www.sciencephoto.com
After reading this CPD article, adapted from Pharmacy Management of Long-term Medical Conditions, published by Pharmaceutical Press, you should be able to:
- Identify when signs and symptoms are indicative of a patient’s asthma not being well-controlled;
- Assess patients and undertake an asthma review;
- Understand how to effectively manage patients, including the process of stepping up treatment and related considerations.
Asthma is a common chronic condition that affects around 5.4 million people in the UK, including 1.1 million children, and 1 in 5 of these people will suffer from severe asthma[1]. It contributes to a large burden on the NHS, including 77,124 UK hospital admissions in 2016/2017, 75% of which were thought to be avoidable, and 1,484 deaths in 2017, two-thirds of which were thought to be preventable[1–3].
Asthma is a reversible obstruction of the airways, characterised by intermittent inflammation that causes narrowing of the airways and limits airflow. Asthma also causes damage to the nerve endings in the airways, making them hypersensitive and easily irritated[4]. People with asthma typically experience periods of chest tightness, shortness of breath and wheeze in between periods of normal lung function[4].
Asthma attacks may occur at random with no obvious precipitating factors or may be caused by poor adherence to treatment, or a trigger such as exposure to allergens (i.e. house dust mites, animal fur, pollen), tobacco smoke, pollution, cold air, exercise or viruses such as cold or influenza[4].
Asthma is a life-long condition that, if treated appropriately, can be controlled to achieve a resolution of asthma symptoms, normal lung function and no limitation of normal activities. However, some patients may experience no relief or easing of symptoms with their current medicines and need their treatment escalated or ‘stepped up’.
Two guidelines are used in the UK: the British Thoracic Society and Scottish Intercollegiate Guidelines Network (BTS/SIGN) and National Institute for Health and Care Excellence (NICE) guidelines. While there are many similarities, there are also differences in stepwise treatment recommendations[4,5]. This article will mainly consider the BTS/SIGN guideline[4], as it is the more established UK guideline, and will outline how pharmacists can review and manage these patients and adjust their regimen to control their symptoms.

Pharmacy Management of Long-term Medical Conditions is designed to support primary care pharmacists working in community and GP practice pharmacy to improve health outcomes and quality of life for people with long-term medical conditions by ensuring they get the best out of their medicines.
Symptoms
Many patients with asthma will experience an exacerbation of their symptoms for a short period of time (e.g. because of a viral infection or owing to stress)[4]. However, around 17% of patients with asthma have difficulty breathing almost all of the time and often have what could be life-threatening asthma attacks[5].
Signs and symptoms that a patient’s asthma is not well-controlled may overlap with those experienced during an attack, and include:
- Chest tightness/wheeze;
- Increased night-time waking with wheeze cough;
- Inability to speak in full sentences;
- Reduced ability to carry out usual daily activities;
- Increased use of reliever inhaler;
- Reduced peak expiratory flow (PEF), especially if reduced below 75% of best;
- Raised fractional exhaled nitric oxide (FeNO) above 30 parts per billion, suggesting poor adherence or response to inhaled corticosteroids (ICS)[4].
Reasons that patients may have asthma that is hard to control include:
- Having other long-term conditions (e.g. a heart condition or diabetes) that make it harder to control asthma;
- Being a smoker;
- Not being able to get into a good routine of taking their inhalers and other asthma medicines regularly;
- Poor or sub-optimal inhaler technique[6].
It is important that patients experiencing any of the symptoms above seek help as soon as possible and do not wait until their next review for a consultation with a healthcare professional.
Assessment
Asthma is best monitored by routine clinical review on at least an annual basis by a healthcare professional with appropriate training in asthma management in primary and/or secondary care. GP pharmacists can undertake annual asthma reviews to optimise the patient’s asthma treatment, ensure that the patient has an up-to-date asthma treatment plan and escalate patients with difficult or severe asthma. Within the outpatient setting, patients may be reviewed by specialist asthma pharmacists, who may alter their asthma prescription and escalate treatment for those with severe or ‘uncontrolled’ asthma. The suggested components of a review are listed in Table 1 and summarised below.
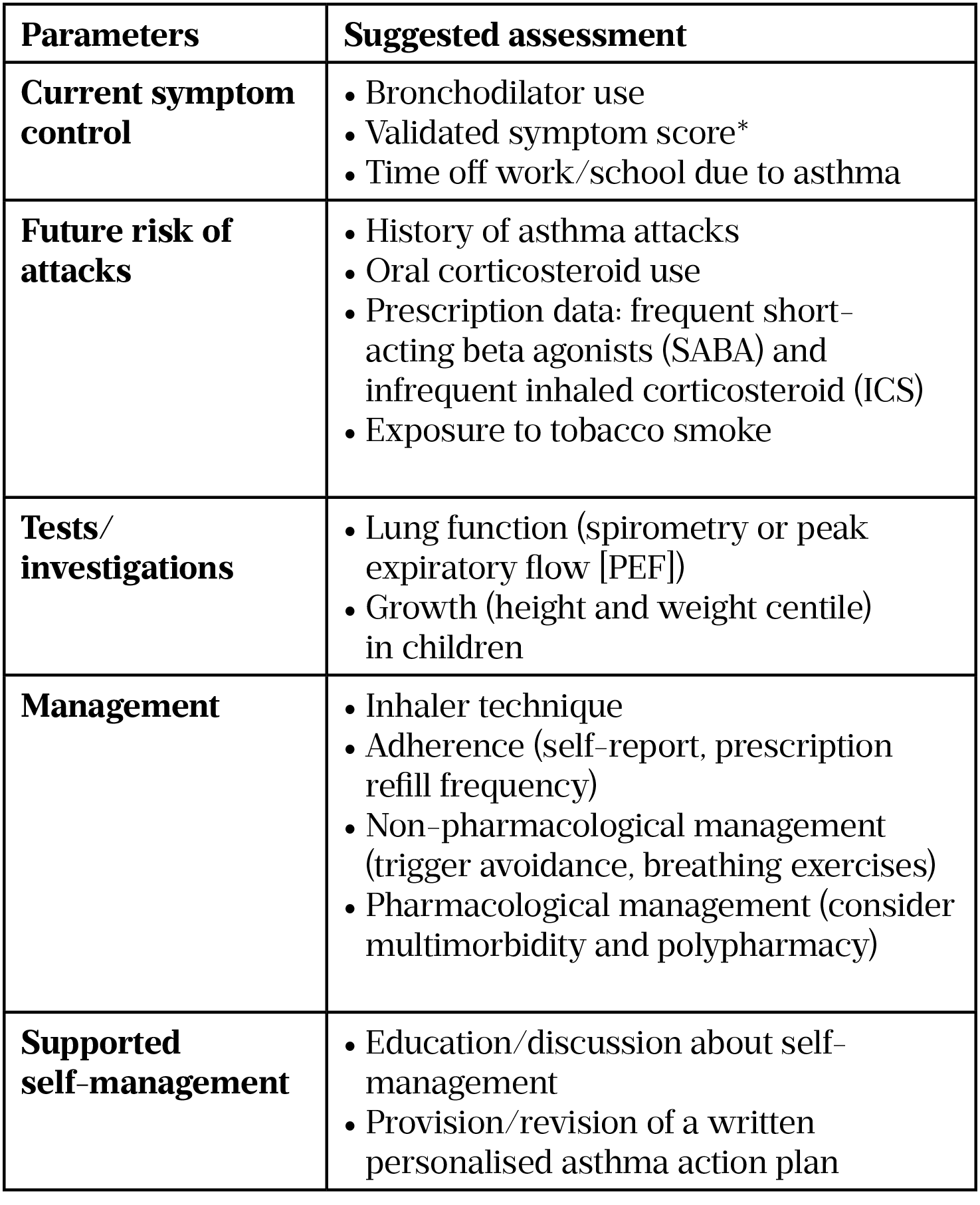
(adapted from BTS/SIGN asthma guidelines, 2019[4])
* Use of the Royal College of Physicians’ ‘3 Questions’ is likely to yield useful information about current control. Symptomatic asthma control is best measured using validated questionnaires (e.g. the Asthma Control Questionnaire, Asthma Control Test or Childhood Asthma Control Test)[4,7–11].
Pharmacists should determine how well patients can control their asthma using specific questions, such as the Royal College of Physicians’ (RCP) ‘3 Questions’:
In the past week/month:
- Have you had difficulty sleeping because of your asthma symptoms (including cough)?
- Have you had your usual asthma symptoms during the day (cough, wheeze, chest tightness or breathlessness)?
- Has your asthma interfered with your usual activities (e.g. housework, work/school etc.)[7]?
If the patient answers yes to any question, further assessment should be made[4]. Symptomatic asthma control is best measured using validated questionnaires (e.g. the Asthma Control Questionnaire, Asthma Control Test or Childhood Asthma Control Test)[9–11].
Pharmacists should also ask the patient about adherence to their preventer inhaler, use of reliever inhaler (whether this has increased in recent days or weeks), how many courses of oral corticosteroids they have required, and use of a PEF meter[4].
During an asthma consultation, inhaler technique should be assessed and optimised using either the patient’s inhalers or placebo devices. If necessary, their inspiratory effort should be checked (e.g. with an In-Check DIAL G16 inspiratory flow meter) to ensure that they can inhale through their prescribed inhaler device at the correct speed. PEF can also be measured to determine or compare with their best PEF as a measure of asthma control and allow the severity of any asthma attack to be determined[4].
Patients should be advised on important non-pharmacological strategies to aid symptom control and reduce the risk of asthma attacks, including smoking cessation advice, breathing exercises and trigger avoidance strategies[4].
Excessive usage of short-acting beta agonists (SABA) inhalers in asthma (more than one canister each month) is associated with a three-fold increased risk of attendance at emergency departments and hospitalisation[12]. Good asthma control is characterised by using a SABA reliever inhaler no more than twice a week, so a 200-dose salbutamol pressurised metered dose inhaler (pMDI) should be expected to last 50 weeks[4].
Management
The aim of asthma management is to control the disease. Complete control of asthma is defined as:
- No daytime symptoms;
- No night-time waking;
- No need for rescue medication;
- No asthma attacks;
- No limitations on activity, including exercise;
- Normal lung function (in practical terms forced expiratory volume [FEV1] and/or PEF>80% predicted or best);
- Minimal adverse effects from medication[4].
General principles
Before initiating a new drug in patients who have uncontrolled asthma, it is important their adherence with existing therapies is confirmed, inhaler technique is checked and any trigger factors eliminated (see Assessment).
Patients should only be prescribed inhalers after they have received training in the use of the device and have demonstrated satisfactory technique[4]. It is good practice to only prescribe inhalers by brand name to ensure that patients are dispensed familiar inhaler devices that they can use properly[4].
In children, a pMDI and spacer is the preferred method of delivering a SABA and ICS, as young children may be unable to produce the inspiratory effort required to use higher resistance dry powder inhalers. A face mask is required until the child can breathe reproducibly using the spacer mouthpiece[4].
However, in some cases, a patient’s current medicines may not provide relief or easing of their symptoms and their treatment will need to be escalated or ‘stepped up’.
Initial add-on therapy
In adult patients whose asthma is uncontrolled on low dose, or children on very low dose ICS, options for ‘stepping up’ treatment include either increasing the ICS dose or the addition of a long-acting beta-2 agonist (LABA) or a leukotriene receptor antagonist (LTRA)[4,5].
Compared with increasing the ICS dose, the addition of a LABA achieves a similar reduction in the risk of exacerbations, but produces greater improvements in lung function, with an increased number of symptom-free days and a lower requirement for rescue SABA therapy[13,14]. The addition of LTRA to low dose ICS is effective in preventing asthma attacks and has a similar effect on asthma control as high dose ICS as single therapy[14].
Recommendations in the current BTS/SIGN and NICE asthma guidelines differ in that the former recommends using LABA as initial add-on therapy, while the latter recommends LTRA[4,5]. While both guidelines agree that LABA add-on therapy produces small but greater improvement in morning and evening PEF and quality of life, a significant reduction in rescue medicine use and an increase in the proportion of symptom-free days, NICE recommends LTRA because the low price of generic montelukast means that LTRA is more cost-effective than LABA[5,15].
When a LABA is used in asthma, it should be prescribed in a combination ICS/LABA inhaler to ensure adherence to both drugs and to prevent patients from only using their LABA inhaler and not their ICS. LABA monotherapy in asthma has been associated with an increase in asthma deaths, but this is not seen when used in combination with an ICS. Otherwise, LABA drugs are well tolerated, with the most common adverse effects including tremor, palpitations and muscle cramps[16].
LTRAs, such as montelukast, are generally well tolerated, with the most common adverse effects including headache and sleep disturbance with vivid dreams[17]. If the patient develops any of these adverse effects, they should be trialled off the leukotriene receptor to assess whether the symptoms resolve[7]. Recently, the Medicines and Healthcare products Regulatory Agency highlighted the risks of neuropsychiatric reactions with montelukast, and patients should be advised to stop treatment and seek medical advice immediately should they occur[7].
Where asthma remains uncontrolled despite low dose ICS with LABA, it is essential to confirm the diagnosis and to check adherence and inhaler technique before stepping up treatment.
Options for increasing treatment include increasing the ICS dose in adults from low dose to medium dose (or in children from very low dose to low dose) or to add in an LTRA[4]. Table 2 provides more information on low, medium and high doses of combination inhalers.

(adapted from BTS/SIGN Asthma Guidelines, 2019[4])
Single combination inhaler for maintenance and reliever therapy (MART)
The use of a single ICS/LABA combination inhaler for maintenance and reliever therapy (MART) is an alternative approach to using fixed-dose ICS/LABA inhaler in addition to a separate SABA reliever inhaler[4]. At present, MART regimen is only licensed for use with budesonide/formoterol or beclomethasone/formoterol inhalers[4].
In this regimen, the ICS/LABA inhaler is used regularly once or twice per day, with an extra dose used as necessary to provide fast relief of symptoms owing to the rapid onset of reliever effect of the LABA formoterol[4]. This additional PRN (as required) dose gives the patient an extra dose of ICS to reduce any early airway inflammation. This ensures that, as the need for a reliever increases, the dose of preventer medication is also increased, with the aim of increasing overall asthma treatment early in deteriorating asthma control and preventing a full asthma attack[4].
It is important that this MART regimen is underpinned by a self-management plan to enable patients to self-monitor their asthma symptoms and understand when it is appropriate to use an extra dose of their ICS/LABA inhaler[4]. There is evidence that by using this regime, there may be a reduction in the overall steroid dose needed to treat an exacerbation[18]. If patients are switched from regular fixed-dose ICS/LABA to a MART regimen, the total regular dose of daily ICS should not be decreased. Patients taking rescue doses of their combination inhaler once per day or more regularly should have their treatment reviewed, as this suggests poor asthma control[4]. Careful education of patients about the specific issues around this management strategy is required to ensure the appropriate use of their inhaler and avoid overuse[4].
Specialist therapies
It should be expected that only a small proportion of patients will continue to have sub-optimal control on low to medium dose ICS with LABA and or LTRA (or very low to low dose ICS in children). There are very few clinical trials in this specific patient group to guide management, and patients should therefore be referred to specialist asthma services[4].
Treatment options in these patients include increasing ICS dose in adults to high doses (see Table 2), or sequential trials of additional controller medicines including the anticholinergic tiotropium, phosphodiesterase inhibitor, theophylline or an LTRA if not already trialled[4].
In adults with severe asthma, the addition of tiotropium to high dose ICS/LABA may have additional benefits in reducing the need for rescue oral corticosteroids, and small improvements in lung function and asthma control[9]. The addition of theophylline may have some beneficial effects but is often limited by significant adverse effects, including nausea, indigestion, headaches, arrhythmias, and seizures requiring 6–12-monthly therapeutic drug monitoring[4].
Continuous oral corticosteroids are rarely recommended, but may be required under specialist care in patients with confirmed asthma who remain uncontrolled despite adequate trials of high dose ICS and all other add-on therapies. Such patients should be monitored closely for systemic adverse effects, including monitoring blood pressure, blood sugar and lipids, bone mineral density, growth in children, cataracts and glaucoma[4].
Patients who are adherent to optimal top step therapy but remain poorly controlled, requiring repeated courses of oral steroids and/or hospital admissions, can be classified as having severe asthma[4]. These patients require referral to a severe asthma centre for review, and may be eligible for monoclonal antibody treatment.
Treatment options include omalizumab as add-on anti-IgE therapy for severe allergic asthma; mepolizumab, reslizumab and benralizumab as add-on anti-IL5 or anti-IL5 receptor therapy for severe eosinophilic asthma; and dupilumab as add-on anti-IL4/anti-IL13 therapy for severe eosinophilic asthma[4].
Patients should be reviewed regularly and, once asthma control has been achieved, consideration should be made to step down treatment to use the lowest effective doses to avoid adverse effects. Stepping down treatment is recommended every three weeks in patients whose asthma is stable. Which drug is reduced or stopped at each review depends on patient preference, response, adverse effects and severity of asthma. When the ICS dose is reduced, it should be done so by 25% to 50% at each step[4].
ICS exhibits a dose–response curve for clinical efficacy, but this starts to plateau at around 100–200 micrograms per day and peaks by 500 micrograms per day[19]. This suggests the majority of adults with asthma would be unlikely to achieve any significant clinical benefits from increasing ICS doses above medium doses. Consequently, the use of high doses in adults (see Tables 2 and 3) should be reserved for specialist care only[4].

(adapted from BTS/SIGN asthma guidelines, 2019[4])
It is important to note that the efficacy of ICS may be reduced by current or past smoking. This may be overcome by increasing the dose. However, the most effective therapy is smoking cessation, and all patients should be offered advice and support[20].
In primary care, pharmacists should closely monitor severe asthmatics, particularly to ensure continued adherence to their preventative therapies. Good communication between primary care, community pharmacists and the severe asthma service is essential to ensure patients remain on the correct therapies and that any changes in adherence or control (increased SABA use or additional courses of oral steroids) are highlighted early to the specialist centre. This will enable the specialist centre to correctly assess the severity of the patient’s asthma. Any change in treatment should be advised by the specialist centre.
Drug interactions
Severe asthma patients are at an increased risk of drug interactions owing to the additional medications they may be taking. The list below is not exhaustive, and any new medications should be checked against the patient’s current treatments for interactions.
- Theophylline and antibiotics — if a patient is on regular oral theophylline and is started on ciprofloxacin or clarithromycin, a reduction in their theophylline dose should be considered, as plasma levels of theophylline can be altered by concomitant use with liver enzyme-inhibiting drugs[21]. Generally, halving the dose is sufficient. The patient’s levels should be closely monitored, and the dose needs to be increased to the previous dose once the course of antibiotics is finished. The patient should be counselled on possible signs of toxicity (e.g. headache, nausea, palpitations).
- Inhaled steroids and antifungals — azoles, such as itraconazole, can increase blood steroid levels following inhaled steroids. A reduction in ICS dose will be required. The dose may need to be halved. It is important to increase the ICS dose if the azole is stopped[22].
Follow-up
Most people with asthma should have an annual asthma review with their GP or healthcare professional to determine their current and recent asthma control, assess their risk of future exacerbations and to optimise (stepping up or down) medication, and reinforce self-management education[4]. Pharmacists can provide this service in primary care.
Patients should also have a follow-up review with their GP within two working days from treatment or discharge from hospital following an asthma attack[4]. This is to ensure that recovery from the acute event is complete; determine the reasons for the asthma attack; and explore strategies to avoid them in the future. Inhaler technique should be re-checked and the importance of maintaining good adherence to treatment reinforced.
Referral
Pharmacists should ensure they are aware of the multidisciplinary team and local services available to help support people with asthma in the community. Physiotherapists can provide education on breathing exercises and dysfunctional breathing reduction techniques, which can improve symptoms and quality of life[4]. Psychological disorders, such as anxiety and depression, are more prevalent in people with asthma and are associated with worse asthma control and increased risk of asthma attacks. Patients should be referred to mental health services for psychological interventions (e.g. cognitive behavioural therapy).
Patients should be referred to specialist asthma services (via the patient’s GP practice) if they have severe asthma requiring treatment with high dose ICS or frequent exacerbations or are at high risk of asthma death. These patients can be assessed by specialist multidisciplinary teams to review their diagnosis, optimise comorbidities and consider treatment with biologic therapies. This can avoid the need for continuous maintenance oral corticosteroids and the risk of associated systemic adverse effects.
Useful organisations and resources
- Asthma UK
- Global Initiative for Asthma
- National Centre for Smoking Cessation Training
- Rightbreathe
- 1Asthma UK. Asthma facts and statistics. Asthma UK. 2017.http://www.asthma.org.uk/asthma-facts-and-statistics (accessed 15 Mar 2021).
- 2Royal College of Physicians. Why asthma still kills: The National Review of Asthma Deaths (NRAD). Royal College of Physicians. 2014.https://www.rcplondon.ac.uk/projects/outputs/why-asthma-still-kills (accessed 15 Mar 2021).
- 3Asthma UK. Time to take action. Asthma UK. 2014.https://www.asthma.org.uk/globalassets/campaigns/compare-your-care-2014.pdf (accessed 15 Mar 2021).
- 4British Thoracic Society and Scottish Intercollegiate Guidelines Network. British guideline on the management of asthma. British Thoracic Society. 2019.https://www.brit-thoracic.org.uk/quality-improvement/guidelines/asthma/ (accessed 15 Mar 2021).
- 5National Institute for Health and Care Excellence . Asthma diagnosis and management. National Institute for Health and Care Excellence. 2017.https://www.nice.org.uk/guidance/ng80 (accessed 15 Mar 2021).
- 6Asthma UK. Difficult to control asthma. Asthma UK. 2019.https://www.asthma.org.uk/advice/understanding-asthma/types/difficult-control-asthma/ (accessed 15 Mar 2021).
- 7Pearson M, Bucknall C. Measuring clinical outcome in asthma: a patient-focused approach. London: : Royal College of Physicians 1999.
- 8Pinnock H, Burton C, Campbell S, et al. Clinical implications of the Royal College of Physicians three questions in routine asthma care: a real-life validation study. Primary Care Respiratory Journal 2012;21:288–94. doi:10.4104/pcrj.2012.00052
- 9Juniper EF, O′byrne PM, Guyatt G h, et al. Development and validation of a questionnaire to measure asthma control. Eur Respir J 1999;14:902. doi:10.1034/j.1399-3003.1999.14d29.x
- 10Nathan RA, Sorkness CA, Kosinski M, et al. Development of the asthma control test☆A survey for assessing asthma control. Journal of Allergy and Clinical Immunology 2004;113:59–65. doi:10.1016/j.jaci.2003.09.008
- 11Liu AH, Zeiger R, Sorkness C, et al. Development and cross-sectional validation of the Childhood Asthma Control Test. Journal of Allergy and Clinical Immunology 2007;119:817–25. doi:10.1016/j.jaci.2006.12.662
- 12Silver HS, Blanchette CM, Kamble S, et al. Quarterly Assessment of Short-Acting β2-Adrenergic Agonist Use as a Predictor of Subsequent Health Care Use for Asthmatic Patients in the United States. Journal of Asthma 2010;47:660–6. doi:10.3109/02770901003702824
- 13Ducharme FM, Ni Chroinin M, Greenstone I, et al. Addition of long-acting beta2-agonists to inhaled steroids versus higher dose inhaled steroids in adults and children with persistent asthma. Cochrane Database of Systematic Reviews Published Online First: 14 April 2010. doi:10.1002/14651858.cd005533.pub2
- 14Joint Formulary Committee. Treatment summary: Asthma, chronic (online). London: : BMJ Group and Pharmaceutical Press 2021. https://bnf.nice.org.uk/treatment-summary/asthma-chronic.html (accessed 15 Mar 2021).
- 15Chauhan BF, Ducharme FM. Addition to inhaled corticosteroids of long-acting beta2-agonists versus anti-leukotrienes for chronic asthma. Cochrane Database of Systematic Reviews Published Online First: 24 January 2014. doi:10.1002/14651858.cd003137.pub5
- 16Morales DR. LABA monotherapy in asthma: an avoidable problem. Br J Gen Pract 2013;63:627–8. doi:10.3399/bjgp13x675250
- 17Calapai G, Casciaro M, Miroddi M, et al. Montelukast-Induced Adverse Drug Reactions: A Review of Case Reports in the Literature. Pharmacology 2014;94:60–70. doi:10.1159/000366164
- 18Sobieraj DM, Weeda ER, Nguyen E, et al. Association of Inhaled Corticosteroids and Long-Acting β-Agonists as Controller and Quick Relief Therapy With Exacerbations and Symptom Control in Persistent Asthma. JAMA 2018;319:1485. doi:10.1001/jama.2018.2769
- 19Holt S, Suder A, Weatherall M, et al. Dose-response relation of inhaled fluticasone propionate in adolescents and adults with asthma: meta-analysis Commentary: Dosage needs systematic and critical. BMJ 2001;323:253–253. doi:10.1136/bmj.323.7307.253
- 20Tomlinson JEM. Efficacy of low and high dose inhaled corticosteroid in smokers versus non-smokers with mild asthma. Thorax 2005;60:282–7. doi:10.1136/thx.2004.033688
- 21National Institute for Health and Care Excellence. Clinical Knowledge Summary. Asthma prescribing information: theophylline. National Institute for Health and Care Excellence. 2020.https://cks.nice.org.uk/topics/asthma/prescribing-information/theophylline/ (accessed 15 Mar 2021).
- 22National Institute for Health and Care Excellence. Clinical Knowledge Summary. Corticosteroids – inhaled. National Institute for Health and Care Excellence. 2020.https://cks.nice.org.uk/topics/corticosteroids-inhaled/management/corticosteroids-inhaled/ (accessed 15 Mar 2021).