Sean Aidan Calderbank / Shutterstock.com
The recent outbreak of meningococcal disease in Kent dominated national news headlines, with the spread of the condition being described as “unprecedented and explosive”.
As of 12:30pm on 1 April 2026, 21 cases had been confirmed to be caused by the meningitis B strain, while 2 people had sadly died of the disease.
The situation in Kent led to a large-scale vaccination programme, with prophylactic antibiotics being given to thousands of people. Although the peak of the outbreak is reported to have passed, it has raised questions about how meningitis is transmitted, its warning signs and the effectiveness of the vaccine.
Meningitis
Meningitis vaccines
- What types of vaccines are available?
- How effective are the vaccines?
- What is the uptake of meningitis vaccines?
- Should MenB vaccination eligibility be expanded?
Kent outbreak and preventative antibiotics
- Where did the outbreak come from?
- Is the strain from this outbreak different to other types of meningitis?
- What antibiotics are being given?
- Are there any antimicrobial resistance considerations with the antibiotics?
Best practice
Meningitis
What is meningitis?
Meningitis is the inflammation of the meninges, membranes that surround the brain and spinal cord1. Bacterial or viral infections are the most common causes of meningitis; fungal, parasitic and non-infective causes are less common.
Viral meningitis is more common than bacterial meningitis: results of a 2018 UK study of 1,126 patients aged over 16 years revealed that 36% of cases were viral, 16% were bacterial and 42% had an unknown cause2. Viral meningitis usually resolves on its own in around seven to ten days with minimal long-term consequences, whereas bacterial meningitis is an immediate life-threatening emergency, potentially leading to sepsis, and has an estimated one in six mortality rate1. There are several different causes of bacterial meningitis (see Box).
Box: Causes of bacterial meningitis
The four most common causes of bacterial meningitis are responsible for around half of deaths from meningitis1. They are caused by the following bacteria:
- Streptococcus pneumoniae (pneumococcal);
- Neisseria meningitidis (meningococcal);
- Group B streptococcus;
- Haemophilus influenzae.
Other notable causes include: Escherichia coli, listeria, salmonella, klebsiella and Mycobacterium tuberculosis
Data suggest that one in five people who contract bacterial meningitis are expected to experience long-term effects, which can include difficulties with language, memory and communication, hearing loss and seizures1.
What are the different strains?
Meningitis has several genetically distinct variants of causative organisms, referred to as strains. For example, Neisseria meningitidis has several serogroups: A, B, C, W, X and Y.
Identifying the causative strain of meningitis is vital for managing outbreaks and disease course, as the implicated strain influences spread, severity of illness, vaccine efficacy and treatment choice. The serogroup is identified by collecting cerebrospinal fluid from patients via lumbar puncture and its genes are analysed using real-time polymerase chain reaction (PCR) analysis.
At the time of going to press, all of the confirmed meningitis cases in Kent had been identified as meningitis B, the leading cause of meningococcal disease. Of 378 confirmed cases of meningococcal disease in England in 2024/2025, 83% (n=313) were determined to be caused by meningitis B3.
How does it spread?
Meningococcal bacteria (i.e. N. meningitidis) are thought to be carried in the nose and throat of one in ten people4. This is usually harmless and can contribute to the development of immunity against these pathogens; however, the bacteria can enter the bloodstream, causing meningococcal disease.
Bacterial meningitis requires prolonged close contact for transmission — it is spread by respiratory droplets via coughing, sneezing and kissing, or by sharing items such as utensils and cutlery. Of particular note from the Kent student outbreak, sharing drinks and vapes can also transmit the disease.
Wearing face masks may be useful in close-contact situations to stop the spread of respiratory droplets, but they do not eliminate risk. At publication, the UKHSA was not recommending the use of facemasks in educational settings, but has advised people to practice good hand hygiene and to avoid sharing items.
What are the symptoms?
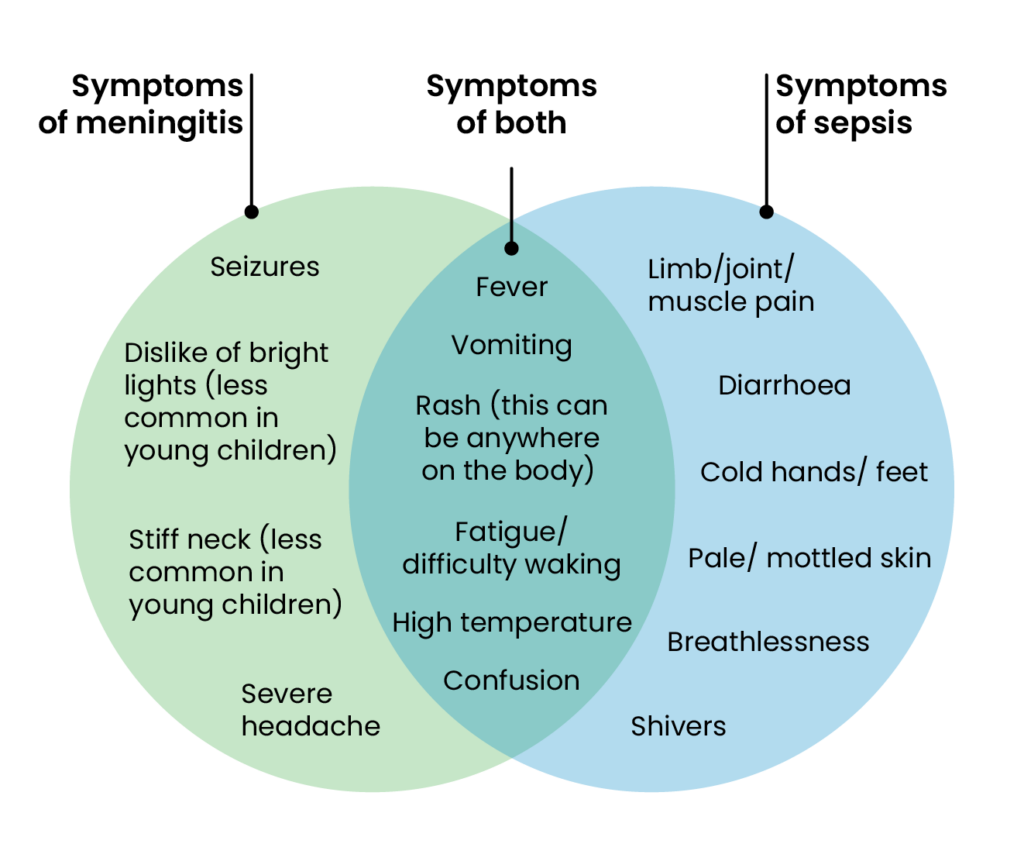
In bacterial meningitis, bacteria often enter the bloodstream, leading to sepsis. However, it should be noted that meningitis can occur without sepsis, which is its own distinct condition. It is important to be aware of the symptoms of both conditions and their overlap (see Figure 1).
Sepsis and meningitis symptoms can appear in any order and do not always occur together. In some cases, especially in young children, sepsis symptoms can appear first, which can be crucial for early identification.
Figure 1: Symptoms of meningitis and sepsis
Meningitis Research Foundation
Figure 2: Photographs of meningitis and sepsis rashes
Meningitis vaccines
What vaccines are available?
Different vaccines protect against different specific viral or bacterial causes of meningitis. Vaccines of note include: MenB (for group B meningococcal); MenACWY (for groups A, C, W, Y meningococcal); the 6-in-1 vaccine which includes Hib (for haemophilus influenzae type b), and the pneumococcal vaccine5–8.
In the UK, the MenACWY vaccine was introduced in 2015 and is routinely offered as a single dose to teenagers aged 13 and 14 years (in UK school year 9)6. It was not found to be cost-effective to vaccinate infants and younger children, as most meningococcal disease in this patient group is caused by meningitis B.
In September 2015, the UK became the first country in the world to routinely vaccinate against meningitis B. Children born after 1 May 2015 receive three doses of the MenB vaccine when they are 8 weeks, 12 weeks and 1 year old. The two vaccines available for meningitis B are Bexsero (GSK) and Trumenba (Pfizer).
In response to the outbreak in Kent, the UKHSA, NHS England and the government commenced a coordinated MenB vaccination programme. As of 10:00 on 30 March 2026, more than 11,400 vaccinations had been given.
The MenB vaccine is being offered to anyone that previously received antibiotic prophylaxis, including:
- Close contacts of any confirmed or suspected meningitis cases;
- University of Kent students and some staff living or working in the halls of residence;
- Students from other Kent universities where there have been cases or who have had close contact;
- Sixth form students in Kent schools and colleges where there have been confirmed/suspected cases of meningitis;
- Anyone who visited Club Chemistry in Canterbury between 5 March and 15 March 2026.
The vaccine is being given as two doses. The second dose should be given at least four weeks after the first for the best results. It takes at least two weeks after the second dose for enough antibodies to be produced to confer good protection.
Vaccination is being deployed as an additional step to protect individuals that have been exposed from developing severe disease. Antibiotic prophylaxis remains the most important intervention and is effective at stopping the spread in 90% of cases.
The outbreak caused a surge in people seeking private MenB vaccination — both in Kent and in other parts of the country — with 20,000 doses for private practice released by the NHS to support continuity of private MenB provision. However, Olivier Picard, chair of the National Pharmacy Association, said is it “unlikely” this will meet demand, as pharmacies are getting “hundreds of thousands of patients seeking vaccination in light of the outbreak in Kent”.
How effective are the vaccines?
According to UKHSA guidance, the MenB infant programme in England has reduced cases of meningitis B by around 75% in vaccinated individuals9. All cases reduced following the commencement of the programme (see Figure 3).
Figure 3: Cases of meningitis in the UK have dropped since vaccinations schemes were introduced in 2015
Cases of strains A, C, W, Y have decreased significantly since the introduction of the vaccine in 2015. In 2022, a study evaluating the impact of the first four years of the MenACWY programme in England reported that overall vaccine effectiveness against MenCWY disease combined was 94% (there were no cases of meningitis A during the reporting period)10.
Unlike the MenACWY vaccine, the MenB vaccine does not prevent individuals from carrying or spreading meningococcal bacteria, therefore it does not provide herd immunity to the wider population. However, it does reduce the risk of developing illness in those carrying the bacteria and reduces severity of illness.
What is the uptake of meningitis vaccines?
In 2024/2025 in England, coverage of the MenB vaccine in children aged 12 months was 91%, a 0.4% increase on the previous year (see Figure 4)11.
Figure 4: Uptake for the MenB vaccine in England in children aged 12 months from financial year ending 2018 to 2025
The MenACWY vaccine is given in schools to children aged 13 and 14 years, in UK school year 9, with catch-up doses offered to children aged 14 and 15 years, in year 10.
For year 10 students, coverage was 75.1% in the 2024/2025 academic year, 2.1% higher than the 2023/2024 cohort; however, rates have not returned to pre-COVID highs of 88% (see Figure 5)10.
Figure 5: Uptake for the MenACWY vaccine in adolescents in school years 9 and 10 by academic year from 2015 to 2025
Should eligibility for the MenB vaccination be expanded?
In 2014, the Joint Committee on Vaccines and Immunisation (JCVI) decided against offering routine meningitis B vaccination for older groups, such as teenagers, because it was determined not to be cost-effective.
This decision has attracted some controversy in the context of the Kent outbreak because the age group primarily affected (teenagers and young adults) have not received the MenB vaccine, with parents of individuals that lost their lives in the outbreak calling for wider availability of the vaccine.
On 17 March 2026, health secretary Wes Streeting asked the Joint Committee on Vaccination and Immunisation (JCVI) to reconsider the case for older children to be given the MenB vaccine.
It is essential that the authorities fully utilise the capacity neighbourhood pharmacies can bring to tackling patient concern
Leyla Hannbeck, chief executive of the Independent Pharmacies Association
And, in the wake of the outbreak, the UKHSA advised JCVI to reconfirm its guidance is correct12. In a technical report published on 24 March 2026, it noted that university students — particularly first-year students — are at 11-times higher risk of meningococcal disease than non-students of the same age13.
“This relative risk is rather greater than I would have guessed,” commented Paul Hunter, professor in medicine at the University of East Anglia.
“To my mind, this relative risk is sufficient that JCVI should model not only giving the vaccine to all adolescents but also model giving it to first-year university students.”
The Independent Pharmacies Association has called for a nationwide catch-up vaccination programme for MenB. Leyla Hannbeck, its chief executive, said: “It is essential that the authorities fully utilise the capacity neighbourhood pharmacies can bring to tackling patient concern.
“Whilst most young children will have had the meningitis B vaccine as part of childhood immunisation since 2015, many teenagers and young adults will not have this vital protection.
“The NHS needs to urgently commission pharmacies to deliver an immediate catch-up programme targeted at university students and teenagers born before 2015.”
Meanwhile, the Company Chemists’ Association and National Pharmacy Association have each called for pharmacy involvement in a catch-up scheme for teenagers who have missed doses of the MenACWY vaccine.
Kent outbreak and preventative antibiotics
Where did the outbreak come from?
Meningitis is a notifiable disease, meaning any suspected cases must be reported to the UK Health Security Agency (UKHSA) by medical practitioners. On 13 March 2026, the UKHSA was first alerted to a possible case involving a student from the University of Kent. The following day, British authorities were notified of a meningitis case in a French exchange student who had recently returned to the Paris region from the University of Kent. Three more University of Kent students were admitted to hospital on the same day with suspected meningitis. On 15 March 2026, the UKHSA issued a rare public health alert. In the days following, cases continued to rise and included students from local sixth forms.
The outbreak is thought to be linked to an event at Club Chemistry, a nightclub in Canterbury, which has since closed temporarily following the outbreak. While specifics about the exact spread of these infections are yet to be fully understood, known patients are said to have visited the venue on 5, 6 or 7 March 2026, attending what have since been termed ‘super-spreader’ events.
Is the strain from this outbreak different to other types of meningitis?
At the time of publication, 18 of the 21 confirmed cases were identified as being the outbreak strain subtype P1.12-1,16-183.
On 24 March 2026, the UKHSA published a technical report focusing on the serogroup of meningitis identified in this outbreak, which states that this strain is a fairly new subtype that has been present in England since 2020, but with “multiple potentially significant genetic differences”.
One of the differences is in some surface proteins called porins that are used to identify different strains
Andrew Smith, professor and honorary consultant microbiologist at the University of Glasgow
“One of the differences is in some surface proteins called porins that are used to identify different strains,” said Andrew Smith, professor and honorary consultant microbiologist at the University of Glasgow, Scotland.
The genetic information from the report suggests changes to a gene that could “affect the infection behaviour of this bacteria”, he added, but more research is needed.
The UKHSA says its analysis suggests the strain is susceptible to antibiotics and covered by Bexsero (GSK) and Trumenba (Pfizer), the two meningitis B vaccines used in the UK.
What antibiotics are being given?
People at risk of contracting meningitis are being given ciprofloxacin, a broad-spectrum antibiotic. This includes close contacts of people with suspected cases, those who have attended settings assessed as high risk, such as people who attended Club Chemistry between 5 and 7 March 2026, and sixth-form students (in UK school years 12 and 13) in schools and colleges in Kent where confirmed or probable cases have been identified.
According to UKHSA, a single course of antibiotics is “highly effective in preventing the contraction and spread of this disease in 90% of cases”12.
It adds that prophylactic antibiotics are a “key intervention” that could help to halt the spread of the outbreak.
As of 10:00 on 30 March 2026, more than 13,500 doses of antibiotics had been administered.
Are there any antimicrobial resistance considerations with the antibiotics?
The prophylactic antibiotics are being described as playing a “crucial role” in preventing the spread of the disease12. But thousands of doses have been handed out, which raises the question of antimicrobial resistance becoming a concern.
“Decisions around antibiotic use must always consider the potential risk of driving antimicrobial resistance if these medications are used in an inappropriate way,” a spokesperson for the UKHSA told The Pharmaceutical Journal.
“The groups that have been identified as eligible for antibiotic chemoprophylaxis have been identified following careful risk assessment and, at this time, we do not recommend antibiotics to be prescribed as chemoprophylaxis to people outside these groups.”
Best practice for pharmacists
Pharmacists play an important role in making the public aware of the symptoms of meningitis, as well as promoting vaccination programmes that prevent the spread of the disease.
A briefing document published by Community Pharmacy Greater Manchester on 19 March 2026 says that pharmacists should maintain “a high level of suspicion”, because flu-like symptoms can worsen rapidly if the patient does have meningitis.
It lists several red-flag symptoms that warrant an immediate referral to 999 or A&E, including:
- Non-blanching rash;
- Reduced consciousness, marked drowsiness, or difficult to wake;
- Seizures;
- Rapid deterioration;
- Pale, mottled, or blotchy skin;
- Severe headache with neck stiffness or photophobia;
- Confusion or delirium;
- Signs of sepsis or significant systemic illness.
Patients who do not have these red-flag symptoms are advised to keep an eye out for these signs occurring in the next 10–14 days.
Hospital pharmacists who are treating patients with meningococcal disease can review The Pharmaceutical Journal’s learning content about managing the condition.
Useful resources
- 1.Meningitis . World Health Organization. Accessed March 2026. https://www.who.int/news-room/fact-sheets/detail/meningitis
- 2.McGill F, Griffiths MJ, Bonnett LJ, et al. Incidence, aetiology, and sequelae of viral meningitis in UK adults: a multicentre prospective observational cohort study. The Lancet Infectious Diseases. 2018;18(9):992-1003. doi:10.1016/s1473-3099(18)30245-7
- 3.Invasive meningococcal disease in England: annual laboratory-confirmed reports for epidemiological year 2024 to 2025. UK Health Security Agency. November 2025. Accessed March 2026. https://www.gov.uk/government/publications/meningococcal-disease-laboratory-confirmed-cases-in-england-2024-to-2025/invasive-meningococcal-disease-in-england-annual-laboratory-confirmed-reports-for-epidemiological-year-2024-to-2025
- 4.Meningococcal meningitis. Meningitis Research Foundation. January 2025. Accessed March 2026. https://www.meningitis.org/about-meningitis/bacterial-meningitis/meningococcal-meningitis/
- 5.MenB vaccine for children. NHS . March 2024. Accessed March 2026. https://www.nhs.uk/vaccinations/menb-vaccine-for-children/
- 6.MenACWY vaccine. NHS. February 2024. Accessed March 2026. https://www.nhs.uk/vaccinations/menacwy-vaccine/
- 7.6-in-1 vaccine. NHS. June 2023. Accessed March 2026. https://www.nhs.uk/vaccinations/6-in-1-vaccine/
- 8.Pneumococcal vaccine. NHS. June 2023. Accessed March 2026. https://www.nhs.uk/vaccinations/pneumococcal-vaccine/
- 9.Meningococcal B vaccination programme for infants: information for healthcare practitioners. UK Health Security Agency. August 2025. Accessed March 2026. https://www.gov.uk/government/publications/meningococcal-b-vaccine-information-for-healthcare-professionals/meningococcal-b-vaccination-programme-for-infants-information-for-healthcare-practitioners
- 10.Summary of MenACWY coverage by year group. UK Health Security Agency. January 2026. Accessed March 2026. https://www.gov.uk/government/publications/meningococcal-acwy-immunisation-programme-vaccine-coverage-estimates/meningococcal-acwy-menacwy-vaccine-coverage-for-adolescents-in-england-academic-year-2024-to-2025#:~:text=Summary%20of%20MenACWY%20coverage%20
- 11.MenB (England) — Coverage in children aged 12 months. UK Health Security Agency. October 2025. Accessed March 2026. https://www.gov.uk/government/statistics/cover-of-vaccination-evaluated-rapidly-cover-programme-annual-reports/vaccination-coverage-statistics-for-children-aged-up-to-5-years-england-cover-programme-report-april-2024-to-march-2025#menb-england
- 12.Meningitis outbreak: what you need to know. UK Health Security Agency. March 2026. Accessed March 2026. https://ukhsa.blog.gov.uk/2026/03/18/meningitis-b-outbreak-what-you-need-to-know/
- 13.Mandal S, Campbell H, Ribeiro S, et al. Risk of invasive meningococcal disease in university students in England and optimal strategies for protection using MenACWY vaccine. Vaccine. 2017;35(43):5814-5818. doi:10.1016/j.vaccine.2017.09.024
You may also be interested in

Case-based learning: meningitis

Everything you need to know about mpox