Shutterstock.com
Open access article
The Royal Pharmaceutical Society has made this article free to access in order to help healthcare professionals stay informed about an issue of national importance.
To learn more about coronavirus, please visit: https://www.rpharms.com/resources/pharmacy-guides/wuhan-novel-coronavirus
Source: Shutterstock.com
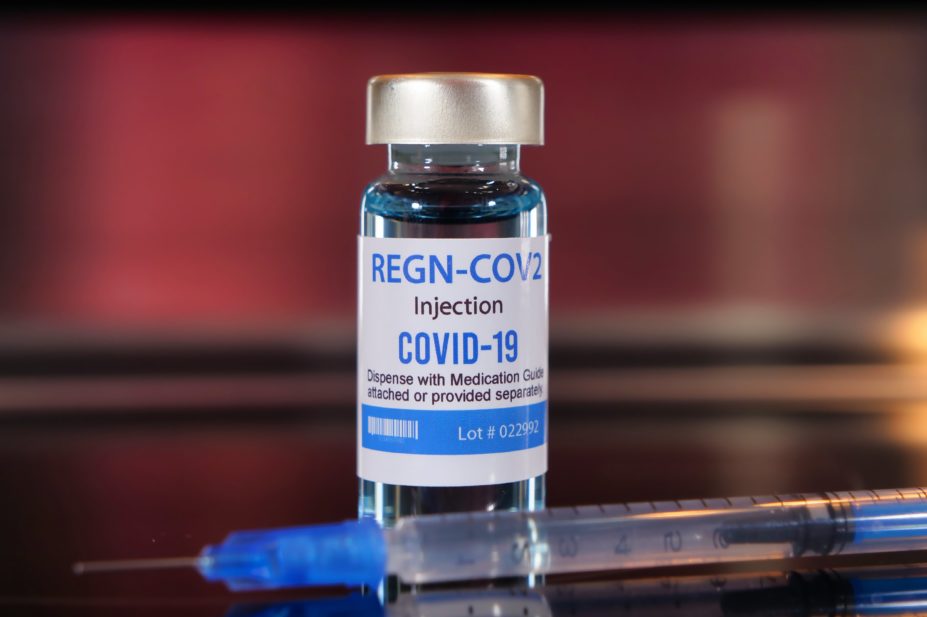
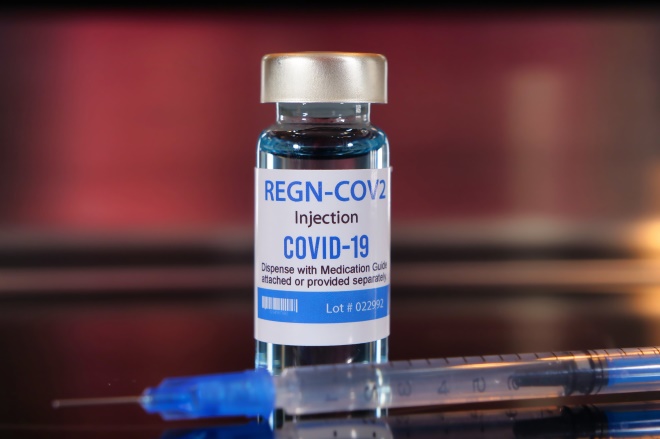
REGN-COV2 combines two antibodies that bind to SARS-COV-2, blocking its entry into host cells
Patients admitted to a hospital trust in North East England with COVID-19 have become the first to receive synthetic neutralising antibodies as part of the ‘Randomised Evaluation of COVID-19 therapy’ (RECOVERY) trial.
North Tees and Hartlepool NHS Foundation Trust introduced the treatment as part of its involvement in the trial overseen by the University of Oxford.
The treatment, REGN-COV2, combines two antibodies that bind to SARS-COV-2, blocking its entry into host cells.
RECOVERY trial researchers announced that the REGN-COV2 arm of the trial would begin phase 3 on 14 September 2020, with patients at North Tees and Hartlepool NHS Foundation Trust the first to receive the treatment.
The trust’s pharmacy aseptic and clinical trials team are responsible for ensuring regulatory compliance with the use of the unlicensed drug, its receipt, appropriate storage, preparation and documentation.
“Together, the team has carried out a significant amount of work to ensure every measure was taken so that this treatment could be delivered to patients,” said Bill Wetherill, associate chief pharmacist in the trust’s aseptic service.
“There have been a number of challenges to overcome,” he added. “This is an experimental drug and — although the likely risks of handling the drug are known — until we have definite proof of its safety in terms of handling, we are preparing the dose within our safety cabinets in the pharmacy clean rooms.
“This allows the product to be protected from contamination and be prepared in an aseptic way, but also ensures protection of the operator from the product in case there are risks we are not yet aware of. It also protects nursing staff and patients on the ward from these risks.”
Mojgan Sani, director of medicines optimisation and chief pharmacist at the trust, said: “The aseptic manufacturing team have played a critical role in helping the trust be the first in the country to offer this medication to patients.”
RECOVERY trial researchers expect that at least 2,000 hospitalised patients will be randomly allocated to receive REGN-COV2 plus usual standard-of-care, and results will be compared with at least 2,000 hospitalised patients who receive standard-of-care on its own.
Researchers will assess the impact of the treatment on mortality 28 days after randomisation, in addition to its impact on hospital stay and the need for ventilation.
Commenting on the launch of the phase III trial, Peter Horby, chief investigator of the RECOVERY trial, said: “The RECOVERY trial was specifically designed so that when promising investigational drugs, such as REGN-COV2, became available they [could] be tested quickly.”