Wikimedia Commons
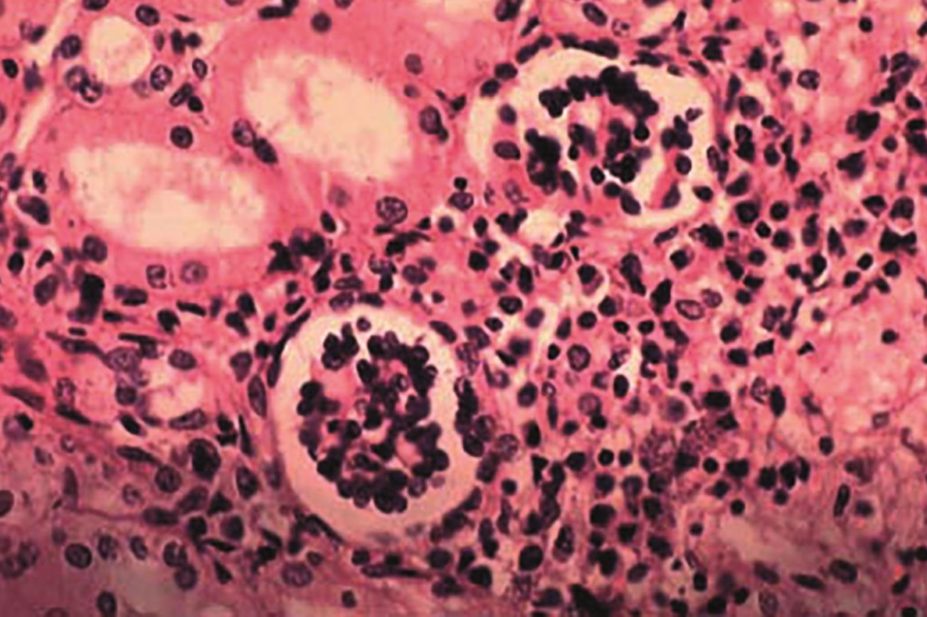
The efficacy and safety of amphotericin plus miltefosine to treat visceral leishmaniasis (VL) patients with HIV will be examined in a Phase III randomized clinical trial in Ethiopia.
The 132-patient open-label study will compare the efficacy and safety of the polyene antifungal amphotericin (AmBisome)(30mg/kg total dose) plus the antineoplastic agent miltefosine (50mg or 100mg per day depending on the patient’s weight) with amphotericin alone (40mg/kg total dose).
It marks the first trial in Africa to implement the World Health Organization’s recommendations for HIV-VL treatment, said AfriCoLeish, an international research & development consortium, formed by six organisations in East Africa and Europe, which includes the London School of Hygiene and Tropical Medicine, and Drugs for Neglected Diseases initiative.
Total treatment duration is 28 days, if the tests show that patients are parasite-negative (or 56 days in case of extended treatment). Afterwards, patients will start a secondary prophylaxis treatment aimed at preventing VL relapses and a one-year follow-up phase.
“The risk of death from VL is nine times higher in patients who are co-infected with HIV. VL also accelerates the progression of HIV,” the consortium said. Some 300,000 new HIV-VL cases are reported each year in 35 countries around the world, with 90% of them occurring in Ethiopia, Sudan, Kenya, Bangladesh, India, Nepal, and Brazil.
You may also be interested in

Recognition and management of whooping cough
Everything you need to know about meningitis B
