Shutterstock.com
After reading this article you should be able to:
- Understand the pathophysiology of cardio-renal metabolic disease (CRMD);
- Identify the risk factors for developing cardio-renal metabolic disease and use risk assessment tools, such as the ‘QRISK-3’ and ‘KFRE’ score, to evaluate the likelihood of future cardiovascular events and end-stage renal failure;
- Know how patients at risk of CRMD are managed and understand strategies for patient education and follow up.
Introduction
Cardiorenal metabolic disease (CRMD) refers to the interconnected dysfunctions of the cardiovascular, renal, and metabolic systems including chronic kidney disease (CKD), diabetes mellitus, heart failure (HF), hypertension and dyslipidaemia. These conditions often co-exist, creating a cycle where damage to one organ system exacerbates damage to others1. A combination of one or more of these conditions worsens prognosis and patient outcomes, including increased risk of cardiovascular events, hospitalisation and death1,2.
Statistics relating to the conditions associated with CRMD can be seen in Figure 13.
Figure 1: The overlap of conditions contributing to cardiorenal metabolic disease
The rising prevalence of CRMD imposes significant challenges on healthcare systems and affects the quality of life for affected individuals, including an increase in mortality, making it a growing concern for public health4. Cardiovascular disease has been recognised as the single biggest condition where improved management can lead to a reduction in the number of premature adult deaths and shorten gaps in health expectancy5. The early diagnosis and management of the overlapping conditions involved in CRMD are essential for improving quality of life and reducing morbidity and mortality, which is aligned with the governments priority of prevention rather than treatment and shifting care from hospitals to community5.
Despite advances in pharmacotherapy and management of CRMD, prognosis remains poor. Challenges in therapeutic management include:
- Limited awareness of the interlinking chronic conditions contributing to CRMD, both by healthcare professionals and patients;
- Inadequate patient screening, causing a delay to the identification of at-risk patients;
- Lack of timely interventions, leading to disease progression and poorer prognosis4,6.
Moreover, limited use of preventative medication has been seen in older patients. Often these patients are more likely to benefit from these medications because they reduce risk of hospitalisation with cardiovascular events7,8.
Pharmacists are integral to managing CRMD through medication management, patient education and supporting lifestyle changes. They are involved in the management of blood pressure, glucose and cholesterol-lowering therapies, HF optimisation and cardio-renal protection, ensuring optimal drug therapy and promoting adherence. Furthermore, pharmacists provide guidance and signposting to services offering lifestyle modifications, such as diet, weight loss and exercise, which are crucial in managing and preventing CRMD.
The benefits of medications to manage CRMD have recently been highlighted by landmark trials for the use of sodium-glucose co-transporter-2 inhibitors (SGLT2is), non-steroidal mineralocorticoid receptor antagonists (MRAs) and glucagon-like peptide-1 (GLP-1) mimetics, further expanding the available treatment options for CRMD9–13.
This article will outline the pathophysiology of CRMD, medication optimisation strategies to manage the associated conditions, as well as an overview of the role of the pharmacist in medication management and patient education.
Risk factors for cardiorenal metabolic disease
Table 1 shows the risk factors that can lead to the development of the conditions contributing to cardiorenal metabolic disease14–16. Often these risk factors are modifiable using lifestyle interventions, medication or a combination of both.
Tools to predict and monitor cardiorenal metabolic disease
The following tools can be used to assess each individual disease state contributing to CRMD. They can be used to diagnose, predict progression or help with ongoing monitoring. By combining these tools, early interventions can be made, with ongoing monitoring to ensure effective treatments are in place for each individual. This can aid healthcare professionals in reducing the burden of CRMD on the patient.
The use of these tools are particularly important for the expanding role of pharmacists within primary and secondary care. Within primary care, these tools can be used to identify patients at risk of CRMD at an early stage, ensuring that medication is initiated to mitigate disease progression. Primary care pharmacists are also able to use these tools to flag any patients who require expert secondary care interventions, referring to appropriate specialist teams in a proactive manner.
Secondary care specialist pharmacists are often embedded in multidisciplinary teams to provide specialist input to manage each aspect of CRMD. The use of these tools is integral to ensure treatment strategies are optimised to improve patient quality of life and long-term outcomes.
Cardiovascular disease prediction
The QRISK3 score is used for predicting the risk of developing CVD leading to a heart attack or stroke over the next ten years. This takes into consideration the patient’s age, smoking status, family history, cholesterol levels and comorbidities (e.g. diabetes, CKD)17. It can be used to determine the benefit of intervening with primary prevention strategies, such as lifestyle changes and lipid-lowering therapies18.
Kidney failure prediction
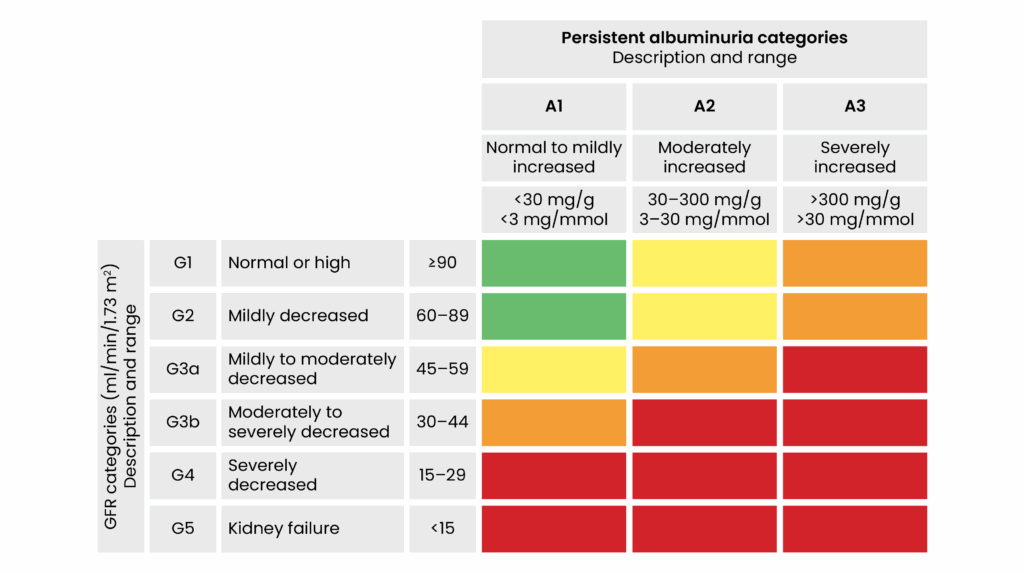
Estimated glomerular filtration rate (eGFR) and urine albumin:creatinine ratio (uACR): ‘Kidney disease: improving global outcomes’ (KDIGO) guidelines outline how the patients eGFR and uACR can determine what stage of CKD a patient is currently classified under, ranging from stages one to five. This also can be used as a predictor for CKD progression to end-stage renal failure and the patient’s risk of developing cardiovascular disease (see Figure 2)19.
Figure 2: KDIGO categorisation of chronic kidney disease (reproduced with permission)
Kidney failure risk equation (KFRE) predicts a patient’s two and five-year absolute risk of reaching end-stage kidney failure. This uses age, biological sex, eGFR and uACR within its calculation. In practice, this can be used to determine if patients require specialist nephrology review, prioritising those patients who require CKD care, including preventative interventions or preparing patients for renal replacement therapies20.
Diabetes diagnosis and monitoring
HbA1C is a reading of the average blood sugar levels from the previous two to three months, used to diagnose and monitor diabetes21.
Heart failure diagnosis and monitoring
N-terminal pro-B-type natriuretic peptide level (NT-Pro-BNP) is a serum test that can be taken to inform the diagnosis of HF and can aid with referral to HF specialists22.
Left ventricular ejection fraction is determined from a patient’s echocardiography test. Patients are categorised into ‘HF with preserved ejection fraction’ (HFpEF), ‘HF with mildly reduced ejection fraction’ (HFmrEF) or ‘HF with reduced ejection fraction’ (HFrEF)23. Diagnosis of HFpEF can be more challenging; therefore, scores such as H2FPEF and HFA-PEFF may be used by specialists to aid diagnosis24.
Symptomatic HF scores can be used to determine the symptomatic severity of HF. There are several scores available, such as the New York Heart Association Classification (NYHA) and the Kansas City Cardiomyopathy Questionnaire (KCCQ-12)24,25. These classify patients based on their limitations to physical activities and can be used to assess progression of disease, impact on quality of life and whether current treatments are optimal for symptom management.
Management of cardiorenal metabolic disease
Patients at risk of developing CRMD may present with one or more of the contributing diseases. To fully understand, prevent and manage CRMD as a whole, it is important to recognise the interplay between these different disease states. Rather than treating each disease independently, a holistic treatment and prevention strategy allows for individualised treatment plans to be made. The interaction of these conditions are outlined below along with considerations to optimise therapy to achieve the best patient outcomes.
Management of diabetes with chronic kidney disease
Diabetes is a significant risk factor for both CKD and CVD. Chronic hyperglycaemia leads to the promotion of inflammation, oxidative stress and tissue damage in both the renal and cardiovascular systems23,26. Diabetic nephropathy, a common cause of CKD, can accelerate kidney damage and impair glucose metabolism, worsening diabetic control23. Additionally, diabetes increases the risk of atherosclerotic plaques, which contribute to the development of ischemic heart disease and HF27.
Pharmacological management of diabetes in chronic kidney disease
The mainstay of treatment for renal protection from hyperglycaemic damage is through controlling blood glucose levels, whether that is with lifestyle interventions, antidiabetic agents or more effectively, a combination of the two. The NICE treatment pathway for diabetes management is dictated by patient risk factors, including chronic HF and/or established atherosclerotic CVD28. A diagnosis of CKD will automatically classify a patient with diabetes as high risk within the NICE algorithm28.
Metformin and SGLT2is should be used as first-line treatment for glycaemic control in CKD, but this will depend on the degree of kidney impairment28. Metformin is to be used with caution if eGFR is <30mL/min/1.73m2. The glycaemic lowering effect of SGLT2is is reduced as kidney function drops below 45mL/min/1.73m2 and is likely to be absent once eGFR is <30mL/min/1.73m2 29,30. Therefore, escalation to other glycaemic lowering agents is often required.
Within the current NICE pathway, GLP‑1 mimetic therapy is only recommended after the use of triple therapy when other antidiabetic drugs have been ineffective, with DPP-4 inhibitors, pioglitazone and sulfonylureas placed higher in the algorithm. International guidance is shifting towards the use of GLP‑1 mimetics earlier in the pathway and they are now recommended second line, if uncontrolled on dual therapy with metformin and SGLT2is. This recommendation is driven by the value of the additional long-term benefits that GLP‑1 mimetics provide, such as the cardiovascular benefits, renal protection and weight loss31–33.
Protecting the kidney in patients with diabetes
There are several mechanisms of CRMD treatments that help to protect the kidney from chronic hyperglycaemic damage independent of HbA1C management, as described below34.
Angiotensin converting enzyme inhibitors, angiotensin-II receptor blockers and sodium-glucose co-transporter-2 inhibitors
High blood glucose levels drive intra-glomerular hypertension, the consequence of which is hyperfiltration and subsequent kidney damage. The pathophysiology for this is multi-mechanistic and complex, although largely hypothesised to be linked with hyperglycaemia induced renal hypertrophy, altered afferent and efferent arteriole tone, and upregulation of proteins, such as SGLT233. Treatments such as angiotensin-converting enzyme inhibitors/angiotensin-II receptor blockers (ARBs) in combination with SGLT2is can help protect the kidney, irrespective of blood glucose levels through their effects on afferent and efferent arteriolar constriction/dilation to reduce hyperfiltration33.
Non-steroidal mineralocorticoid receptor antagonists
Finerenone is a non-steroidal MRA. Finerenone is more selective for the mineralocorticoid receptor and is associated with lower rates of hyperkalaemia, in comparison with other MRAs such as spironolactone and eplenerone34. It is currently only licensed for the treatment of CKD in those with type 2 diabetes mellitus. It can be used as an add-on therapy if albuminuria is still present (uACR >3mg/mmoL) despite maximum tolerated doses of ACEi/ARB and SGLT2i35. The reno-protective mechanism of finerenone is through antagonism of the mineralocorticoid receptor reducing pro-inflammatory and pro-fibrotic mediators36. The ongoing FIND-CKD study is assessing effectiveness in those patients without diabetes37.
Prevention of cardiovascular disease in chronic kidney disease
Primary prevention of CVD using statins is recommended in all patients with CKD (if eGFR <60mL/min/1.73m2) irrespective of diabetes status with a high-intensity statin18. The presence of albuminuria is indicative of kidney damage and systemic vascular disease, increasing the risk of a cardiovascular event in this patient population. Even in those patients without any major cardiovascular risk factors (e.g. no hypertension, diabetes or CKD), the presence of albumin in the urine serves as an independent cardiovascular risk factor, with each doubling of uACR increasing the risk of cardiovascular death by 36%38. This demonstrates the importance of statin use for cardio-protection, even in those with no perceived increased cardiovascular risk other than albuminuria.
Management of heart failure with chronic kidney disease
Impaired kidney function leads to the loss of the kidney’s ability to regulate fluid balance, blood pressure, electrolytes and waste products. As kidney function declines, fluid overload, increased vascular resistance and hypertension will contribute to an increased risk in CVD39. CKD also contributes to systemic inflammation and oxidative stress, promoting endothelial dysfunction and arterial stiffness, increasing the risk of atherosclerosis, myocardial infarction and HF39.
HF often develops resulting from longstanding hypertension, ischaemic heart disease and metabolic dysfunction. Hyperglycaemia and insulin resistance lead to myocardial stiffening, impaired contractility and adverse ventricular remodeling40. In HFpEF, systemic inflammation and metabolic stress lead to diastolic dysfunction. In HFrEF, myocardial fibrosis plays a dominant role. Additionally, endothelial dysfunction impairs nitric oxide-mediated vasodilation, leading to increased vascular stiffness and cardiac workload2.
HFrEF is managed using four primary classes of medications — also known as the “four pillars” of HF treatment (see Figure 3)9,11,21,37,41–44. These medications are aimed to reduce symptoms, prevent hospitalisations and improve survival, even in patients with CKD, low eGFRs or those on dialysis41. Careful monitoring and dose adjustments are necessary to optimise therapy and reduce risks. Robust evidence-based practice is often limited in this patient group and, with the absence of guidelines, close collaboration is needed between the HF and nephrology teams to create individualised treatment plans.
Figure 3: Four pillars of heart failure pharmacological management
Loop diuretics
In people with HF with low eGFR, adjuvant therapy with loop diuretics may be used at higher doses for effective fluid control owing to reduced renal function21. Patients on diuretics with low eGFRs require regular monitoring of electrolytes, renal function, blood pressure and volume status to prevent complications. Loop diuretics are ineffective in patients with anuria45.
Management of cardiovascular disease with diabetes
CVD is a leading cause of morbidity and mortality in individuals with diabetes7,46. The increased risk is multifactorial, with atherosclerosis, oxidative stress and diabetic cardiomyopathy being central contributors. To mitigate these risks, early comprehensive management is essential47. This includes optimising glycaemic control, managing hypertension and dyslipidaemia, and adopting lifestyle modifications, such as weight management, regular exercise and smoking cessation.
Diabetic cardiomyopathy is characterised by structural and functional changes in the heart muscle, independent of hypertension or coronary artery disease. Chronic hyperglycaemia and insulin resistance lead to overactivation of the renin-angiotensin-aldosterone system, causing cardiac dysfunction through myocardial fibrosis, left ventricular hypertrophy and impaired diastolic function. Over time, this can progress to HF48. The following strategies are vital for the management of CVD in people with diabetes.
Control hyperglycaemia
Persistent hyperglycaemia generates excess reactive oxygen species (ROS), leading to oxidative stress. Oxidative stress can damage blood vessels, contributing to the development of diabetic complications such as retinopathy (eye damage), nephropathy (kidney damage) and cardiovascular disease. Oxidative stress can trigger inflammation, which further exacerbates the damage caused by diabetes31. SGLT2is and GLP-1 mimetics have shown cardiovascular benefits and antioxidant effects. Current NICE guidance recommends the addition of an SGLT2i, alongside metformin, in patients with an increase cardiovascular risk27.
Lipid management
Diabetes accelerates the development of atherosclerosis, a condition characterised by the build-up of plaques in the arterial walls. Chronic hyperglycaemia leads to endothelial dysfunction, increased inflammation and abnormal lipid metabolism, all of which contribute to plaque formation31. Additionally, individuals with diabetes often have coexisting hypertension, obesity and dyslipidaemia, further increasing atherosclerotic risk. These factors increase the risk of coronary artery disease, myocardial infarction and stroke. Statins reduce LDL cholesterol and decrease the risk of cardiovascular events18.
Control blood pressure
Effective pharmacologic management of hypertension in individuals with type 1 and type 2 diabetes mellitus is crucial to mitigate the heightened risk of cardiovascular and renal complications associated with these conditions. NICE guidelines for hypertension recommend ACEis or ARBs as first-line treatments for people living with type 1 and type 2 diabetes mellitus49. Blood pressure targets are lower than those people who do not have diabetes, especially if there is albuminuria detected. Many patients with diabetes require multiple antihypertensive agents to achieve optimal blood pressure control. Combining medications from different classes can provide synergistic effects and minimise side effects49.
Management of chronic kidney disease
The primary aim for managing patients with CKD is to reduce the rate at which the kidney function declines, extending the life of the kidney to delay, or hopefully prevent altogether, the need for dialysis and/or kidney transplantation50.
Lifestyle interventions are essential for helping to manage kidney disease and prevent further renal damage. Smoking cessation, limiting alcohol intake, salt restriction, regular exercise, maintaining a healthy weight and eating a healthy balanced diet can all help to keep the kidneys as healthy as possible51. Diet management can often be the most complicated of these. Depending on the current degree of kidney dysfunction and individual patient factors, other dietary restrictions (such as potassium and phosphate) may become necessary — referral to specialist renal dietitians can help to navigate this51.
The pharmacological management for slowing the progression of kidney disease is moving towards a treatment strategy similar to that of the ‘pillar’ metaphor used for HF, utilising maximum tolerated doses of ACEi/ARBs, SGLT2is, non-steroidal MRAs and could soon include GLP-1 mimetics52.
Current practice is to monitor uACR as a predictor for the likelihood for disease progression (see Figure 2). A healthy kidney should not allow albumin into the urine and, therefore, if albuminuria is present at increased levels, it is a sign of glomerular injury and endothelial barrier dysfunction. The treatment strategies for this are centred around medications that can help protect the glomerulus from damage. Treatments should be sequentially added in, starting with ACEi/ARBs, uptitrated to the maximum tolerated dose with monitoring of albuminuria to decide on adding in further treatments50. The KFRE calculator is used to help predict individualised risk of kidney function decline. This helps to translate the risk to patients but also can be used to prioritise those patients who need specialist nephrology interventions20.
Once managing a healthy lifestyle and on optimum pharmacological therapy, ongoing treatment continues to focus on symptom management for the complications of kidney disease, such as mineral bone disease, anaemia of CKD, fluid and electrolyte disorders50.
Potassium management in patients with CRMD
Potassium management in patients with CRMD presents a significant clinical challenge owing to the complex interplay between heart, kidney and metabolic dysfunction. Hyperkalaemia in this population often arises from impaired renal potassium excretion, reduced insulin production and the use of medications such as ACE inhibitors, ARBs and MRAs19,53. Dietary strategies to modify potassium intake should only be considered when all non-dietary causes of hyperkalaemia have been excluded, such as constipation, acidosis and poorly controlled diabetes. If dietary modifications are required, inform patients to limit their intake of processed and ultra-processed foods before restricting plant-based foods53. Figure 4 shows potassium absorption rates from dietary sources19.
Figure 4: Potassium absorption rates from dietary sources (reproduced with permission)
However, when hyperkalaemia persists despite dietary measures and limits the ability to continue or optimise RAASi’s, further intervention is needed53.
Potassium binders should be considered in patients with CKD stage 3b to 5, or HF, who develop hyperkalaemia (defined as serum potassium >6.0 mmol/L) that restricts the use or optimal dosing of RAASi therapy. Agents, such as sodium zirconium cyclosilicate and patiromer bind to potassium in the gastrointestinal tract, effectively lowering serum potassium levels and enabling patients to maintain optimal RAASi treatment19,53. Further information about potassium management can be found in the UK Kidney Association’s clinical practice guidelines53.
Pharmacists’ role in managing cardiorenal metabolic disease
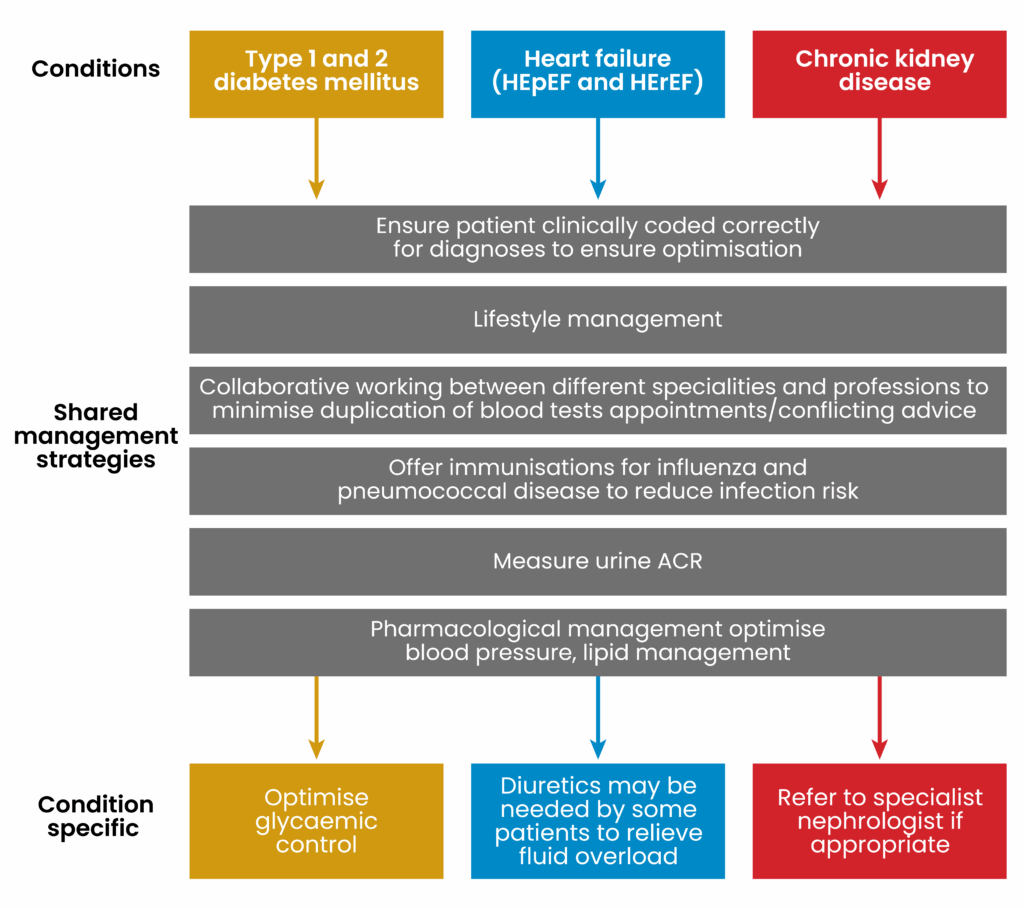
Early identification and management of CRMD improves patients’ quality of life and reduces risk of premature death. ACEi/ARBs, SGLT2is and statins are the mainstay of management of the three conditions that encompass CRMD (see Figure 5)4. These medications, along with lifestyle interventions, help prevent the progression of disease and therefore good patient education, and collaborative medication management efforts between the multidisciplinary teams in primary and secondary, can help to optimise therapy.
Figure 5: Overview of management in patients with cardiorenal metabolic disease
Lifestyle advice
Delivering these messages should be integrated into consultations and, where possible, include patient and family engagement to enable consistency:
- Avoiding the use of tobacco products — encourage patients to stop smoking, signpost or provide smoking cessation counselling and appropriate nicotine replacement therapy;
- Physical activity — patients should be encouraged to do some physical activity for a cumulative duration of at least 150 minutes per week, or to a level compatible with their cardiovascular, frailty and physical tolerance. Patients should be encouraged to avoid sedentary behaviour;
- Optimal weight – advise/encourage patients to lose weight where appropriate. Signpost to some free community support groups of initiatives that may be available within your borough;
- Diet – encourage patients to adopt healthy and diverse diet with a higher consumption of plant-based foods compared with animal-based foods and a lower consumption of processed and ultra processed foods22. Encourage hydration with water and reduction in the intake of sugary or sugar sweetened beverages, including fizzy drinks and limit alcohol. Restriction of food groups is not usually recommended but can be advised by specialist dieticians on a case-by-case basis24,54;
- Limit salt intake — recommended sodium chloride intake for adults is <5g (1 teaspoon) per day. Action on Salt, a government initiative, runs campaign and awareness weeks. Consider using posters to raise awareness at your place of work. Patients can also be signposted to the website for improved education18,19,55–58.
Sick day medication guidance
People living with CKD are more likely to develop an acute kidney injury. It is therefore important to counsel them on sick day rules to reduce risk of dehydration and AKI. If patients are unwell with vomiting, diarrhoea, fever, sweats and shaking, they should temporarily stop taking the medicines listed below. They should be encouraged to seek medical advice early if they are unsure what to do or continue to remain unwell after 48 hours. Patients can restart their medication two to three days after feeling well enough to eat and drink normally again59.
- ACE inhibitors;
- ARBs, including ARB/ARNI;
- Non-steroidal anti-inflammatory drugs;
- Diuretics;
- SGLT2is;
- Finerenone;
- Metformin;
- Sulfonylureas (e.g. gliclazide, glimepiride).
Best practice
- Initiate the patient on a statin (unless contraindicated) for primary prevention of CVD;
- Inform and educate the patient on their diagnosis and therapy if this has not previously been done:
- Most people do not have symptoms of kidney disease, especially in the early stages and so may not understand the importance of the health interventions that are being made;
- Spend time discussing healthy lifestyle choices with patients — this is essential in reducing cardiovascular risk factors. Check the patient’s health literacy levels and their understanding of the information they are presented with. Patient facing websites, such as Kidney Care UK, Diabetes UK and British Heart Foundation, provide information in a simplified format which a patient can be signposted to;
- Reinforce education on medication and the importance of medication adherence.
- Use tools such as KFRE, QRISK3 to recommend interventions and help patients make informed decisions;
- Consider using frameworks, such as ‘Make every contact count’, to deliver health messages and signpost patient to appropriate services;
- If a patient has diabetes, hypertension or CKD, NSAIDs should be avoided or used with caution, owing to the risk of deterioration of renal function or worsening hypertension;
- Age should not hinder the initiation of these preventative medications. Often the older patients are more likely to benefit from these medications because it reduces risk of hospitalisation with CV events. Use a patient’s frailty and their informed decision to guide management of CRMD. For more information on medicines use in frailty, see ‘Case-based learning: frailty’17–20,52,54,56,60.
Conclusion
Pharmacists as part of a multidisciplinary team can play an important role in the management of CRMD. Pharmacists’ strengths lie in the understanding of the disease, awareness of guidelines and their application, and understanding of key lifestyle factors and pharmacotherapy, all of which can improve outcomes for patients.
Furthermore, pharmacists are better versed in the nuances of drug licensing in the UK compared with other healthcare professionals. Within primary and secondary care, having access to several tools can enable patient screening, medicines optimisation and deprescribing where needed.
Being experts in medication counselling allows pharmacists to be able to identify and manage non-adherence to medications and provide good patient education about drug and disease alike to achieve the best outcomes for patients.
- 1.Marassi M, Fadini GP. The cardio-renal-metabolic connection: a review of the evidence. Cardiovasc Diabetol. 2023;22(1). doi:10.1186/s12933-023-01937-x
- 2.Rangaswami J, Bhalla V, Blair JEA, et al. Cardiorenal Syndrome: Classification, Pathophysiology, Diagnosis, and Treatment Strategies: A Scientific Statement From the American Heart Association. Circulation. 2019;139(16). doi:10.1161/cir.0000000000000664
- 3.Vijay K, Neuen BL, Lerma EV. Heart Failure in Patients with Diabetes and Chronic Kidney Disease: Challenges and Opportunities. Cardiorenal Med. 2021;12(1):1-10. doi:10.1159/000520909
- 4.Krentz A, Jacob S, Heiss C, et al. Rising to the challenge of cardio-renal-metabolic disease in the 21st century: Translating evidence into best clinical practice to prevent and manage atherosclerosis. Atherosclerosis. 2024;396:118528. doi:10.1016/j.atherosclerosis.2024.118528
- 5.Ten-year health plan for England: fit for the future. Department of Health and Social Care. 2025. Accessed November 2025. https://www.gov.uk/government/publications/10-year-health-plan-for-england-fit-for-the-future
- 6.Czupryniak L, Mosenzon O, Rychlík I, et al. Barriers to early diagnosis of chronic kidney disease and use of sodium‐glucose cotransporter‐2 inhibitors for renal protection: A comprehensive review and call to action. Diabetes Obesity Metabolism. 2024;26(10):4165-4177. doi:10.1111/dom.15789
- 7.Gregg EW, Gu Q, Cheng YJ, Venkat Narayan KM, Cowie CC. Mortality Trends in Men and Women with Diabetes, 1971 to 2000. Ann Intern Med. 2007;147(3):149-155. doi:10.7326/0003-4819-147-3-200708070-00167
- 8.Usman MS, Khan MS, Butler J. The Interplay Between Diabetes, Cardiovascular Disease, and Kidney Disease. Compendia. 2021;2021(1):13-18. doi:10.2337/db20211-13
- 9.Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N Engl J Med. 2015;373(22):2117-2128. doi:10.1056/nejmoa1504720
- 10.Neal B, Perkovic V, Mahaffey KW, et al. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med. 2017;377(7):644-657. doi:10.1056/nejmoa1611925
- 11.Heerspink HJL, Stefánsson BV, Correa-Rotter R, et al. Dapagliflozin in Patients with Chronic Kidney Disease. N Engl J Med. 2020;383(15):1436-1446. doi:10.1056/nejmoa2024816
- 12.Bakris GL, Agarwal R, Anker SD, et al. Effect of Finerenone on Chronic Kidney Disease Outcomes in Type 2 Diabetes. N Engl J Med. 2020;383(23):2219-2229. doi:10.1056/nejmoa2025845
- 13.Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes. N Engl J Med. 2016;375(19):1834-1844. doi:10.1056/nejmoa1607141
- 14.Ibrahim M, Ba‐Essa EM, Baker J, et al. Cardio‐renal‐metabolic disease in primary care setting. Diabetes Metabolism Res. 2023;40(3). doi:10.1002/dmrr.3755
- 15.Li J, Lei L, Wang W, et al. Social Risk Profile and Cardiovascular‐Kidney‐Metabolic Syndrome in US Adults. JAHA. 2024;13(16). doi:10.1161/jaha.124.034996
- 16.Dal Canto E, Farukh B, Faconti L. Why are there ethnic differences in cardio-metabolic risk factors and cardiovascular diseases? JRSM Cardiovascular Disease. 2018;7. doi:10.1177/2048004018818923
- 17.Hippisley-Cox J, Coupland C, Brindle P. Development and validation of QRISK3 risk prediction algorithms to estimate future risk of cardiovascular disease: prospective cohort study. BMJ. Published online May 23, 2017:j2099. doi:10.1136/bmj.j2099
- 18.Cardiovascular disease: risk assessment and reduction, including lipid modification. National Institute for Health and Care Excellence . Accessed November 2025. https://www.nice.org.uk/guidance/ng238
- 19.Stevens PE, Ahmed SB, Carrero JJ, et al. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney International. 2024;105(4):S117-S314. doi:10.1016/j.kint.2023.10.018
- 20.Tangri N, Grams ME, Levey AS, et al. Multinational Assessment of Accuracy of Equations for Predicting Risk of Kidney Failure. JAMA. 2016;315(2):164. doi:10.1001/jama.2015.18202
- 21.Chronic heart failure in adults: diagnosis and management. National Institute for Health and Care Excellence . Accessed November 2025. https://www.nice.org.uk/guidance/ng106
- 22.McDonagh TA, Metra M, Adamo M, et al. 2021 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure. European Heart Journal. 2021;42(36):3599-3726. doi:10.1093/eurheartj/ehab368
- 23.Low Wang CC, Hess CN, Hiatt WR, Goldfine AB. Clinical Update: Cardiovascular Disease in Diabetes Mellitus. Circulation. 2016;133(24):2459-2502. doi:10.1161/circulationaha.116.022194
- 24.Soto GE, Jones P, Weintraub WS, Krumholz HM, Spertus JA. Prognostic Value of Health Status in Patients With Heart Failure After Acute Myocardial Infarction. Circulation. 2004;110(5):546-551. doi:10.1161/01.cir.0000136991.85540.a9
- 25.Rask-Madsen C, King GL. Vascular Complications of Diabetes: Mechanisms of Injury and Protective Factors. Cell Metabolism. 2013;17(1):20-33. doi:10.1016/j.cmet.2012.11.012
- 26.Kumar M, Dev S, Khalid MU, et al. The Bidirectional Link Between Diabetes and Kidney Disease: Mechanisms and Management. Cureus. Published online September 20, 2023. doi:10.7759/cureus.45615
- 27.Type 2 diabetes in adults: management. National Institute for Health and Care Excellence. Accessed November 2025. https://www.nice.org.uk/guidance/ng28
- 28.Jardiance 10 mg film-coated tablets: summary of product characteristics. Electronic Medicines Compendium (eMC). Accessed November 2025. https://www.medicines.org.uk/emc/product/5441/smpc#gref
- 29.Forxia 10 mg film-coated tablets: summary of product characteristics. Electronic Medicines Compendium . 2024. Accessed November 2025. https://www.medicines.org.uk/emc/product/7607/smpc#gref
- 30.Rossing P, Caramori ML, Chan JCN, et al. KDIGO 2022 Clinical Practice Guideline for Diabetes Management in Chronic Kidney Disease. Kidney International. 2022;102(5):S1-S127. doi:10.1016/j.kint.2022.06.008
- 31.Marx N, Federici M, Schütt K, et al. 2023 ESC Guidelines for the management of cardiovascular disease in patients with diabetes. European Heart Journal. 2023;44(39):4043-4140. doi:10.1093/eurheartj/ehad192
- 32.ElSayed NA, McCoy RG, et al. 9. Pharmacologic Approaches to Glycemic Treatment: Standards of Care in Diabetes—2025. Diabetes Care. 2024;48(Supplement_1):S181-S206. doi:10.2337/dc25-s009
- 33.Tonneijck L, Muskiet MHA, Smits MM, et al. Glomerular Hyperfiltration in Diabetes: Mechanisms, Clinical Significance, and Treatment. JASN. 2017;28(4):1023-1039. doi:10.1681/asn.2016060666
- 34.Pitt B, Filippatos G, Agarwal R, et al. Cardiovascular Events with Finerenone in Kidney Disease and Type 2 Diabetes. N Engl J Med. 2021;385(24):2252-2263. doi:10.1056/nejmoa2110956
- 35.Finerenone for treating chronic kidney disease in type 2 diabetes. National Institute for Health and Care Excellence . 2023. Accessed November 2025. https://www.nice.org.uk/guidance/ta877
- 36.10 mg film-coated tablets: summary of product characteristics. Electronic Medicines Compendium . 2023. Accessed November 2025. https://www.medicines.org.uk/emc/product/13437/smpc/
- 37.Heerspink HJL, Agarwal R, Bakris GL, et al. Design and baseline characteristics of the Finerenone, in addition to standard of care, on the progression of kidney disease in patients with Non-Diabetic Chronic Kidney Disease (FIND-CKD) randomized trial. Nephrology Dialysis Transplantation. 2024;40(2):308-319. doi:10.1093/ndt/gfae132
- 38.Claudel SE, Waikar SS, Schmidt IM, Vasan RS, Verma A. The relationship between low levels of albuminuria and mortality among adults without major cardiovascular risk factors. European Journal of Preventive Cardiology. 2024;31(17):2046-2055. doi:10.1093/eurjpc/zwae189
- 39.Jankowski J, Floege J, Fliser D, Böhm M, Marx N. Cardiovascular Disease in Chronic Kidney Disease. Circulation. 2021;143(11):1157-1172. doi:10.1161/circulationaha.120.050686
- 40.Borghetti G, von Lewinski D, Eaton DM, Sourij H, Houser SR, Wallner M. Diabetic Cardiomyopathy: Current and Future Therapies. Beyond Glycemic Control. Front Physiol. 2018;9. doi:10.3389/fphys.2018.01514
- 41.Chronic heart failure in adults quality standard. National Institute for Health and Care Excellence . 2025. Accessed November 2025. https://www.nice.org.uk/guidance/qs9
- 42.Entresto 24 mg/26 mg, 49 mg/51 mg and 97 mg/103 mg film-coated tablets: summary of product characteristics. Electronic Medicines Compendium . Accessed November 2025. https://www.medicines.ie/medicines/entresto-24mg-26mg-49mg-51mg-and-97mg-103mg-film-coated-tablets-32015/spc
- 43.Ando K, Ohtsu H, et al. Rationale and design of the Eplerenone combination Versus conventional Agents to Lower blood pressure on Urinary Antialbuminuric Treatment Effect (EVALUATE) trial: a double-blinded randomized placebo-controlled trial to evaluate the antialbuminuric effects of an aldosterone blocker in hypertensive patients with albuminuria. Hypertens Res. 2010;33(6):616-621. doi:10.1038/hr.2010.46
- 44.Siddiqui R, Obi Y, Dossabhoy NR, Shafi T. Is There a Role for SGLT2 Inhibitors in Patients with End-Stage Kidney Disease? Curr Hypertens Rep. 2024;26(12):463-474. doi:10.1007/s11906-024-01314-3
- 45.Furosemide 10 mg/ml solution for injection/infusion: summary of product characteristics. Electronic Medicines Compendium . Accessed November 2025. https://www.medicines.org.uk/emc/product/13171/smpc
- 46.Official statistics: Diabetes profile. Department of Health and Social Care. 2021. Accessed November 2025. https://www.gov.uk/government/statistics/diabetes-profile-update-march-2025
- 47.Faria A, Persaud SJ. Cardiac oxidative stress in diabetes: Mechanisms and therapeutic potential. Pharmacology & Therapeutics. 2017;172:50-62. doi:10.1016/j.pharmthera.2016.11.013
- 48.Bellemare M, Bourcier L, Iglesies‐Grau J, Boulet J, O’Meara E, Bouabdallaoui N. Mechanisms of diabetic cardiomyopathy: Focus on inflammation. Diabetes Obesity Metabolism. 2025;27(5):2326-2338. doi:10.1111/dom.16242
- 49.Hypertension in adults: diagnosis and management. National Institute for Health and Care Excellence. 2023. Accessed November 2025. https://www.nice.org.uk/guidance/ng136
- 50.Chronic kidney disease: assessment and management. National Institute for Health and Care Excellence. 2021. Accessed November 2025. https://www.nice.org.uk/guidance/ng203
- 51.Chronic kidney disease: Prevention. NHS. 2023. Accessed November 2025. https://www.nhs.uk/conditions/kidney-disease/prevention/
- 52.Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes. N Engl J Med. 2024;391(2):109-121. doi:10.1056/nejmoa2403347
- 53.UKKA clinical practice guideline – Management of Hyperkalaemia in Adults. UK Kidney Association. 2023. Accessed November 2025. https://www.ukkidney.org/sites/renal.org/files/FINAL%20VERSION%20-%20UKKA%20CLINICAL%20PRACTICE%20GUIDELINE%20-%20MANAGEMENT%20OF%20HYPERKALAEMIA%20IN%20ADULTS%20-%20191223_0.pdf
- 54.Kidney Care UK. Kidney Care UK. Accessed November 2025. https://www.kidneycareuk.org/
- 55.Information and support. British Heart Foundation. Accessed November 2025. https://www.bhf.org.uk/informationsupport/
- 56.Simple signposting to better health and wellbeing. MECC Link. Accessed November 2025. https://www.mecclink.co.uk/
- 57.Lifestyle Changes to Prevent a Heart Attack. American Heart Association. Accessed November 2025. https://www.heart.org/en/health-topics/heart-attack/life-after-a-heart-attack/lifestyle-changes-for-heart-attack-prevention
- 58.Action on Salt. Queen Mary University of London. Accessed November 2025. https://www.actiononsalt.org.uk/
- 59.“Sick day” guidance in patients at risk of acute kidney injury: position statement. Think Kidneys Programme Board. 2018. Accessed November 2025. https://www.thinkkidneys.nhs.uk/aki/wp-content/uploads/sites/2/2018/01/Think-Kidneys-Sick-Day-Guidance-2018.pdf
- 60.Diabetes UK. Diabetes UK. Accessed November 2025. https://www.diabetes.org.uk/