
Wes Mountain
This case example explores the complexity of prescribing high-cost drugs in the context of patient-centred care.
A list of the skills required to manage such situations — mapped to the Royal Pharmaceutical Society’s ‘Competency framework for all prescribers’ — can be found at the end of this article.

Case presentation
A 35-year-old female patient with a five-year history of ulcerative colitis (UC) presented to a specialist inflammatory bowel disease (IBD) clinic with worsening symptoms over the previous eight weeks, including up to eight loose stools per day, intermittent rectal bleeding, abdominal cramping and fatigue.
Despite optimal use of mesalazine (oral and rectal formulations) and a recent short course of oral corticosteroids, her symptoms persisted. A recent flexible sigmoidoscopy confirmed moderate-to-severe disease activity, while her faecal calprotectin was significantly above the reference range of 100–250 μg/g. Her inflammatory markers were also elevated, with a CRP 27mg/L greater than reference range of 0–5mg/L. She had no previous exposure to biologic therapies, was up to date with vaccinations and had no contraindications to immunosuppressive treatment.

The dilemma
The patient was referred to the pharmacist prescriber and gastroenterology consultant to discuss escalation of therapy, where she expressed significant concern over work absences and the impact on her role as a full-time teacher. According to National Institute for Health and Care Excellence (NICE) guidelines and British Society of Gastroenterology (BSG) guidance, patients with moderate-to-severe UC who fail to respond to aminosalicylates and steroids should be considered for advanced therapies, such as biologics (e.g. infliximab, adalimumab, vedolizumab) or small-molecule inhibitors (e.g. tofacitinib)1,2.
The patient met the clinical criteria for advanced therapy; however, the first-line options in the local commissioning formulary were limited to biosimilar infliximab and adalimumab, which was based on cost-effectiveness considerations under the local high-cost drugs commissioning policy.
While these tumour necrosis factor (TNF) inhibitors are effective, the patient expressed concern over the need for intravenous infusions, owing to the risk of serious infections or systemic adverse effects (i.e. infliximab), and reported significant needle phobia, raising potential adherence issues with subcutaneous therapies (i.e. adalimumab). Oral therapies were also considered; however, the patient declared that she has issues with adherence to oral agents and did not want to take medication every day. After discussing all the options, the patient showed a strong preference for vedolizumab, which has a more gut-specific mechanism and a potentially more favourable side-effect profile3–5.
However, according to the locally agreed pathway, vedolizumab is only approved for patients who have failed or are intolerant to anti-TNF agents. Although national guidance indicates similar clinical efficacy, anti-TNF therapies remain the first-line option owing to their greater cost-effectiveness6. This created a dilemma: should the prescriber initiate an anti-TNF therapy per formulary guidance and locally agreed pathways, knowing the patient may struggle with adherence and administration, or support an application for vedolizumab based on individual patient factors?

Consultation
During the consultation, the mechanisms of action, routes of administration, potential side effects and monitoring requirements for anti-TNF agents versus vedolizumab were discussed.
The patient explained her needle phobia and the psychological impact it had on her past healthcare experiences. She also raised practical concerns about infusion centre access and time off work for treatment. Importantly, she expressed a willingness to attend infusions if they were less frequent, as with vedolizumab’s longer maintenance interval of every eight weeks, according to NICE guidance3.
A shared decision-making approach was used, and the patient was encouraged to ask questions and express concerns. Although she acknowledged that infliximab may be a faster-acting agent in some patients, her personal priorities were minimising psychological stress, ensuring treatment adherence and limiting disruption to her career. She was also concerned about infection risk with systemic immunosuppressants and was reassured by the gut-selective action of vedolizumab3–5.
The pharmacist prescriber consulted the IBD multidisciplinary team (MDT) and the local formulary advisor to discuss an individual funding request (IFR). The IFR process ensures that patients with exceptional clinical circumstances can access treatments not routinely commissioned.
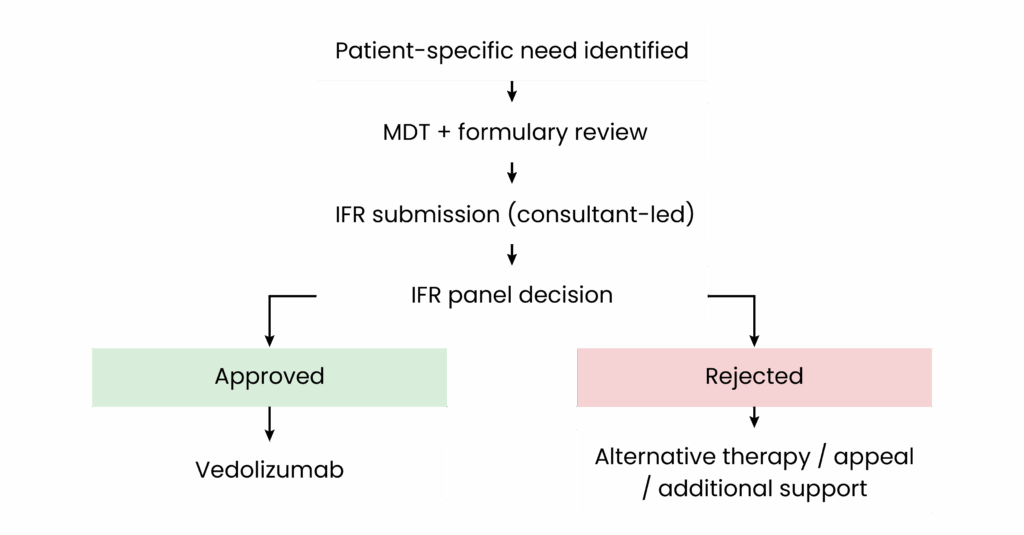
The typical IFR workflow can be seen in Box 17.
Box 1: Individual funding request workflow
- Identification of clinical exceptionality: The prescribing clinician or pharmacist identifies that standard commissioned therapies are unsuitable owing to patient-specific factors (e.g. intolerance, psychological burden, adherence issues);
- Multidisciplinary discussion: The case is reviewed with the inflammatory bowel disease multidisciplinary team (MDT) and local formulary advisors to confirm appropriateness and gather supporting clinical evidence;
- Preparation and submission: The responsible consultant or prescribing clinician submits the individual funding request (IFR) to the local clinical commissioning group/NHS England, including:
- Clinical history and exceptionality;
- Evidence of patient preference and quality-of-life considerations;
- Supporting literature (e.g. National Institute of Health and Care Excellence guidance, trial evidence).
- Review and decision-making: The IFR panel evaluates the request based on:
- Clinical exceptionality;
- Alignment with national guidance;
- Cost implications and NHS resource stewardship;
- Outcome and next steps: If approved, the patient receives the requested treatment. If rejected:
- The MDT may consider alternative routinely commissioned therapies;
- The decision can be appealed with additional clinical justification;
- Additional support strategies (e.g. adherence, psychological support) may be implemented to optimise standard therapies.
The Pharmaceutical Journal

Appropriate monitoring and additional actions agreed
Baseline investigations, including TB screening, hepatitis B/C serology and varicella status, were completed.
In parallel, the patient was referred to the IBD nurse specialist for biologic counselling, while psychological support services were signposted to help manage treatment-related anxiety.
Sick-day rules, infection risk precautions and vaccine advice, including influenza and pneumococcal immunisation, were discussed. The patient was advised on signs of infection, infusion reactions and when to seek medical advice.

Follow-up
Within two weeks, the IFR was approved. Vedolizumab was initiated with support from the hospital day case team. The patient tolerated induction infusions well, reported significant symptom improvement after the second dose, and returned to full-time work without further absences.
Her calprotectin reduced to <150μg/g, and she reported only 2–3 formed stools per day without blood. She also experienced no infusion reactions or infections. The patient reported improved quality of life and satisfaction with her care plan.
If the IFR had not been approved, the MDT would have needed to consider alternative therapies that are routinely commissioned, such as anti-TNF agents, alongside additional support for adherence and psychological wellbeing. Further options could include appealing the decision with additional clinical justification or exploring trial enrolment if appropriate.
Reflective practice
This case illustrates the complexity of prescribing high-cost drugs in the context of patient-centred care, commissioning frameworks and clinical efficacy. It raised an important question: how do we balance guideline adherence, formulary restrictions and patient-centred care when prescribing high-cost biologics?
The decision to pursue vedolizumab — despite it being more expensive than anti-TNF therapy — reflected prioritisation of the patient’s individual needs, minimising psychological stress, supporting adherence and allowing continued employment alongside evidence of comparable efficacy. While infliximab or adalimumab may have been appropriate from a cost and guideline perspective, the patient’s preferences and lifestyle circumstances justified a deviation from the default pathway. IFRs exist to enable clinicians to advocate for treatments that are clinically appropriate in exceptional circumstances, even when not routinely commissioned.
By collaborating with the MDT and advocating for the patient, we ensured she received a treatment she could engage with long term, reducing the risk of non-adherence, disease flares and hospitalisation. Reflectively, this highlights the importance of balancing policy with personalised care. While cost-effective prescribing is essential, patient engagement, treatment sustainability and functional outcomes are equally critical in real-world decision-making.

RPS Competency Framework for All Prescribers
This article is aimed at supporting the development of knowledge and skills related to the following competencies:
Domain 1.5: Building rapport and understanding the patient’s lived experience with inflammatory bowel disease;
Domain 1.7: Conducting appropriate clinical assessment and understanding disease severity;
Domain 2.3 and 2.4: Evaluating the risks and benefits of different biologics in the context of adherence and patient-specific factors;
Domain 2.7 and 2.8: Using evidence-based guidance alongside local policies to support individualised care;
Domain 3.1 and 3.3: Involving the patient in shared decision-making and clearly communicating therapeutic options;
Domain 4.1 and 4.2: Prescribing high-cost biologics with full awareness of mechanisms, risks and required monitoring;
Domain 4.3: Navigating and working within local prescribing frameworks and funding mechanisms;
Domain 5.4 and 5.5: Supporting patient self-management and providing contingency advice (e.g. infection signs);
Domain 6.1 and 6.2: Establishing a clear monitoring and review plan with multidisciplinary input.
Disclaimer
The information in this dilemma draws on the prescriber’s own experience in practice but the patient information has been changed to protect anonymity. The author aims to support others to navigate ‘grey areas’ within active prescribing by stimulating discussion through the sharing of their clinical approach.
The content contained in this dilemma is for educational purposes only and does not constitute clinical advice, guidance or recommendation. Other clinical approaches may be more appropriate for similar patients based on a full exploration of shared decision-making and person-centred care.
- 1.Ulcerative colitis: management. The National Institute for Health and Care Excellence . 2019. https://www.nice.org.uk/guidance/ng130
- 2.Lamb CA, Kennedy NA, Raine T, et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68(Suppl 3):s1-s106. doi:10.1136/gutjnl-2019-318484
- 3.Vedolizumab for treating moderately to severely active Crohn’s disease after prior therapy . The National Institute for Health and Care Excellence . 2015. https://www.nice.org.uk/guidance/ta352](https://www.nice.org.uk/guidance/ta352)
- 4.Feagan B. Safety and Positioning of Vedolizumab in Patients With Inflammatory Bowel Disease. National Library of Medicine . 2018. https://pmc.ncbi.nlm.nih.gov/articles/PMC6009184/
- 5.Solitano V, Facciorusso A, Jess T, et al. Comparative Risk of Serious Infections With Biologic Agents and Oral Small Molecules in Inflammatory Bowel Diseases: A Systematic Review and Meta-Analysis. Clinical Gastroenterology and Hepatology. 2023;21(4):907-921.e2. doi:10.1016/j.cgh.2022.07.032
- 6.Commissioning framework for biological medicines. NHS England . 2025. https://www.england.nhs.uk/publication/commissioning-framework-for-biological-medicines/
- 7.Individual Funding Requests (IFR) Policy. NHS England . 2023. https://www.england.nhs.uk/publication/individual-funding-request-ifr-policy/
5 comments
You must be logged in to post a comment.





Thank you for this article. I agree that the IFR process is a powerful tool for patient's who have presentations which are so unusual and distinct from their peers that the commissioning policy would be unable to realistically foresee their management. It is a tool that, as clinicians, we should be aware of as to best support the management of our patients.
My main queries with this are three fold:
1. I sympathise with this patient and their significant needle phobia. However, (significant) needle phobia is not at all an exceptional trait to have. A cursory glance suggests that at least 10% of the population may be needle phobic. I would argue that 1 in 10 patient's would not reach the bar for clinical exceptionality, how do you propose to treat the following patients who come into your care who are similarly needle phobic?
2. Vedolizumab is also an injection. Indeed, initiation of infliximab is very similar to that of vedolizumab in UC. Both medications also have subcutaneous preparations. If needle phobia was the prime "exceptionality" why pick another injectable therapy? Especially when we take into account the existence of multiple options for oral escalation (JAK inhibitors)? I am unsure why this particular patient is able to be injected with vedolizumab but Infliximab is a step too far.
3. I note that you have referenced the NHS England IFR policy. Assuming this is an adult gastroenterology service, and therefore an adult gastroenterology pathway, this would fall under the commissioning responsibility of your local ICB. Could you kindly clarify which organisation processed the IFR case and reference accordingly?
Thank you for your time.
Ultimately, my query is why vedolizumab when from a patient factors POV it suffers from all the same problems associated with the first line options in the commissioning policy.
Thank you very much for your thoughtful and detailed comments. I really appreciate the engagement and the opportunity to clarify the context and rationale behind this case. The intention was never to be prescriptive, but rather to stimulate constructive discussion around how prescribing frameworks can be applied in real-world clinical practice.
1. Needle phobia and clinical exceptionality
I agree entirely that needle phobia is not uncommon, and on its own would not usually meet the threshold for clinical exceptionality. In this case, however, the funding request was not based solely on the presence of needle phobia. What felt important was the severity of the phobia, the clear psychological impact it had had on the patient’s previous healthcare experiences, and how this interacted with other practical and clinical factors. These included difficulties attending infusion centres, the need to take repeated time off work, and a documented history of adherence issues with oral therapies. The assessment of exceptionality was therefore very much patient-specific and multifactorial, rather than an attempt to suggest that needle phobia in itself is exceptional.
2. Choice of vedolizumab in the context of needle phobia
You are absolutely right that vedolizumab is also an injectable therapy, and that both infliximab and vedolizumab have subcutaneous formulations. In this case, the choice of vedolizumab was less about avoiding injections altogether and more about treatment delivery and acceptability. The patient was clear that she did not want to self-inject, but she was willing to attend for infusions provided they were less frequent. Vedolizumab’s eight-weekly maintenance schedule, in line with NICE guidance, felt more manageable for her than alternative options.
Oral escalation strategies, including JAK inhibitors, were considered. However, the patient had previously struggled with compliance to tablet-based treatments. Alongside this, vedolizumab’s gut-selective mechanism and established safety profile in ulcerative colitis were felt to be clinically appropriate. Ultimately, this was a shared decision-making process that balanced efficacy, safety, adherence, and patient preference, rather than placing needle phobia as the sole or overriding factor.
3. IFR policy and commissioning responsibility
Thank you for highlighting the commissioning distinction. As this was an adult gastroenterology pathway, the individual funding request was processed locally via the relevant Integrated Care Board (ICB), rather than directly by NHS England. The reference to the NHS England IFR policy was included to reflect some of the overarching principles that often inform local IFR processes, rather than to imply that NHS England was the responsible commissioner in this case. As you note, local commissioning arrangements continue to evolve and can vary between regions in response to population and demographic needs.
In summary, the purpose of the case was to highlight the complexities that can arise when applying established prescribing pathways to individual patients, and to support discussion around how flexibility can be exercised appropriately within those frameworks. Thank you again for your comment. it’s genuinely helpful to have this level of engagement and to encourage productive discussion.
I read with interest this article which thoughtfully illustrates the tension between patient-centred care and local commissioning constraints when prescribing high-cost biologics.
The case highlights excellent shared decision-making, multidisciplinary working and robust clinical governance. However, I would caution against framing this scenario as one that should routinely be resolved through the individual funding request (IFR) route.
IFRs are intended to support access to treatment in genuinely exceptional circumstances, where a patient’s clinical needs are materially different from those of the wider cohort for whom a commissioned pathway has been developed. In this case, the patient clearly meets NICE criteria for advanced therapy escalation and has no contraindications to treatment. Her concerns, needle phobia, impact on work, preference for less frequent administration and desire to minimise systemic immunosuppression are all valid and important, but they are also increasingly common among patients with ulcerative colitis.
As such, this patient arguably belongs to a clearly identifiable cohort rather than representing true exceptionality. Many working-age adults with inflammatory bowel disease experience similar psychological, occupational and adherence-related barriers to anti-TNF therapy. If such factors consistently require IFR approval to access gut-selective or alternative first-line biologics, this suggests a misalignment between commissioning pathways and real-world patient needs, rather than an appropriate use of exceptionality mechanisms.
While IFR approval in this case led to an excellent clinical outcome, reliance on IFRs risks creating inequity, administrative burden and unwarranted variation in care. From a system perspective, repeated IFR submissions for similar patients should prompt reconsideration of formulary positioning and pathway flexibility, particularly where national guidance recognises comparable efficacy across biologic classes and where patient-centred outcomes influence adherence and long-term effectiveness.
Thank you for such a thoughtful and well-considered response. I agree with much of what you’ve said, particularly the concern that IFRs risk being used to manage what is increasingly a recognisable group of patients, rather than genuinely exceptional cases. If we start to see these situations coming up regularly, then I agree the issue is less about individual exceptionality and more about whether local pathways and agreements are keeping pace with the needs of the population we’re actually treating. Psychological factors, work pressures, treatment acceptability and adherence challenges are now very common in working-age adults with inflammatory bowel disease, and commissioning frameworks need to reflect that reality.
At present, not all regions have local agreements that capture these cohorts, and many services still rely heavily on NICE guidance to steer commissioning decisions. In this particular case, the IFR route was therefore the only realistic mechanism available to progress care in a way that aligned with both clinical judgement and the patient’s preferences. It was very much a pragmatic response to a local gap at that point in time, rather than a suggestion that IFRs should be the default solution.
I would fully agree that repeated IFRs for similar scenarios should act as a clear signal to commissioners that a more consistent and equitable approach is needed, whether through greater pathway flexibility, revised formulary positioning, or locally agreed criteria that better reflect real-world practice. It is encouraging that many commissioning teams are already evolving their processes to accommodate the breadth of clinical and psychosocial factors we now routinely encounter, and I expect this will continue as services adapt to changing population needs.
Thank you again for such a balanced and system-aware contribution. These kinds of discussions are exactly what help move pathways forward in a constructive way.