Blocking the immune system in rheumatoid arthritis
Download a PDF of the full infographic
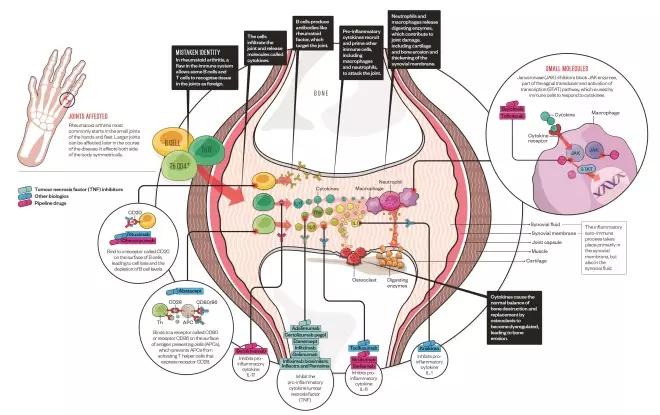
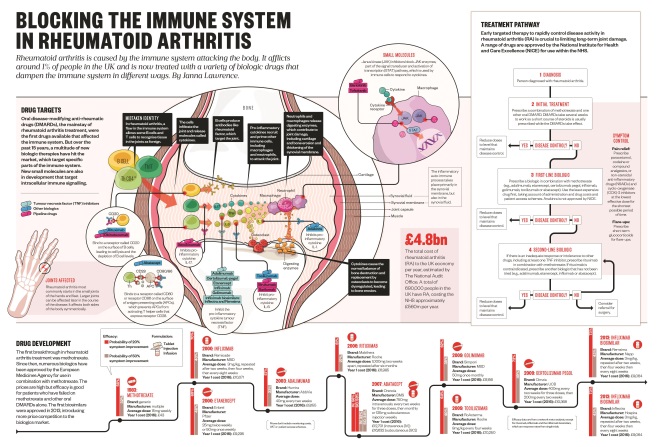
Drug targets in rheumatoid arthritis
Source: Summary of product characteristics and Nature
Oral disease-modifying anti-rheumatic drugs (DMARDs), the mainstay of rheumatoid arthritis treatment, were the first drugs available that affected the immune system. But over the past fifteen years, a multitude of new biologic therapies have hit the market, which target specific parts of the immune system. New small molecules are also in development that target intracellular immune signalling
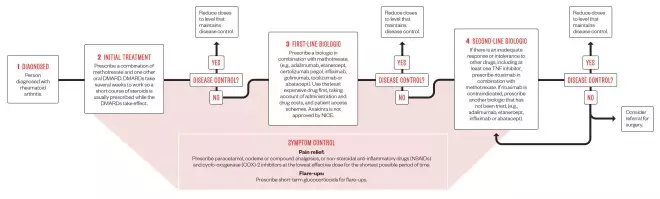
Treatment pathway for rheumatoid arthritis
Source: National Institute for Health and Care Excellence (NICE)
Early targeted therapy to rapidly control disease activity in rheumatoid arthritis (RA) is crucial to limiting long-term joint damage. A range of drugs are approved by the National Institute for Health and Care Excellence (NICE) for use within the NHS
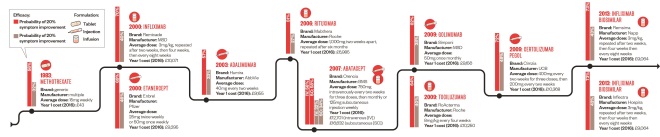
Timeline of drug development for rheumatoid arthritis
Source: NICE, British National Formulary, Arthritis Research and Therapy
The first big breakthrough in RA treatment was methotrexate. Since then, numerous biologics have been approved by the European Medicines Agency for use in combination with methotrexate. The prices are high but efficacy is good for patients who have failed on methotrexate and other oral DMARDs alone. The first biosimilars were approved in 2013, introducing more price competition to the biologics market.
- This article was updated on 1 June 2016 to clarify that rituximab is known as Mabthera and is manufactured by Roche.
References
Graphics: Alisdair Macdonald
Editorial advisers: Stewart Glaspole, NHS Brighton and Hove CCG; Robert Benson, Institute of Infection, Immunity and Inflammation, Glasgow University