Roger Hutchings / Alamy Stock Photo
Healthcare professionals involved in the supply of Prenoxad (naloxone; Martindale Pharma) injection kits to patients have been advised to contact “all likely holders” of the kits, following an alert that some may be missing needles.
However, service providers have told The Pharmaceutical Journal that they “will never be able to contact every person who has been issued with a kit” as some are provided anonymously.
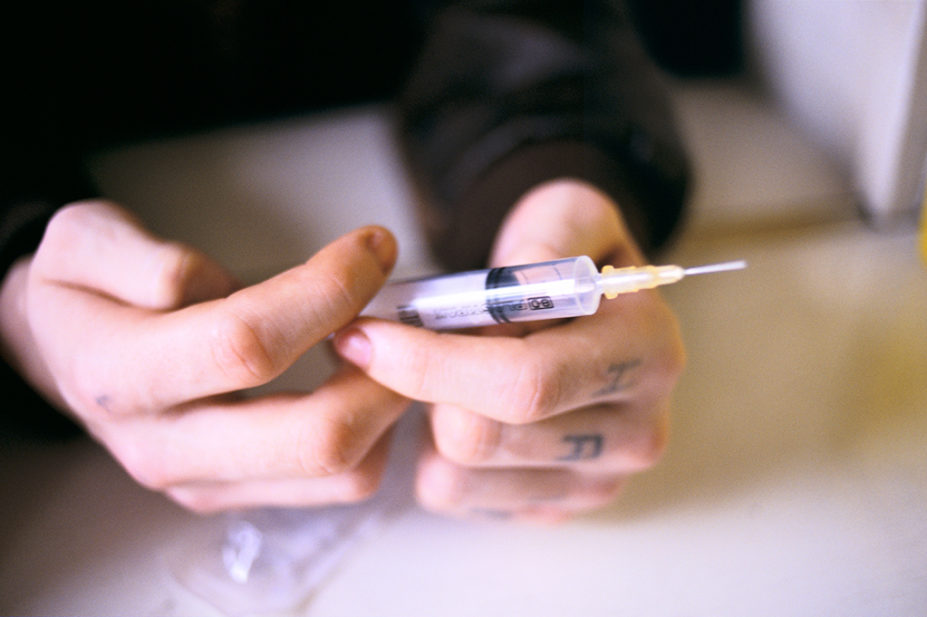
The Medicines and Healthcare products Regulatory Agency (MHRA) issued a National Patient Safety Alert for 24 batches of Prenoxad kits on 10 November 2022 after Martindale Pharma alerted the regulator to an issue with “a limited number” of kits in France that contained fewer than two needles.
Prenoxad kits are usually packed with two needles, along with a pre-filled syringe containing naloxone hydrochloride, which reverses the effects of an opioid overdose.
“If no needles are present in the kit, there is a risk that patients, members of the public and/or healthcare professionals may not be able to administer life-saving doses of naloxone from these kits in an emergency,” the patient safety alert says.
“This may impede the treatment for a patient with an opioid overdose, which may result in delay to intervention and possible death.”
As a result, the MHRA has advised anyone with a Prenoxad kit to check it “against the batches specified” in the alert and quarantine any with missing needles. The batches date back to March 2020.
The alert adds that “where healthcare professionals, service providers and local teams (including those involved in needle and syringe programmes), are able to contact patients and members of the public who have been supplied with Prenoxad, they should inform them to check their kits to ensure they contain two needles in each kit”.
“This action will depend on the local procedures for record keeping, but efforts should be made to inform all likely holders of Prenoxad.”
Suzanne Gallagher, helpline development officer at the charity Scottish Families Affected by Alcohol and Drugs, told The Pharmaceutical Journal: “People have the right to anonymously access a naloxone kit for whatever reason and these individuals may not be contactable.
“You will never be able to contact every person who has been issued with a kit, but services who provide naloxone have the means to contact their clients and also ensure that accurate information on how to externally check your kit is communicated through all communications to reach the wider public.”
Roz Gittins, director of pharmacy at Humankind, said the charity has “been checking all of the kits we have in stock”, adding that “whilst we have some of the affected batches, so far all contain the required two needles”.
“Where we record the person’s name, we are in the process of contacting individuals to inform them about how to check the kits and, if we need to, we’ll arrange for replacements to be issued. However, sometimes we supply kits without taking the person’s details; for example, via our needle and syringe provision schemes, which the person may wish to access anonymously,” she said.
“Therefore, we’ve produced posters, which are on display in our services and via social media to help raise awareness and inform people that they need to check their kits and what action they need to take.”
Nicola Torr, former branch pharmacist at The Pharmacy Group in Wakefield, which became one of the top naloxone-supplying pharmacies in England in 2021, said “a significant proportion” of needle exchange patients at the pharmacy do not have a phone, regularly change their phone numbers or “don’t have an address to enable contact, so we are dependent on patients accessing the service and communications in person”.
She added that her “concern is that, if these kits go back to March 2020, how many kits this affects and how long it’s taken to be realised”.
“Two needles are usually supplied with each kit in case one of them snaps,” she said. “Many patients don’t open the kit until needed and, if there are no other needles available, this could lead to a negative outcome.”
A report from the Scottish Drug Deaths Taskforce found that the number of naloxone kits handed out in Scotland increased by “at least” 31% in 2020 compared with the previous year.
Alison Cave, chief safety officer at the MHRA, said that checking the kits “is vitally important”, adding that the MHRA has “taken prompt action to ensure kits with missing needles will no longer be given to patients.
“There are no concerns about the medicine in these kits and other products containing naloxone have not been affected by this manufacturing error,” she said.
A spokesperson for Ethypharm, which owns Martindale Pharma, said the alert was “issued following receipt of three reports of missing needles, all on a single batch distributed in France”, adding that “no patients were affected in any of the three reported incidents”.
“Significant improvements to our packing in-process controls to eliminate risk of missing needles have already been made thus, batches not listed … will not require checking,” they said.
1 comment
You must be logged in to post a comment.



It is important to highlight to anyone using needle and syringe programmes in pharmacies to check their kits as they may not be engaged with drugs services. It is a balance to try to reach the most disengaged people and pharmacy teams have the privilege to be able to do that. I think the bottom line from this is, spread the word as much as you can. I have known people to use 1ml insulin syringes to withdraw naloxone when no needle was available (because it had been used). I have also heard of nasal administration of the IM solution. Not advocating either approaches, but just to illustrate the ability of people to be innovative in a crisis. What we need of course is more pharmacies supplying naloxone, that is important to have more even availability across the country and national commissioning would be ideal.