Shutterstock.com
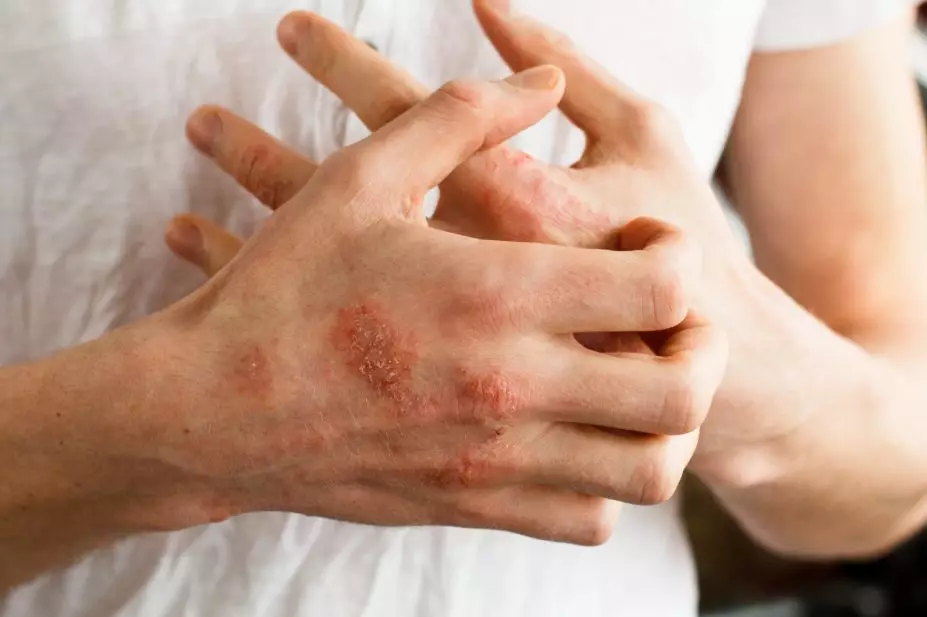
Adding the oral selective Janus kinase (JAK) 1 and 2 inhibitor, baricitinib (4mg), to topical corticosteroid (TCS) treatment significantly improves the signs and symptoms of moderate-to-severe atopic dermatitis (AD), a phase III study in JAMA Dermatology has suggested (30 September 2020)[1]
.
In the trial, 329 adults with moderate-to-severe AD who had an inadequate response to TCS therapy were randomised to receive either 2mg of baricitinib once daily, 4mg of baricitinib once daily, or placebo, for 16 weeks in combination with TCS.
At week 16, 31% of patients who received 4mg of the drug and 24% of those who received 2mg had achieved a validated Investigator Global Assessment for Atopic Dermatitis score of 0 (clear) or 1 (almost clear), compared with 15% in the placebo group.
Adverse events were reported in 38% of participants in the placebo group compared with 58% and 56% in the 4mg and 2mg groups, respectively. However, serious adverse events affected the same number of participants in the 4mg group as the placebo group.
“Treatment with 4mg of baricitinib in combination with background TCS therapy significantly improved the signs and symptoms of moderate to severe AD in adults compared with placebo, with a rapid onset of action and a safety profile consistent with prior findings from baricitinib clinical development in AD,” the researchers concluded.
References
[1] Reich K, Kabashima K, Peris K, et al. Efficacy and safety of baricitinib combined with topical corticosteroids for treatment of moderate to severe atopic dermatitis. JAMA Dermatol 2020. doi: 10.1001/jamadermatol.2020.3260