STEVE GSCHMEISSNER / SCIENCE PHOTO LIBRARY
After reading this learning article, you should be able to:
- Differentiate between hyperthyroidism and thyrotoxicosis and understand their causes;
- Recognise the symptoms and understand how the conditions are diagnosed;
- Know the different treatment options, including how complications are managed.
The thyroid is a small butterfly-shaped gland found at the front base of the neck[1]. The primary function of the thyroid is the synthesis and release of two main thyroid hormones that work to regulate metabolic rate, triiodothyronine (T3) and tetraiodothyronine (T4) — T4 is more commonly known as thyroxine[1,2]. Dietary iodine is essential for thyroid hormone production. T3 and T4 synthesis and regulation is summarised in ‘Hypothyroidism: causes, diagnosis and treatment‘.
The terms ‘hyperthyroidism’ and ‘thyrotoxicosis’ are often used interchangeably; however, hyperthyroidism refers specifically to excess production and secretion of T3 and T4 from the thyroid gland, disproportionate to requirement, while thyrotoxicosis is the clinical manifestation of symptoms owing to excess T3 and T4 action in peripheral tissues[2,3].
Similarly to hypothyroidism, hyperthyroidism is ten times more prevalent in females than in males and is most likely to present between the ages of 30–50 years, with UK prevalence reported as 0.5%–2.0% and 0.2%, respectively[4]. In contrast to hypothyroidism, incidence of hyperthyroidism does not increase with advancing age[5].
Causes
Thyrotoxicosis can be classified according to its aetiology and origin of dysfunction, which can be either within the thyroid or from other tissues. Hyperthyroidism is specifically characterised by increased synthesis and secretion of thyroid hormones from the thyroid gland[2]. Graves’ disease is the most common cause of hyperthyroidism in the UK, accounting for around 70–80% of cases[2,6]. Graves’ disease arises from the autoimmune-mediated abnormal production of immunoglobulin G (IgG) that occupies and activates the thyroid stimulating hormone (TSH) receptor of the thyroid follicles, stimulating inappropriate T3 and T4 secretion[2]. The second most common cause of hyperthyroidism is thyroid nodular disease[2]. This can be multinodular, leading to swelling or formation of a lump at the front of the neck known as a goitre, which accounts for 15–20% of cases, or a single nodular adenoma, which is found in 5% of hyperthyroid patients[2].
Thyrotoxicosis may arise transiently, without the presence of hyperthyroidism, in response to acute autoimmune inflammation destruction of thyroid follicles, which is known as thyroiditis[7]. In these cases, thyroid hormones briefly rise, owing to the release of stored T3 and T4 when the thyroid follicles are damaged[7]. Thyroiditis is occasionally self-limiting and will spontaneously resolve in less than six months with no significant impact on thyroid function; however, more commonly it will lead to sub-therapeutic thyroid function and hypothyroidism[6].
Hyperthyroidism may also be induced by medication[2]. Amiodarone, an anti-arrhythmic, is known to cause hypo- and hyperthyroidism owing to its structural similarity to iodine; this is known as amiodarone-induced thyrotoxicosis (AIT)[8]. Additionally, patients with hypothyroidism treated with levothyroxine may experience thyrotoxicosis if their dosage is too high[6].
Signs and symptoms
As hyperthyroidism causes an increased metabolic rate, the symptoms of thyrotoxicosis are often secondary to the augmented stimulation of cardiac beta-adrenergic receptors[2]. Therefore, symptoms are widespread across many organ systems (see Table 1).
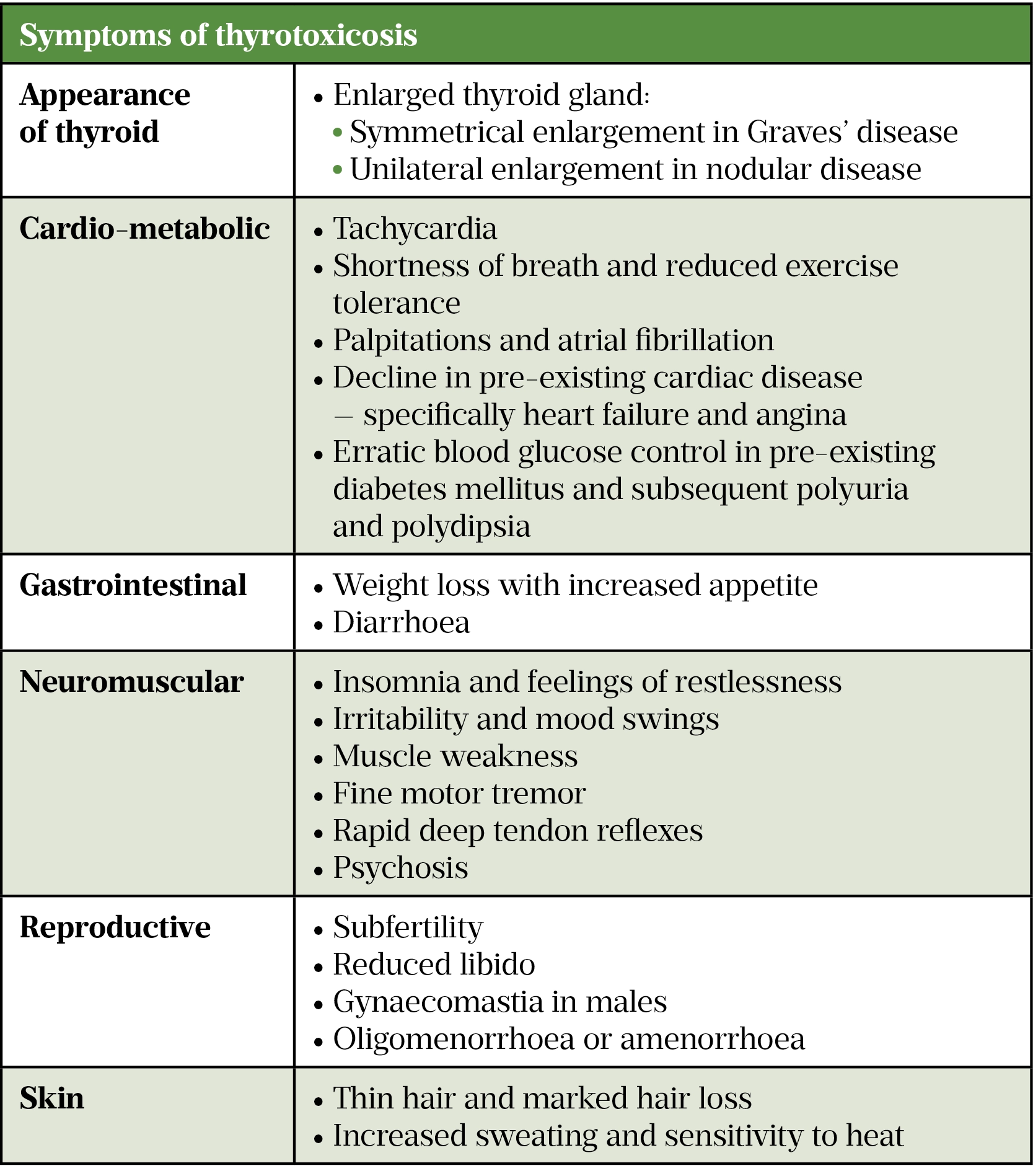
Some symptoms are more specific to the individual cause. Graves’ disease can present with specific symptoms of ophthalmopathy, while symptoms such as hoarse voice or dysphagia may arise owing to compression from an enlarged thyroid, most notable in nodular disease[2,6]. During acute attacks of thyroiditis, patients may experience neck or thyroid-specific pain[6].
Diagnosis
National Institute for Health and Care Excellence (NICE) guidance recommends thyroid function tests (TFTs) for patients presenting with symptoms of thyrotoxicosis, as well as for those with pre-existing autoimmune diseases, such as type 1 diabetes mellitus, or with new onset atrial fibrillation[9].
Initially, only a TSH level is required to screen for thyroid dysfunction, as a TSH value within the reference range is sufficient to rule out thyroid dysfunction; see Table 2 for TFT reference ranges[9]. However, if TSH is outside these values, circulating thyroid hormone levels known as free T3 (FT3) and free T4 (FT4) are required to guide differential further[9].
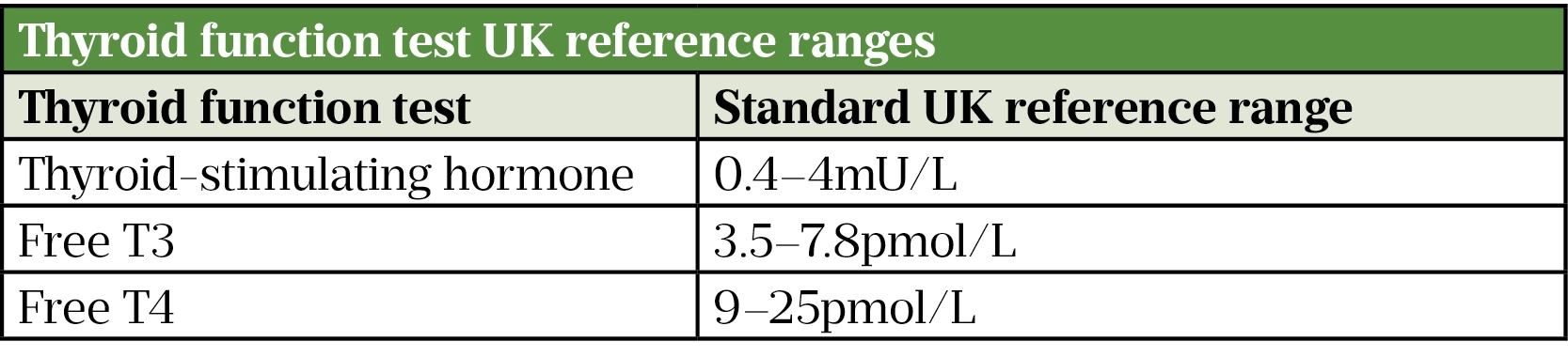
In nearly all variations of thyrotoxicosis, FT3 and FT4 are raised and TSH levels are suppressed owing to the negative feedback loop in response to high circulating thyroid hormone levels; excluding remarkably rare cases of TSH-secreting pituitary adenomas, known as TSHomas, which account for less than 1% of all pituitary adenomas[2,11].
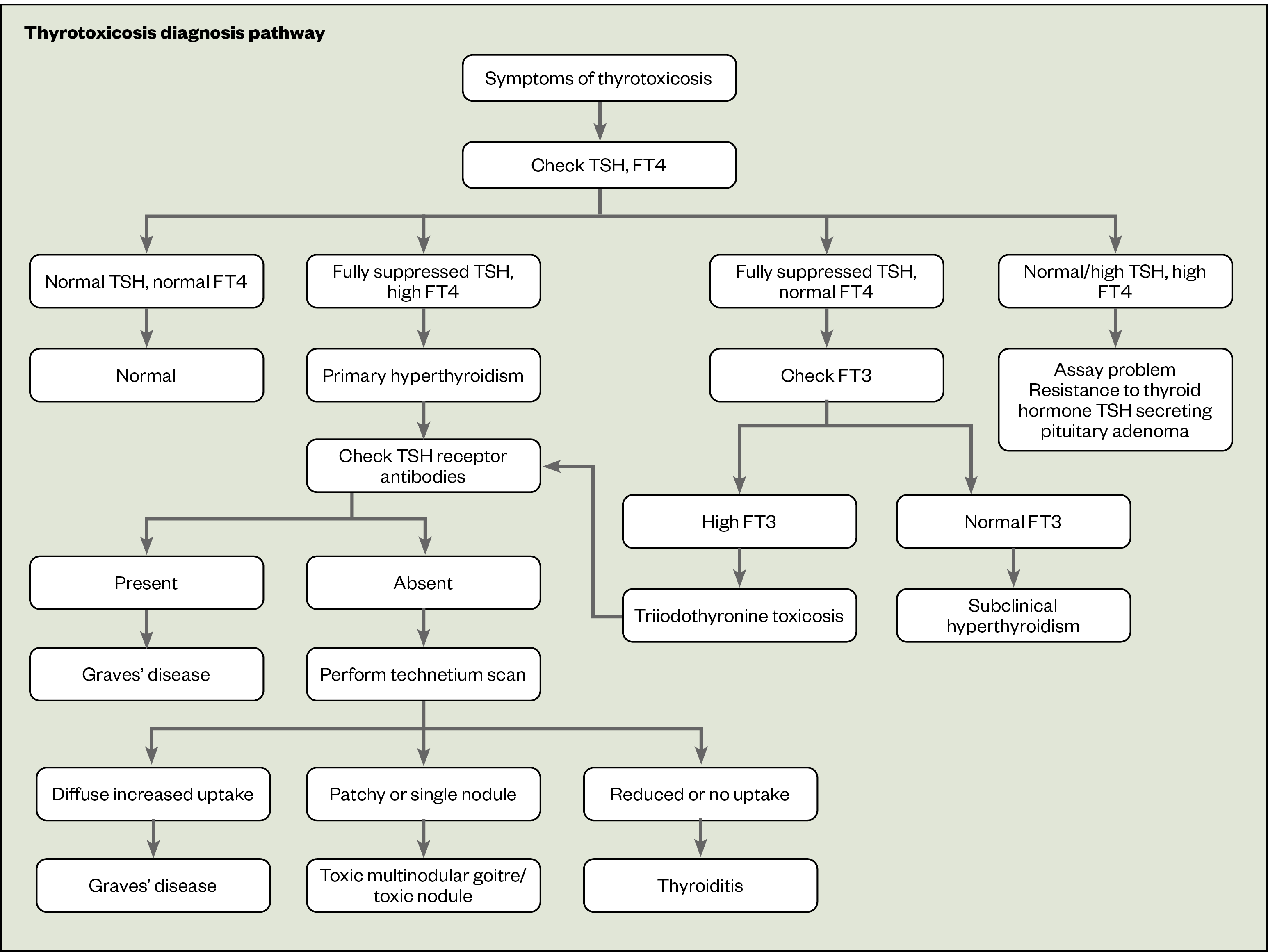
Two sets of TFTs at least six weeks apart are adequate to confirm diagnosis of thyrotoxicosis; TFTs repeated sooner than six weeks are unlikely to provide new clinical information[2,9]. Once thyrotoxicosis is confirmed, cause should be further investigated. Patient history should be taken to identify any medications that are known to impact thyroid function or any recent exposure to radioactive iodine[2]. Testing of TSH receptor antibodies (TRAs) is also recommended by NICE to differentiate between Graves’ and nodular disease, with a positive TRA result indicating Graves’ disease with 95% accuracy[9,12]. In cases where aetiology is still unclear, technetium scanning (whereby a technetium 99m pertechnetate tracer is used as a radioactive label) of the thyroid gland can be performed to distinguish between causes of dysfunction[9].
A summary of the thyrotoxicosis diagnosis pathway is outlined in Figure 1.
Depending on severity at presentation, patients will require either:
- Urgent admission to hospital if there are symptoms of thyroid storm;
- Urgent referral to a secondary care endocrinology specialist if hypothalamic-pituitary pathology suspected;
- Two-week wait referral via suspected cancer pathway if goitre, nodular or structural changes are suggestive of thyroid cancer;
- Routine referral to endocrinology specialists for all other cases.
Treatment
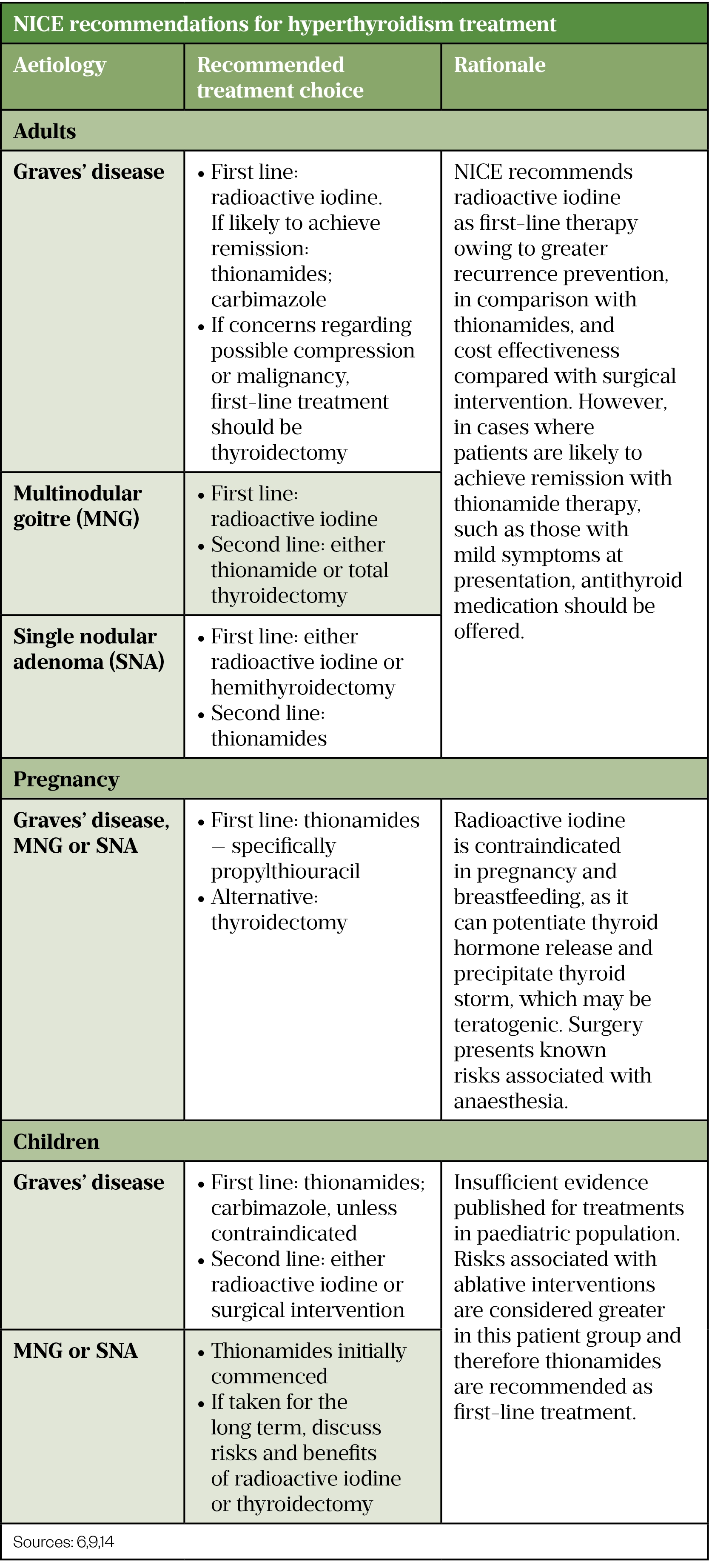
Initial management of thyrotoxicosis consists of symptomatic relief, through counteracting the augmented adrenergic activity with beta-blockers, for which nadolol, metoprolol and propranolol are all licensed, as well as more definitive intervention to treat the underlying cause[13,14]. These interventions can be pharmacological, involving the use of anti-thyroid medications, known as thionamides, or ablative through radioactive iodine or surgery (e.g. thyroidectomy)[9]. Ideally, ablative therapies should not be performed until the patient has become euthyroid (i.e. thyroid hormone levels are within reference range) with thionamide treatment, because of risk of precipitating thyroid storm[2].
Regardless, patients should be fully informed of the risks and benefits of all options so that they can make informed decisions regarding their choice of treatment. A summary of NICE recommendations for hyperthyroidism treatment are included in Table 3.
Antithyroid medications
The oral thionamides, carbimazole and propylthiouracil, are equally effective in treating hyperthyroidism in adults and children[2]. These drugs take up to six to eight weeks to demonstrate a clinical benefit as they prevent thyroid hormone synthesis by inhibiting oxidation of iodine ions required for T3 and T4 production, but do not impact existing T3 and T4 levels[15,16]. Although propylthiouracil inhibits peripheral conversion of T4 to T3, this is rarely clinically significant[17]. Propylthiouracil may also impair radioactive iodine efficacy by increasing thyroid tissue resistance to radioactivity[18]. Carbimazole is the most commonly used first-line treatment, owing to quicker correction of hyperthyroidism and less prominent side effects[17,19].
Initial doses of carbimazole in adults range from between 40–60mg daily, depending on severity of presenting symptoms[2,15]. Levels of TSH, FT3 and FT4 should be checked every six weeks until symptoms have resolved and thyroid hormone levels are within reference range. TSH levels may take four to eight weeks to correct[2]. Once stabilised, patients must then elect for either a ‘titration’ or ‘blocking’ dose regimen:
- Those on a titration regimen have 5–15mg once daily. Despite a short half-life of 4–6 hours, carbimazole exhibits a biological effect up to 40 hours post-dose so is suitable for 24-hourly dosing[15]. Patients require regular TFT monitoring and appropriate dose modification alongside symptom reviews[13];
- Patients on a blocking dose remain on the high initiation dose of 20–60mg to fully suppress endogenous thyroid function and are supplemented with oral levothyroxine to achieve euthyroidism[2]. In the blocking regimen levothyroxine is often given at a dose of 50–150 micrograms once daily for the duration of the treatment[15]. Blocking is thought to be an easier option for patients owing to the reduced need for blood monitoring hospital attendance, with no greater risk of adverse reaction[13].
Patients should be fully informed of the benefits, associated risks and impracticalities or each regimen, such as additional medication required for blocking regimen or frequent hospital attendance on titration regimen, so they can be supported to make an informed decision to fit their lifestyle.
Regardless of regimen, TFTs should be monitored at least once every three months once therapy has stabilised[6].
Carbimazole is contraindicated in patients with severe hepatic impairment, pre-existing blood disorders (e.g. anaemia) and those with a history of pancreatitis. It is not recommended in the first trimester of pregnancy owing to concerns over teratogenicity[15,20].
Propylthiouracil is used for patients who suffer adverse drug reactions with carbimazole, have a history of pancreatitis and in pregnancy[9]. This can be initiated at 200–400mg daily in divided doses, dependent on severity of symptoms on presentation, until euthyroidism is achieved, at which point the dose is gradually reduced to a maintenance dose of 50–150mg daily[14]. Propylthiouracil is only available in 50mg tablets and requires multiple daily dosing, which may mean that patients struggle to comply. Pharmacists should support patients’ concordance through effective counselling on initiation to reinforce the importance of adherence and educate on how to take the medication correctly.
It is important that patients are counselled appropriately on the risks of these medications. Fatal cases of thionamide-induced agranulocytosis have been reported; therefore, prior to commencement of anti-thyroid drugs, a full blood count and liver function tests should be completed[19]. Patients should be educated on the symptoms of blood dyscrasias and advised to immediately report any of these classic signs, such as rashes, mouth ulcers or a new sore throat, for urgent investigation[14]. Additionally, following the observed increased risk of congenital malformations seen with carbimazole, a 2019 Medicines and Healthcare products Regulatory Agency alert recommends the use of effective contraception in all women of childbearing potential taking carbimazole[20]. If a patient is planning pregnancy this should be discussed with their specialist.
Neither propylthiouracil or carbimazole should be used in patients with Lapp lactase deficiency, galactose or glucose-galactose intolerance owing to contents of lactose in formulations[6,15,16]. As with any oral medications that contain lactose as a carrier, caution is required in those with lactose intolerance. While some patients may tolerate this small amount of lactose without remarkable side effects, others may be required to consider ablative therapies.
After 12–18 months of treatment around 50% of patients achieve remission; those who do not should be considered for ablative intervention with either radioactive iodine or surgery[6].
Radioactive iodine
Patients may be concerned by the term ‘radioactive’ when discussing treatment options, but radioactive iodine is safe and effective and has been used to treat thyroid disorders since the 1940s[21]. Self-education via internet searches often lead to patients’ misconceptions about radioactive iodine[22]. This, paired with the scientific nature and complexity of radioactive iodine, can lead to challenging consultations with patients when outlining available options, avoiding use of medical jargon when educating patients to appropriately support their informed decision making[22].
The aim of treatment is to resolve hyperthyroidism without inducing hypothyroidism, but there is no single correct dose to achieve this[2]. Radioactive iodine is readily absorbed and primarily collects within the thyroid gland, where the radiation destroys thyroid tissue and any cancerous cells present within the gland. As a result, almost all patients given radioiodine for Graves’ disease, and up to 50% of those treated for multinodular goitre, will require lifelong levothyroxine following treatment[2]. Additionally, radioactive iodine has been shown to exacerbate Graves’ ophthalmopathy and while the mechanism of this is not well understood, clinicians and patients may not choose this therapy in the presence of thyroid eye disease[23,24]. Risk of exacerbation may be somewhat mitigated through appropriate treatment of ophthalmopathy with corticosteroids and swift thyroxine supplementation should hypothyroidism occur following ablation[2].
Antithyroid drugs should temporarily be ceased one week prior to treatment, to allow adequate absorption of the isotope into the thyroid, and patients should be informed of this by clinicians when opting for this therapy[19]. It will often take two to three months before radioiodine provides clinical benefit, so those with milder forms of hyperthyroidism may not need to recommence their antithyroid drug therapies[18]. Those with severe thyrotoxicosis should restart no sooner than day three post-radioiodine and should have this periodically stopped to assess if still required[19].
Although treatment is safe and effective, the regulations regarding contact post-radiation exposure may concern patients and often leads patients to elect for surgical ablative therapy[2].
Box 1: Guidelines for personal contact following radioiodine exposure
Following a standard 555 MBq dose of radioiodine, patients are considered radioactive for up to six weeks and therefore:
- Patients should not have continuous close contact (within two metres) for periods over an hour with others for 14 days;
- Avoid contact with pregnant women and children for 24 days[2].
Surgery
Surgical intervention for thyrotoxicosis involves removal of part or the entire thyroid gland. This is known as either a hemi- (i.e. near total) or total thyroidectomy, depending on the amount of tissue resected, with some patients requiring removal of the isthmus (the thyroid tissue connecting each lobe)[25]. A thyroidectomy is indicated for large goitres causing compression which results in severe symptoms of dysphagia or breathing difficulties[26]. Additionally, thyroidectomy can be utilised for patients who do not respond to thionamides or who have declined radioiodine therapy[9].
Ideally, patients’ thyroid hormones will be controlled pre-operatively by thionamides; however, in refractory cases or those who experience adverse reactions or intolerance to thionamides, alternative therapies may sometimes be used to control pre-operative hormone levels and reduce peri-operative adverse events (e.g. induction of thyroid storm)[27].
Lithium, a medication used to treat bipolar disorder, has been shown to inhibit thyroid hormone release[28,29]. When used to treat bipolar disorder, this effect can adversely induce hypothyroidism; however, in those with thyrotoxicosis it offers a suitable alternative for essential pre-operative thyroid hormone control[27]. It is prescribed at a dose of 800–1,200mg daily in two divided doses, with appropriate therapeutic drug monitoring[27]. Beta-blockers are also administered to ensure heart rate remains below 80bpm; if heart rate is not adequately controlled, it can contribute to precipitation of a thyroid storm[2].
Complications
There are a number of common complications of uncontrolled hyperthyroidism, including:
- Atrial fibrillation, which occurs in 15% of patients;
- Associated congestive cardiac failure resulting from augmented cardiac output;
- Tachycardia and enhanced left ventricular function;
- Osteoporosis second to over-stimulation of osteoblast turnover;
- Impacts of compression from a large goitre[30–32].
Graves’ ophthalmopathy
A characteristic feature of Graves’ disease, Graves’ ophthalmopathy occurs in around 25 to 50% of cases; see Figure 2[23,33,34]. While often mild, causing merely unpleasant ‘grittiness’, it can be severe and progressive in around 2–5% of cases, resulting in subsequent difficulties, such as protrusion of the eye ball known as proptosis, conjunctival oedema, eye muscle weakness known as ophthalmoplegia or even optic nerve compression, leading to irreparable damage or blindeness[2,34]. Treatment is initially high-dose prednisolone, but those who fail to respond may require surgical decompression[2,23].
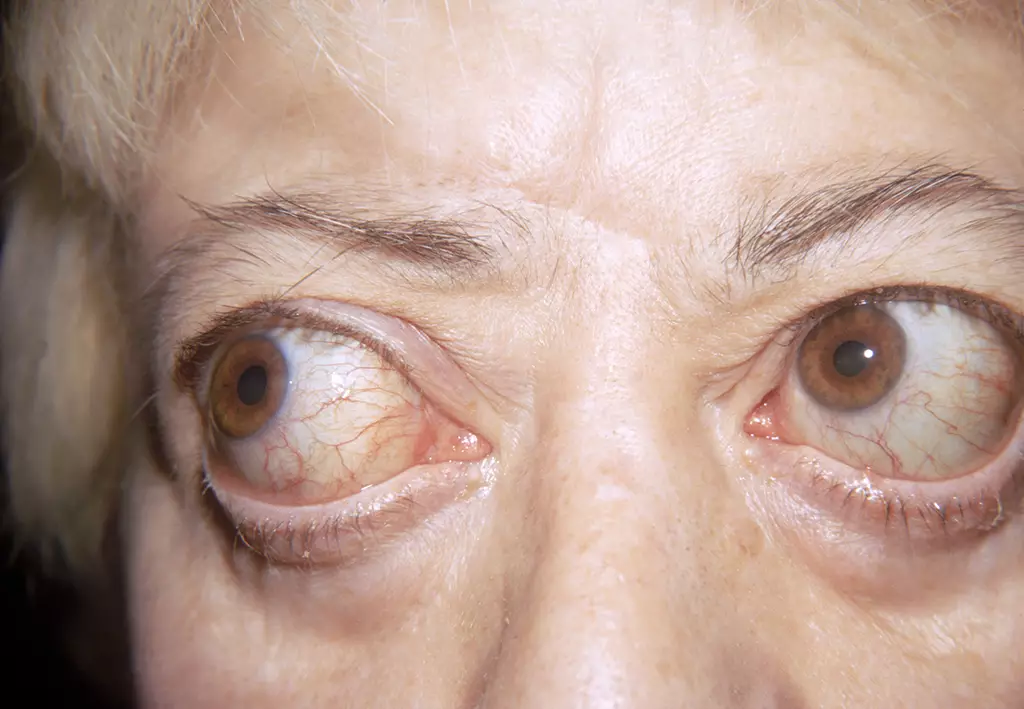
RALPH C. EAGLE, JR.’/SCIENCE PHOTO LIBRARY
Thyroid storm
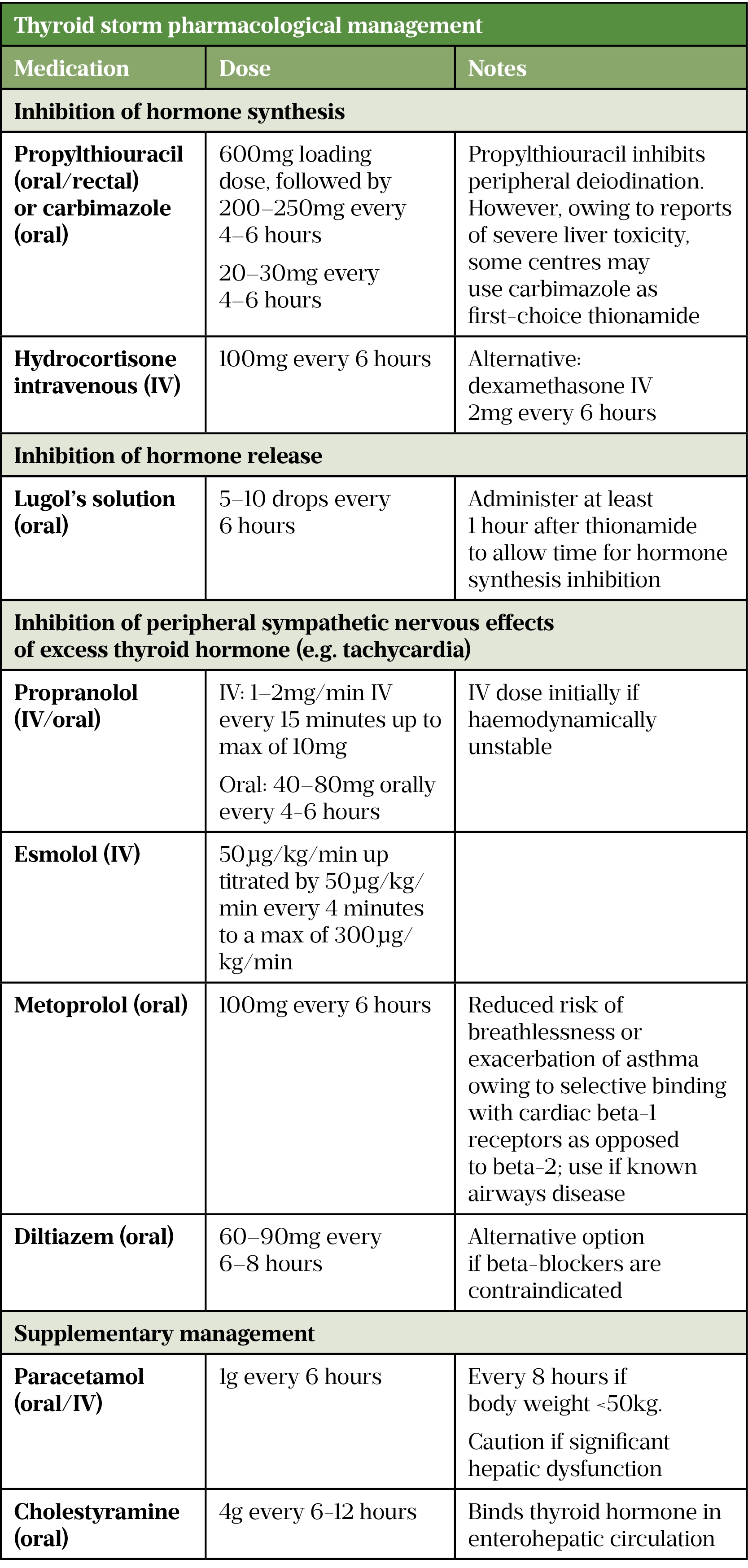
A potentially fatal complication of thyrotoxicosis is a thyroid storm, also known as thyroid crisis[13]. It can be triggered in patients with or without pre-existing hyperthyroidism by infection, trauma, pregnancy, surgery or in those who suddenly stop their antithyroid medication[6]. It presents with tachycardia, muscle weakness, hyperthermia, sweating, vomiting and severe agitation, which can precipitate episodes of psychosis[2,35]. Management requires a multifactorial approach and is summarised in Table 4[36,37].
Role of the pharmacist
Pharmacists’ can support patients’ management of hyperthyroidism. Those experienced with thyroid disorders may provide direct patient care through provision of thyroid clinics, directly assessing and managing patients as part of the multidisciplinary team.
Pharmacists in the community sector should look to provide symptom identification, signposting, monitoring advice and counselling; the latter of which is vital for those on antithyroid medications. Appropriate patient education on symptoms of blood disorders with carbimazole, complex management in pregnancy and regimen concordance support are essential to safeguard patients and facilitate adequate self-care[6].
This article has been reviewed by the expert authors to ensure it remains relevant and up to date, following its original publication in The Pharmaceutical Journal in September 2021.
- 1Betts G, Young K, Wise J, et al. The Endocrine System: The Thyroid Gland . In: Anatomy and Physiology. Housten: : Rice University 2017. N/A.https://openstax.org/details/books/anatomy-and-physiology (accessed Sep 2021).
- 2Kalhan A, Page M. Thyroid and parathyroid disorders. In: Whittlesea C, Hodson K, eds. Clinical Pharmacy and Therapeutics. London: : Churchill Livingstone 2018. 744–61.
- 3De Leo S, Lee SY, Braverman LE. Hyperthyroidism. The Lancet 2016;388:906–18. doi:10.1016/s0140-6736(16)00278-6
- 4Vanderpump MPJ, Tunbrldge WMG, French JM, et al. The incidence of thyroid disorders in the community: a twenty-year follow-up of the Whickham Survey. Clin Endocrinol 1995;43:55–68. doi:10.1111/j.1365-2265.1995.tb01894.x
- 5Samuels M. Hyperthyroidism in Aging. In: Feingold K, Anawalt B, Boyce A, eds. Endotext [ebook]. South Dartmouth 2018. N/A.https://www.ncbi.nlm.nih.gov/books/NBK278986/ (accessed Sep 2021).
- 6Clinical Knowledge Summaries: Hyperthyroidism. National Institute for Health and Care Excellence. 2021.https://cks.nice.org.uk/topics/hyperthyroidism/ (accessed Sep 2021).
- 7Thyroiditis. British Thyroid Foundation. 2018.https://www.btf-thyroid.org/thyroiditis (accessed Sep 2021).
- 8Narayana SK, Woods DR, Boos CJ. Management of amiodarone-related thyroid problems. Therapeutic Advances in Endocrinology 2011;2:115–26. doi:10.1177/2042018811398516
- 9Thyroid disease: assessment and management: NICE guideline [NG145]. . National Institute for Health and Care Excellence . 2019.https://www.nice.org.uk/guidance/ng145 (accessed Sep 2021).
- 10Thyroid function tests. British Thyroid Foundation. 2021. https://www.btf-thyroid.org/thyroid-function-tests (accessed Sep 2021).
- 11Tjörnstrand A, Nyström HF. DIAGNOSIS OF ENDOCRINE DISEASE: Diagnostic approach to TSH-producing pituitary adenoma. European Journal of Endocrinology 2017;177:R183–97. doi:10.1530/eje-16-1029
- 12Graves’ diseas. BMJ Best Practice. 2020.https://www.british-thyroid-association.org/sandbox/bta2016/bta_statement_on_the_management_of_primary_hypothyroidism.pdf (accessed Sep 2021).
- 13Vaidya B, Pearce SHS. Diagnosis and management of thyrotoxicosis. BMJ 2014;349:g5128–g5128. doi:10.1136/bmj.g5128
- 14British National Formulary (online). Joint Formulary Committee. 2021.https://www.medicinescomplete.com (accessed Sep 2021).
- 15Carbimazole 5mg Tablets Summary of Product Characteristics. Accord UK TLD. 2020.https://www.medicines.org.uk/emc/product/10283/smpc (accessed Sep 2021).
- 16Propylthiouracil 50mg Tablets Summary of Product Characteristics. Wockhardt UK LTD. 2020.https://www.medicines.org.uk/emc/product/3123/smpc (accessed Sep 2021).
- 17Cooper DS. Antithyroid Drugs. N Engl J Med 2005;352:905–17. doi:10.1056/nejmra042972
- 18Wong KK, Shulkin BL, Gross MD, et al. Efficacy of radioactive iodine treatment of graves’ hyperthyroidism using a single calculated 131I dose. Clin Diabetes Endocrinol 2018;4. doi:10.1186/s40842-018-0071-6
- 19Thionamides. In: Meyler’s Side Effects of Drugs. Elsevier 2016. 874–89. doi:10.1016/b978-0-444-53717-1.01537-7
- 20Carbimazole: Increased risk of congenital malformations; strengthened advice on contraception. Medicines and Healthcare Products Regulatory Agency. 2019.https://www.gov.uk/drug-safety-update/carbimazole-increased-risk-of-congenital-malformations-strengthened-advice-on-contraception#new-advice-on-contraception-and-pregnancy (accessed Sep 2021).
- 21Treatment of an overactive or enlarged thyroid gland with radioactive iodine. British Thyroid Foundation. 2019.https://www.btf-thyroid.org/treatment-of-an-over-active-or-enlarged-thyroid-gland-with-radioactive-iodine (accessed Sep 2021).
- 22Rosenthal M. Patient misconceptions and ethical challenges in radioactive iodine scanning and therapy. J Nucl Med Technol 2006;34:143–50; quiz 151–2.https://www.ncbi.nlm.nih.gov/pubmed/16951283
- 23Walsh JP, Dayan CM, Potts MJ. Radioiodine and thyroid eye disease. BMJ 1999;319:68–9. doi:10.1136/bmj.319.7202.68
- 24Bartalena L, Marcocci C, Bogazzi F, et al. Relation between Therapy for Hyperthyroidism and the Course of Graves’ Ophthalmopathy. N Engl J Med 1998;338:73–8. doi:10.1056/nejm199801083380201
- 25British Thyroid Foundation. Thyroid surgery. 2018.https://www.btf-thyroid.org/thyroid-surgery (accessed Sep 2021).
- 26Banks CA, Ayers CM, Hornig JD, et al. Thyroid disease and compressive symptoms. The Laryngoscope 2011;122:13–6. doi:10.1002/lary.22366
- 27Nair G, C. Babu M, Menon R, et al. Preoperative preparation of hyperthyroidism for thyroidectomy – Role of supersaturated iodine and lithium carbonate. Indian J Endocr Metab 2018;22:392. doi:10.4103/ijem.ijem_3_18
- 28LAZARUS JH. The Effects of Lithium Therapy on Thyroid and Thyrotropin-Releasing Hormone. Thyroid 1998;8:909–13. doi:10.1089/thy.1998.8.909
- 29Kibirige D, Luzinda K, Ssekitoleko R. Spectrum of lithium induced thyroid abnormalities: a current perspective. Thyroid Research 2013;6:3. doi:10.1186/1756-6614-6-3
- 30Bielecka-Dabrowa A, Mikhailidis DP, Rysz J, et al. The mechanisms of atrial fibrillation in hyperthyroidism. Thyroid Res 2009;2. doi:10.1186/1756-6614-2-4
- 31Fazio S. Effects of Thyroid Hormone on the Cardiovascular System. Recent Progress in Hormone Research 2004;59:31–50. doi:10.1210/rp.59.1.31
- 32Thyroid disease and osteoporosis. British Thyroid Foundation. 2020.https://www.btf-thyroid.org/thyroid-disease-and-osteoporosis (accessed Sep 2021).
- 33Hiromatsu Y, Eguchi H, Tani J, et al. Graves’ Ophthalmopathy: Epidemiology and Natural History. Intern Med 2014;53:353–60. doi:10.2169/internalmedicine.53.1518
- 34Wiersinga WM, Bartalena L. Epidemiology and Prevention of Graves’ Ophthalmopathy. Thyroid 2002;12:855–60. doi:10.1089/105072502761016476
- 35Bathgate G, Karra E, Khoo B. New diagnosis of hyperthyroidism in primary care. BMJ 2018;:k2880. doi:10.1136/bmj.k2880
- 36Carroll R, Matfin G. Review: Endocrine and metabolic emergencies: thyroid storm. Therapeutic Advances in Endocrinology 2010;1:139–45. doi:10.1177/2042018810382481
- 37Bahn RS, Burch HB, Cooper DS, et al. Hyperthyroidism and Other Causes of Thyrotoxicosis: Management Guidelines of the American Thyroid Association and American Association of Clinical Endocrinologists. Endocrine Practice 2011;17:456–520. doi:10.4158/ep.17.3.456