Shutterstock.com
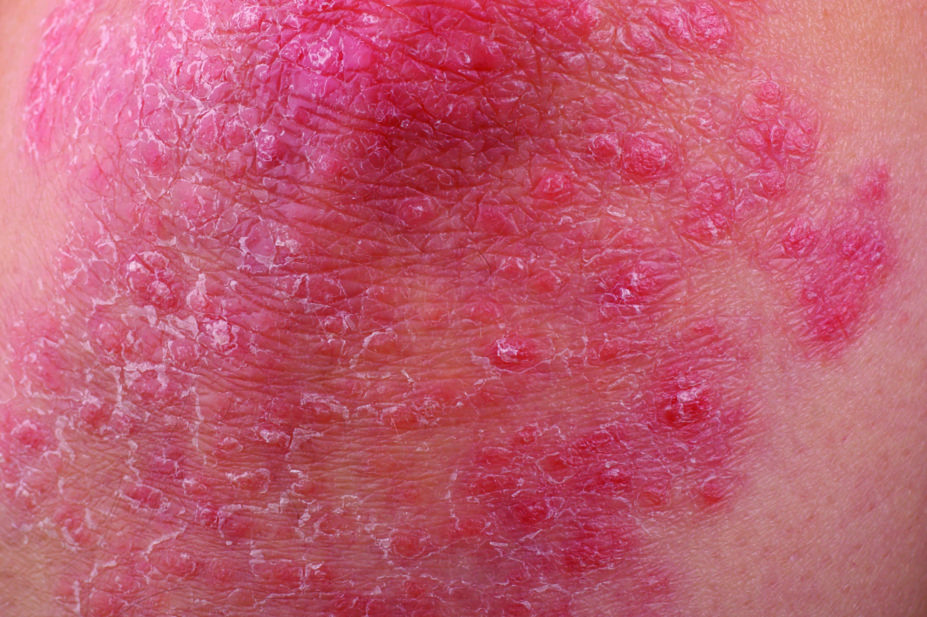
The US Food and Drug Administration (FDA) has approved Tremfya (guselkumab) for the treatment of adults with moderate to severe plaque psoriasis.[1]
Developed by US biotechnology company Janssen Biotech, Tremfya is the first and only biologic approved that selectively blocks interleukin (IL)-23 — a cytokine that plays a key role in plaque psoriasis.
Approval comes after an expedited regulatory review following application of an FDA Priority Review Voucher.
The biologic is administered as a 100mg subcutaneous injection every eight weeks, following two starter doses at weeks 0 and 4. In clinical studies of more than 2,000 patients, those receiving Tremfya experienced at least 90% improvement in skin clearance and greater improvement in symptoms of plaque psoriasis, including itch, pain, stinging, burning and skin tightness when compared with a placebo at week 16.
Tremfya demonstrated superior results in skin clearance compared with Humira (adalimumab) in head-to-head analysis at weeks 16, 24 and 48. Seven out of ten patients receiving Tremfya achieved at least 90% improvement in skin clearance at week 16. Nearly three quarters of Tremfya-treated patients demonstrated at least 90% clearer skin at week 48.
Dermatologist Andrew Blauvelt, president of Oregon Medical Research Center, and study investigator said: “Tremfya represents a significant milestone in the treatment of moderate to severe plaque psoriasis as evidenced by the proven skin clearance demonstrated in the majority of study patients receiving this IL-23-specific therapy at week 16 and up to week 48.”
“We continue to make progress in understanding the science of psoriasis and the important role IL-23 plays in the pathogenesis of this disease,” he added.
A spokesperson for Janssen said the company will work closely with payers, providers and pharmacy benefit managers to ensure Tremfya is broadly accessible and affordable for patients and that the cost for payers is competitive with currently available biologic therapies for psoriasis.
References
[1] Janssen Biotech, Inc. Janssen announces U.S. FDA approval of Tremfyaâ„¢ (guselkumab) for the treatment of moderate to severe plaque psoriasis. 13 July 2017. Available at: https://www.jnj.com/media-center/press-releases/janssen-announces-us-fda-approval-of-tremfya-guselkumab-for-the-treatment-of-moderate-to-severe-plaque-psoriasis (accessed July 2017)