With a new ‘recombinant’ strain being reported in England in December 2025, healthcare professionals’ focus has returned to mpox.
In October 2025, the UK Health Security Agency (UKHSA) urged gay, bisexual and other men who have sex with men to come forward for mpox vaccination, after cases of locally acquired clade Ib mpox were identified in Europe for the first time, suggesting community transmission. The UKHSA’s latest technical assessment, published in October 2025, indicates the high probability of importation of cases to the UK.
Pharmacists have a role to play in identifying potential cases of mpox and increasing public awareness of the vaccine, as well as its high efficacy rate.
This article gives a brief overview of transmission, prevention and identification of mpox and will be updated periodically. It provides answers to the following questions:
- What is mpox?
- What do we know about the spread of mpox?
- How is mpox transmitted?
- What are the clinical features of mpox?
- What strains of mpox are currently circulating?
- Who is most at risk of infection?
- How many cases are there in the UK?
- What treatments are available?
- Who can get the mpox vaccine?
- How effective is the mpox vaccine?
- What is best practice for pharmacists in preventing mpox?
What is mpox?
Mpox — previously known as monkeypox — is a viral infection that causes a rash, often beginning on the face or genital area, with flu-like symptoms such as a fever, muscle pains and headache. It is an enveloped double-stranded DNA virus and part of the Orthopoxvirus genus, which is the same family as the smallpox virus.
There are two major genetic groups (i.e. clades) of the virus: clade II mpox (formerly known as ‘West African clade’); and clade I mpox (formerly known as ‘Central African or Congo basin clade’), which is considered to be more severe.
What do we know about the spread of mpox?
The first human case of mpox was detected in 1970 in the Democratic Republic of Congo1, and the disease was originally most prevalent in West and Central Africa. Its natural animal reservoir is likely to be a small rodent such as a squirrel, dormouse or giant pouched rat.
There were 558 suspected cases in Nigeria between 2017 and April 2022, during which time the virus was exported to several countries, including the United States, the UK, Israel and Singapore.
Since May 2022, cases of human mpox have been reported in multiple countries worldwide, including in the UK. These cases are primarily caused by clade IIb, which is mostly spread through close physical contact such as sexual activity. A total of 3,732 ‘highly probable’ and confirmed cases were reported in the UK by 31 December 2022, with 3,553 of those cases being in England. Questionnaire data from 1,050 confirmed cases in England found that just under 97% of these cases were reported in people who were gay, bisexual, or other men who have sex with men.
How is mpox transmitted?
Mpox can be spread through:
- Close physical or intimate contact with people who may have mpox;
- Sharing bedding or towels with people who may have mpox;
- Coughs and sneezes of a person with mpox.
Although mpox has not been found in animals in the UK, it can be caught via infected animals through:
- Being bitten;
- Touching wild or stray animals, including animals that appear unwell or are dead;
- Eating or touching meat from wild animals that has not been cooked through.
There is some concern that mpox can also be transmitted asymptomatically. In August 2023, the UKHSA launched the £1m PResymptomatIc MonkEypox (PRIME) study, funded by the Medical Research Council, to examine whether people who tested positive for mpox but did not have symptoms that would be able to pass the disease on. At the time, the UKHSA said the results would support future vaccination and testing strategies. To date, no results have been reported.
What are the clinical features of mpox?
The incubation period varies: symptoms tend to start within a week of exposure but can begin anywhere between 1–21 days from exposure.
Most symptoms are mild and self-limiting, with the prodromal phase lasting between one and five days, followed by a second phase, which is commonly characterised by the appearance of a rash.
The second phase starts as raised spots and spreads rapidly to other parts of the body. It develops from macules and papules to vesicles and pustules, which then crust over and fall off in two to three weeks. Lesions can occur on the oral mucous membranes, genitals, conjunctivae and, in some patients, the cornea, which can present as a single lesion or thousands of lesions.
Other common symptoms include a fever, sore throat, tiredness and headache: see Figure 1 for a full list of symptoms.
Figure 1: Symptoms of mpox
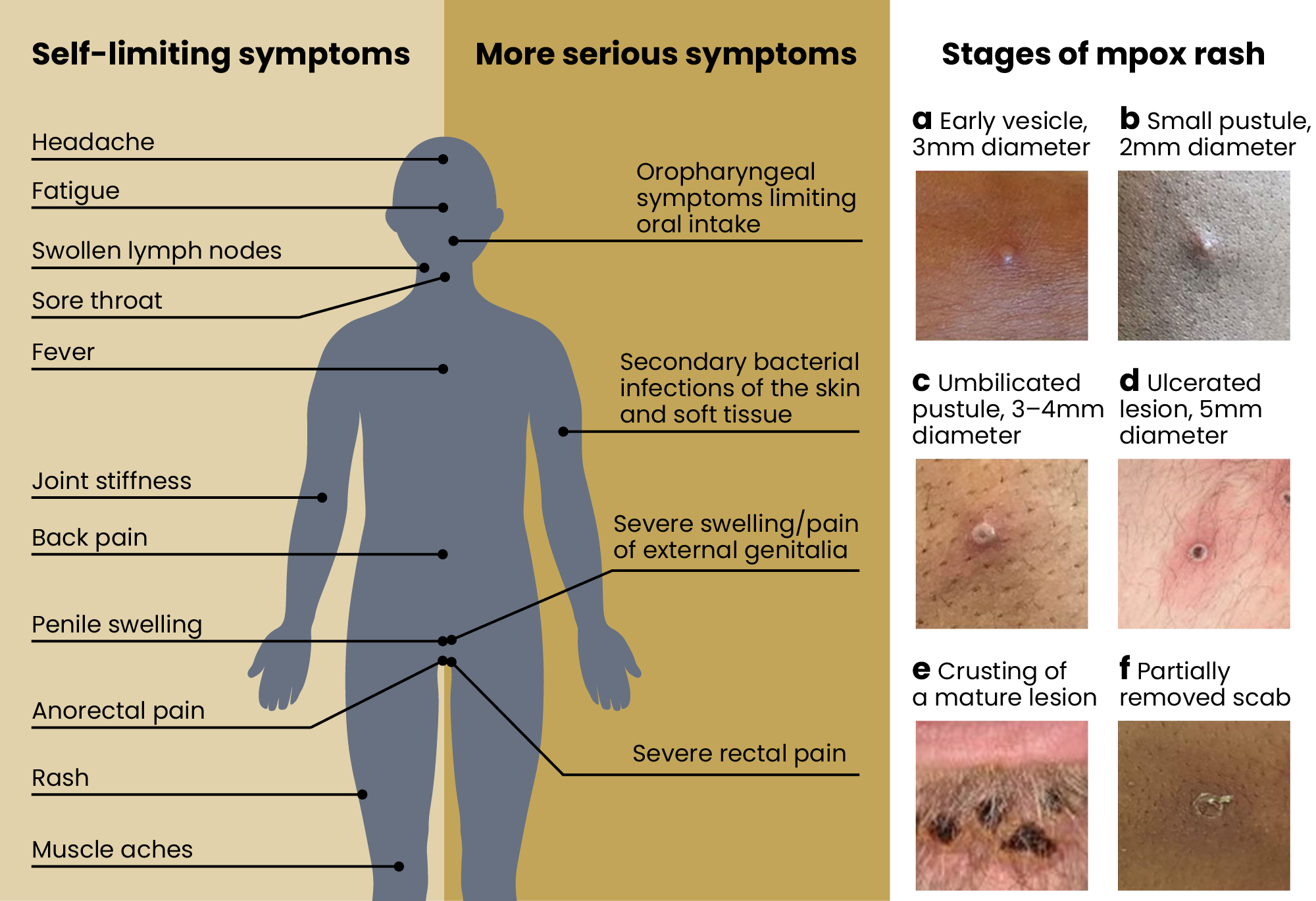
The Pharmaceutical Journal/Photographs: courtesy of the UK Health Security Agency
Symptoms last for between two and four weeks; however, people with weakened immune systems may experience symptoms for longer.
Mpox can be easily confused with chickenpox and can only be diagnosed following laboratory testing of swabs taken from a vesicle or ulcer.
What strains of mpox are currently circulating?
In December 2025, the UKHSA identified a new “recombinant” mpox virus in England, which contained genetic elements of both clade Ib and IIb. The virus was identified in an individual who had recently travelled to Asia. A spokesperson for the UKHSA said at the time that this was “not unexpected” as both clades were in circulation.
The World Health Organization has reported that, as of 18 January 2026, four countries: Czechia, Israel, Madagascar and Nepal, as well as the French territory of Mayotte, have reported mpox cases caused by clade Ib, which is the more severe strain. It noted that clade Ib cases continue to be reported in France, Italy and Spain.
Who is most at risk of infection?
According to the UKHSA, there is a low risk to the general public. However, it advises people who have travelled to areas affected by clade I mpox to be aware of its symptoms and to contact a healthcare professional if they have any concerns.
How many cases are there in the UK?
The number of cases in the UK remains low, with around 250 cases being diagnosed in England in 2025 (see Figure 2). Cases of clade IIb are more common, while clade Ib cases are typically in the single figures each month.
Figure 2: Number of mpox cases in the UK
What treatments are available?
Mpox can get better without treatment after a few weeks, but treatment offered is mainly supportive (e.g. pain relief, keeping hydrated, etc.).
People who experience more severe symptoms or who are at risk of more severe disease, such as older people, young children and immunocompromised people, may need treatment in hospital, which can involve antivirals such as cidofovir and tecovirimat. There are no approved medications specifically for mpox.
Tecovirimat has not been studied in human efficacy trials for mpox. However, it has demonstrated efficacy against orthopoxviruses, including mpox, in animal models, while there are pharmacokinetic and safety data in humans2.
Tecovirimat inhibits the activity of the VP37 envelope protein, which is encoded by a highly conserved gene in all members of the orthopoxvirus genus — this stops the virus from leaving the infected cell, therefore, slowing spread of the infection.
Preventative measures are available through vaccination.
Who can get the mpox vaccine?
The mpox vaccine is available on the NHS to people who are at increased risk of contracting the virus. This includes men who have sex with men and also have multiple partners, engage in group sex or attend sex-on-premises venues. People who have had close contact with someone who has mpox, or who have a high risk of being exposed to the virus, are also eligible for the vaccine.
The mpox vaccine is being offered in sexual health clinics. Most people will be offered two doses of the vaccine, at least 28 days apart.
How effective is the mpox vaccine?
There is one vaccine available in the UK, Imvanex, which is known as a Modified Vaccinia Ankara–Bavarian Nordic (MVA-BN) vaccine.
Imvanex contains a replication defective virus and should be considered as an inactivated vaccine.
The results of a 2024 systematic review of mpox vaccines showed that one dose of MVA-BN had an efficacy of 76% from twelve studies, while two doses had an efficacy of 82%, as shown by six studies. Where the vaccine was given as post-exposure prophylaxis, its efficacy was 20%, as shown by seven studies3.
According to the NHS, anyone who contracts mpox after being vaccinated will typically have milder symptoms than someone who has not been vaccinated.
What is best practice for pharmacists in preventing mpox?
Pharmacists have an important role to play in ensuring that eligible people are informed about the mpox vaccine and its effectiveness.
They can also help to spot potential signs of mpox and ensure patients are on the right treatment pathway.
When should I suspect a potential case of mpox?
The UKHSA says healthcare professionals should consider mpox if an individual presents with any of the following:
1. A prodrome (i.e. fever, chills, headache, exhaustion, myalgia, arthralgia, backache, lymphadenopathy), in an individual with contact with a confirmed or suspected case of mpox in the 21 days before symptom onset; or
2. An mpox-compatible rash anywhere on the skin (e.g. face, limbs, extremities, torso), mucosae, including oral, genital, anal or symptoms of proctitis and at least one of the following in the 21 days before symptom onset:
- A recent new sexual partner;
- Contact with known or suspected case of mpox;
- A travel history to a country where mpox is currently common — this does not include people transiting through the affected country where they do not leave the airport;
- A link to an infected animal or meat; or
3. An mpox-compatible rash anywhere on the skin (e.g. face, limbs, extremities, torso), mucosae, including oral, genital, anal, or symptoms of proctitis, where there is no risk factor and no alternative common differential diagnosis1. These patients should be discussed with local infection services to determine the approach to investigation and management.
If community pharmacists suspect that a patient needs a clinical assessment to determine if they are a suspected case, NHS England recommends that they determine whether the patient is clinically stable or requires immediate intervention.
If stable, the patient should be advised to return home, isolate themselves and contact 111.
If the patient requires immediate intervention, the pharmacist should advise the patient to dial 999 if urgent, or contact a regional ambulance service to arrange transfer, clearly stating the patient is a suspected mpox case.
Further reading
- 1.Srivastava S, Kumar S, Jain S, et al. The Global Monkeypox (Mpox) Outbreak: A Comprehensive Review. Vaccines. 2023;11(6):1093. doi:10.3390/vaccines11061093
- 2.Grosenbach DW, Honeychurch K, Rose EA, et al. Oral Tecovirimat for the Treatment of Smallpox. N Engl J Med. 2018;379(1):44-53. doi:10.1056/nejmoa1705688
- 3.Pischel L, Martini BA, Yu N, et al. Vaccine effectiveness of 3rd generation mpox vaccines against mpox and disease severity: A systematic review and meta-analysis. Vaccine. 2024;42(25):126053. doi:10.1016/j.vaccine.2024.06.021
You may also be interested in
Norovirus and strategies for infection control

Case-based learning: insect bites and stings

