwalking and confusional arousals in the general population: their frequency and relationship to other sleep and mental disorders. J Clin Psychiatry 1999;60(4);268–276. doi:10.4088/jcp.v60n0413
[5] Colonna A, Smith AM, Pal DK et al. Novel mechanisms, treatments, and outcome measures in childhood sleep. Front Psychol 2015;6:602. doi:10.3389/fpsyg.2015.00602
[6] Dijk DJ, Groeger JA, Stanley N et al. Age-related reduction in daytime sleep propensity and nocturnal slow wave sleep. Sleep 2010;33(2):211–223. PMCID: PMC2817908
[7] Dijk DJ, Beersma DG & van der Hoofdakker RH. All night spectral analysis of EEG sleep in young adult and middle-aged male subjects. Neurobiol Aging 1989;10(6):677–682. doi:10.1016/0197-4580(89)90004-3
[8] Irwin MR. Why sleep is important for health: a psychoneuroimmunology perspective. Annu Rev Psychol 2014;66:143–172. doi:10.1146/annurev-psych-010213-115205
[9] Boake PA, Olechowski C, Rashiq S et al. A critical review of neurobiological factors involved in the interactions between chronic pain, depression and sleep duration. Clin J Pain 2015. doi:10.1097/ajp.0000000000000260
[10] Gagnon K, Baril AA, Gagnon JF et al. Cognitive impairment in obstructive apnea. Pathol Biol (Paris) 2014;62(5):233–240. doi:10.1016/j.patbio.2014.05.015
[11] Horne J. Working through the night: beyond “sleepiness” – impairments to critical thinking. Neurosci Biobehav Rev 2012;36(10):2226–2231. doi:10.1016/j.neubiorev.2012.08.005
[12] Wang P, Ren FM, Lin Y et al. Night-shift work, sleep duration, daytime napping and breast cancer risk. Sleep Med 2015;16(4):462–268. doi:10.1016/j.sleep.2014.11.017
[13] International Classification of Sleep Disorders. 3rd ed. Darien, IL: American Academy of Sleep Medicine; 2014.
[14] Loudon RJ, Muurlink O, Peetz D et al. Does age affect the relationship between control at work and sleep disturbance for shift workers? Chronobiol Int 2014;31(10):1190–1200. doi:10.3109/07420528.2014.957307
[15] Roth T, Coulouvrat C, Hajak G et al. Prevalence and perceived health associated with insomnia based on DSM-IV-TR. Biol Psychiatry 2011;69(6):592–600. doi:10.1016/j.biopsych.2010.10.023
[16] Spielman AJ, Caruso LS & Glovinsky PB. A behavioural perspective on insomnia treatment. Psychiatr Clin North Am 1987;10(4):541–553. PMID: 3332317
[17] Alessi C & Vitiello MV. Insomnia (primary) in older people: non-drug treatments. BMJ Clin Evid 2015. PMID: 25968443
[18] Harvey AG & Tang NK. (Mis)perception of sleep in insomnia: a puzzle and a resolution. Psychol Bull 2012;138(1):77–101. doi:10.1037/a0025730
[19] Wyatt JK. Delayed sleep phase syndrome: pathophysiology and treatment options. Sleep 2004;27(6):1195–1203. PMID: 15532214
[20] Cunnington D, Junge MF & Fernando AT. Insomnia: prevalence, consequences and effective treatment. Med J Aust 2013;199(8):S36–S40. doi:10.5694/mja13.10718
[21] Leschziner G & Gringras P. Restless legs syndrome. BMJ 2012;344:e3056 doi:10.1136/bmj.e3056
[22] Allen RP. Controversies and challenges in defining the etiology and pathophysiology of restless legs syndrome. Am J Med 2007;120(1):S13–S21. doi:10.1016/j.amjmed.2006.11.003
[23] Lee W, Nagubai S, Kryger MH et al. Epidemiology of obstructive sleep apnea: a population-based perspective. Expert Rev Respir Med 2008;2(3):349–364. doi:10.1586/17476348.2.3.349
[24] Paiva T & Attarian H. Obstructive sleep apnea and other sleep-related syndromes. Handb Clin Neurol 2014;119:251–271. doi:10.1016/b978-0-7020-4086-3.00018-7
[25] Spicuzza L, Caruso D & Di Maria G. Obstructive sleep apnoea syndrome and its management. Ther Adv Chronic Dis 2015;6(5):273–285. doi:10.1177/2040622315590318
[26] Gibson GJ. Obstructive sleep apnoea syndrome: underestimated and undertreated. Br Med Bull 2005;72:49–65. doi:10.1093/bmb/ldh044
[27] Zamarron C, Garcia Paz V & Riveiro A. Obstructive sleep apnea syndrome is a systemic disease. Current evidence. Eur J Intern Med 2008;19(6):390–398. doi:10.1016/j.ejim.2007.12.006
[28] Rosenzweig I, Williams SC & Morrell MJ. The impact of sleep and hypoxia on the brain: potential mechanisms for the effects of obstructive sleep apnea. Curr Opin Pulm Med 2014;20(6):565–571. doi:10.1097/mcp.0000000000000099
[29] Degache F, Sforza E, Dauphinot V et al. Relation of central fat mass to obstructive sleep apnea in the elderly. Sleep 2013;36(4):501–507. doi:10.5665/sleep.2532
[30] Lin CC, Tsan KW & Chen PJ. The relationship between sleep apnea syndrome and hypothyroidism. Chest 1992;102(6):1663–1667. doi:10.1378/chest.102.6.1663
[31] Castro-Anon O, Perez de Llano LA, De la Fuente Sanchez S et al. Obesity-hypoventilation syndrome: increased risk of death over sleep apnea syndrome. PLoS One 2015;10(2):e0117808. doi:10.1371/journal.pone.0117808
[32] Sanjuan-Lopez P, Valino-Lopez P, Ricoy-Gabaldon J et al. Amyotrophic lateral sclerosis: impact of pulmonary follow-up and mechanical ventilation on survival. A study of 114 cases. Arch Bronchoneumol 2014;50(12):509–513. doi:10.1016/j.arbr.2014.10.008
[33] Dauvilliers Y, Arnulf I & Mignot E. Narcolepsy with cataplexy. Lancet 2007;369(9560):499–511. doi:10.1016/s0140-6736(07)60237-2
[34] Inocente CO, Gustin MP, Lavault S et al. Quality of life in children with narcolepsy. CNS Neurosci Ther 2014;20(8):763–771. doi:10.1111/cns.12291
[35] Miller E, Andrews N, Stellitano L et al. Risk of narcolepsy in children and young people receiving AS03 adjuvanted pandemic A/HINI 2009 influenza vaccine: retrospective analysis. BMJ 2013;346f794. doi:10.1136/bmj.f794
[36] Nishino S & Kanbayashi T. Symptomatic narcolepsy, cataplexy and hypersomnia and their implications in the hypothalamic hypocretin/orexin system. Sleep Med Rev 2005;9(4):268–310. doi:10.1016/j.smrv.2005.03.004
[37] Vrethem M, Malmgren K & Lindh J. A patient with both narcolepsy and multiple sclerosis in association with Pandemrix vaccination. J Neurol Sci 2012;321:89–91. doi:10.1016/j.jns.2012.07.025
[38] Ali M, Auger RR, Slocumb NL et al. Idiopathic hypersomnia: clinical features and response to treatment. J Clin Sleep Med 2009;5(6):562–568. PMCID: PMC2792973
[39] Martin JL & Hakim AD. Wrist actigraphy. Chest 2011;139(6):1514–1527. doi:10.1378/chest.10-1872
[40] Senthilvel E, Auckley D & Dasarathy J. Evaluation of sleep disorders in the primary care setting: history taking and questionnaires. J Clin Sleep Med 2011;7(1):41–48. PMCID: PMC3041630
[41] Backhaus J, Junghanns K, Broocks A et al. Test-retest reliability and validity of the Pittsburgh Sleep Quality Index in primary insomnia. J Psychosom Res 2002;53(3):737–740. doi:10.1016/s0022-3999(02)00330-6
[42] Sleepdex. Resources for better sleep. Available at: http://www.sleepdex.org/stages.htm (accessed October 2015).
[43] Bonnett MH. ACNS clinical controversy: MSLT and MWT have limited clinical utility. J Clin Neurophysiol 2006;23(1):50–58. doi:10.1097/01.wnp.0000190415.83841.17
[44] Nishino S, Ripley B, Overeem S et al. Hypocretin (orexin) deficiency in human narcolepsy. Lancet 2000;355(9197):39–40. doi:10.1016/s0140-6736(99)05582-8
[45] Howell MJ & Schenck CH. Rapid eye movement sleep behaviour disorder and neurodegenerative disease. JAMA Neurol 2015;72(6):707–712. doi:10.1001/jamaneurol.2014.4563
You might also be interested in…

Insomnia disorder: management strategies
Sleep disorders: treatment
