TEK IMAGE/SCIENCE PHOTO LIBRARY
The Competition and Markets Authority (CMA) is investigating price increases for antibiotics used to treat group A streptococcus (Strep A) infection during a surge in cases.
As a result of demand, the government has banned the export or hoarding of four different antibiotics.
The increase in cases of Strep A among children since the beginning of December 2022 has led to increased demand for the drugs, particularly in solution. Pharmacists have reported shortages of antibiotics and increased prices, with some saying that they have been forced to dispense prescriptions at a financial loss.
David Webb, chief pharmaceutical officer for England, wrote to pharmacists on 8 December 2022, advising that “sufficient stock” of relevant antibiotics was available at a national level, but that local pharmacy teams may be experiencing “temporary interruption of supply” owing to high demand.
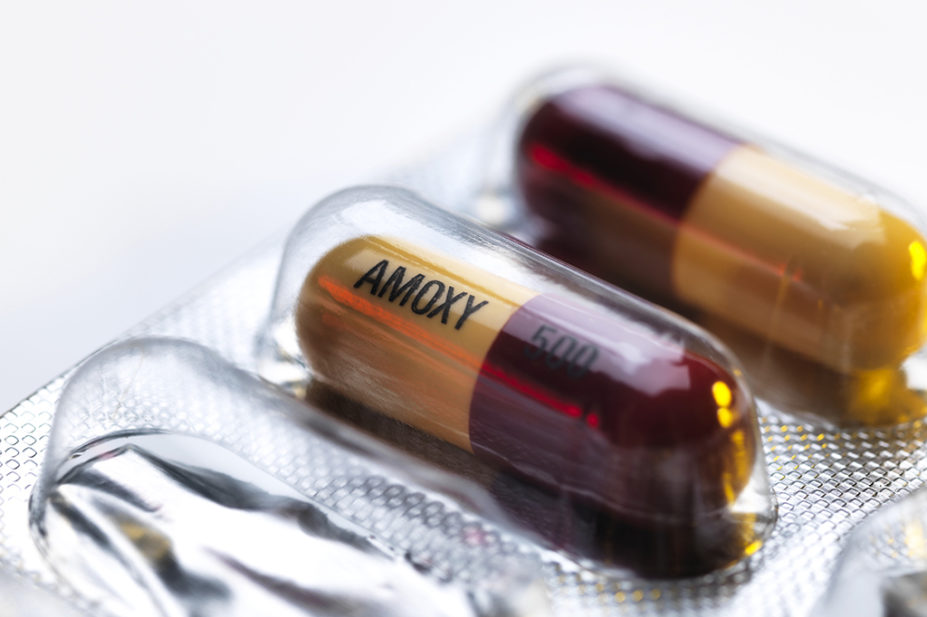
On 14 December 2022, the Medicines and Healthcare products Regulatory Agency added the antibiotics amoxicillin, cefalexin, phenoxymethylpenicillin (penicillin V) and azithromycin oral suspension to the list of medicines that cannot be exported from the UK or hoarded.
On the same day, the CMA said on Twitter that it was “working to establish the facts of what is currently happening in the market and welcome new information as part of our work.”
“We stand ready to take action if there is evidence of anti-competitive behaviour that breaks the law,” it warned.
“People have got real concerns about the price of antibiotics used to treat Strep A, and we want companies to be clear about their obligations under the law. There should be no doubt that it is illegal for a dominant company to charge excessive prices, or for any companies to collude to drive up prices.”
However, manufacturers said that the shortages were the result of a “huge and very sudden spike in demand”.
Martin Sawer, executive director of the Healthcare Distribution Association (HDA), said he knew of one large pharmacy group that ordered one normal month’s worth of Strep A-related antibiotic products in one day.
He added: “Wholesale distributors have been forced to cap orders now to even out distribution. In some cases, HDA wholesale distributors have supplied more packs of these products than they did in the whole of December [2021] in the first few days of December 2022, hence why the products have dropped out of stock this week.”
Leyla Hannbeck, chief executive of the Association of Independent Multiple Pharmacies, said that pharmacies were frustrated at the delays in dealing with supply problems.
“I raised this issue about antibiotic supplies ten days ago.
“Last week, the minister went on national media to say there were no shortages … we were right all along to say there were supply issues.”
She added that the action to protect supplies was “a step in the right direction in terms of managing stock, but why could they not do that earlier?”
The situation has only got worse in the past ten days, she said. “As soon as stock becomes available, bang! It’s gone off the shelves. At least last week we still had some stock — now it’s gone.”
Hannbeck added that the government needed to get ahead of medicines supply issues by sitting down with the profession to make plans when stock levels showed signs of getting low or demand was expected to rise.
She pointed out that the current shortages affected not just children with Strep A, but also anyone who needed the antibiotics for any infection.
On 14 December 2022, the Pharmaceutical Services Negotiating Committee said in a statement that the medicine supply notification and involvement of the CMA “are steps in the right direction”.
The statement continued: “The PSNC has also pressed [the Department of Health and Social Care] to think broadly about what measures could help to manage the current crisis, such as allowing pharmacists more freedom to change strengths or formulations without prescriber approval (outside of Serious Shortage Protocols [SSPs]).
“We have also been clear that more communications are needed to reassure patients and to protect pharmacy staff from the abuse that they are reporting.”
The shortages of particular formulations and types of antibiotics led GP leaders to call for community pharmacists to be given the power to substitute antibiotics if they do not have the prescribed product in stock. This can only happen without going back to the prescriber if the government has issued a serious shortage protocol.

