Shutterstock.com
Tens of thousands of people with moderate rheumatoid arthritis (RA), whose disease is inadequately controlled on conventional therapies, are set to benefit from treatment with biologic therapies for the first time on the NHS.
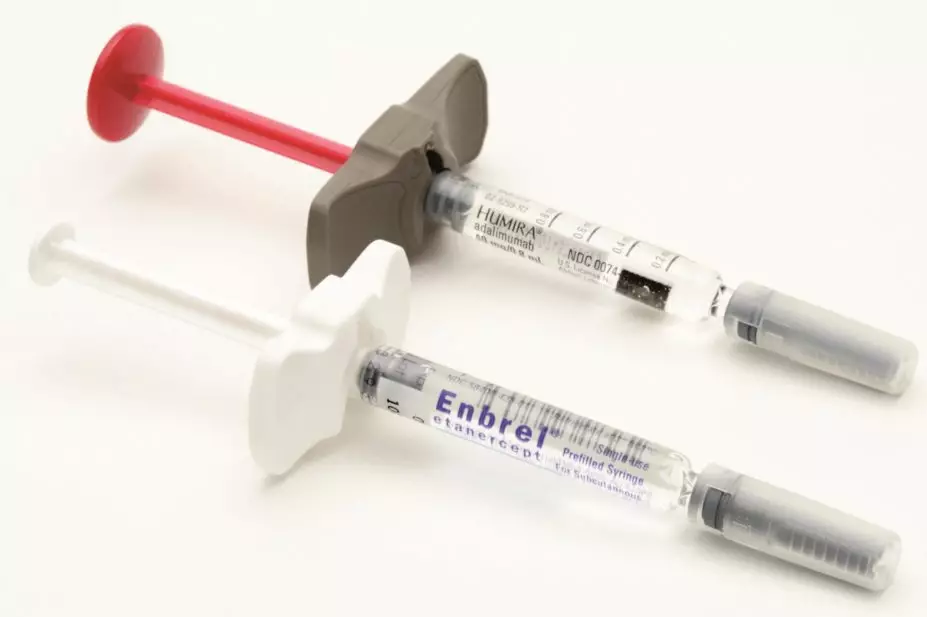
The National Institute for Health and Care Excellence (NICE) updated its guidance on 14 July 2021 to say that adalimumab, etanercept and infliximab, alongside methotrexate, are recommended if patients have a disease activity score (DAS28) of 3.2 to 5.1 after intensive therapy with two or more conventional disease-modifying antirheumatic drugs (DMARDs).
Adalimumab and etanercept can be used as monotherapy in the same patients if methotrexate is contraindicated or not tolerated.
Previously, NICE only recommended biological treatments for severe RA; however, the guidance was reviewed in the context of the increasing availability of biosimilars — medicines that are developed to be similar to an existing biological medicine — meaning that these treatments are now available to the NHS at a lower price.
The recommendations could benefit around 25,000 patients in the UK, according to NICE estimates, and follows the NICE approval of another advanced therapy, filgotinib (a targeted synthetic DMARD), in February 2022 for people with both severe and moderate rheumatoid arthritis.
Ailsa Bosworth, chief executive at the National Rheumatoid Arthritis Society (NRAS), said that the recommendations would mean that people with moderate disease no longer have to wait, sometimes for many years, for more advanced therapies.
“This decision is a real milestone in our efforts to improve equity of access to effective treatments for the many thousands of people who live with disease that is inadequately controlled, and therefore has a detrimental impact on the quality of their day-to-day lives,” she said.
Mark Samuels, chief executive of the British Biosimilars Association, said the “hugely significant” recommendation could address previously unmet medical needs and reduce health inequalities across the country.
“It is also an important milestone for the entire biosimilars industry as it sets a precedent for the use of such medicines for other conditions going forward,” he said.
What are biologics and biosimilars?
Biological medicines, or biologics, have revolutionised treatment for many patients living with chronic conditions, such as diabetes, autoimmune disease and cancer. Derived or manufactured from a living source, biologics work by targeting specific chemicals or cells involved in the body’s immune system response.
A biosimilar is a biologic that is highly similar to an existing, already approved biologic (the ‘originator’). Although not an exact copy, the biosimilar has no clinically meaningful differences to the originator and is approved according to the same standards of pharmaceutical quality, safety and efficacy that apply to all biologics.
READ MORE: Preparing for the big biologic switch