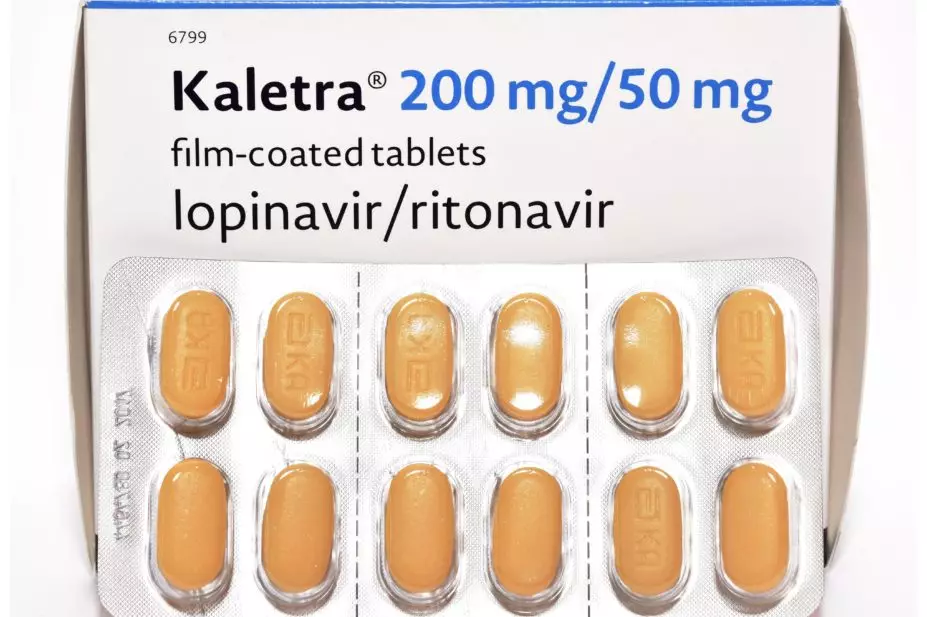
DR P. MARAZZI / SCIENCE PHOTO LIBRARY
Open access article
The Royal Pharmaceutical Society has made this article free to access in order to help healthcare professionals stay informed about an issue of national importance.
To learn more about coronavirus, please visit: https://www.rpharms.com/resources/pharmacy-guides/wuhan-novel-coronavirus
Source: DR P. MARAZZI / SCIENCE PHOTO LIBRARY
Researchers from the World Health Organization examined 20 observational studies of the effects of antiretroviral medicines, such as lopinavir/ritonavir, on the clinical outcomes of several respiratory diseases
There is insufficient evidence in the literature to indicate whether antiretroviral medicines, such as lopinavir/ritonavir (LPV/r), are effective in treating or preventing coronavirus disease infections, the authors of a systematic review published in the Journal of the International AIDS Society (26 March 2020) have concluded[1]
.
The team, made up of researchers from the World Health Organization, identified 20 observational studies that reported the effects of antiretroviral medicines on clinical outcomes in severe acute respiratory syndrome (SARS; n=3), Middle East respiratory syndrome (MERS; n=6), and COVID-19 (n=11), along with one randomised trial in COVID-19.
The randomised trial showed no clinical benefit of LPV/r on clinical outcomes. Across the remaining studies, the evidence was inconclusive and the certainty of evidence for outcomes was low.
LPV/r was suggested for potential use in COVID-19 in rapid guidance from Wuhan University, China, based on previous experience against SARS and MERS. However, the researchers noted it was important that antiretroviral drugs were not directed away from people with HIV.
Several randomised trials are planned to assess the safety and efficacy of antiretroviral medicines for the treatment of coronaviruses.
“While the conduct of such trials is challenging, high-quality evidence is needed to improve clinical and programmatic decisions to use antiretroviral drugs for current and future coronavirus outbreaks,” the researchers concluded
.
References
[1] Ford N, Vitoria M, Rangaraj, A et al. J Int AIDS Soc 2020;23(4):e25489. doi: 10.1002/jia2.25489