Shutterstock.com
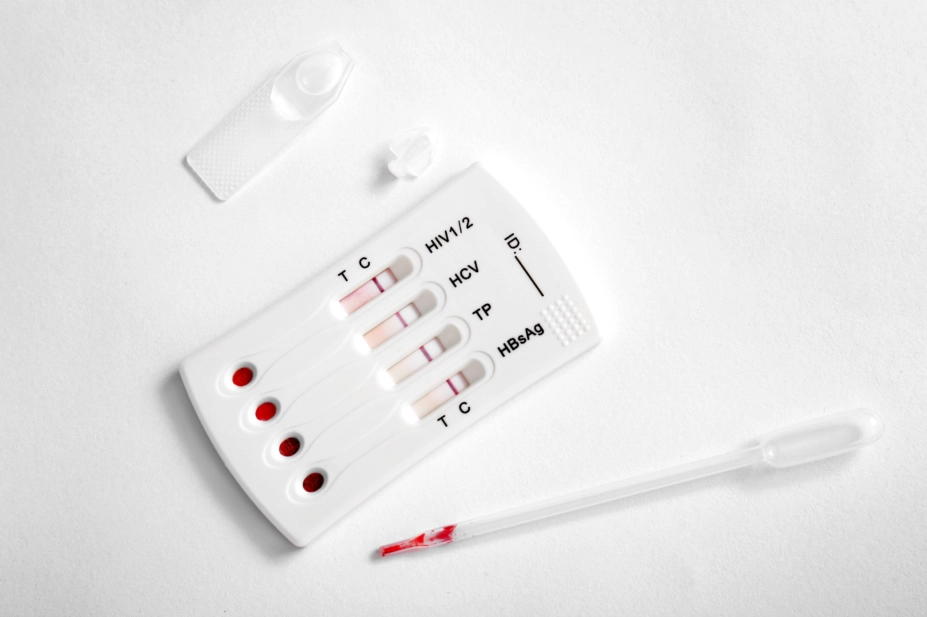
The UK’s drugs safety watchdog has seized nearly 10,000 fake sexually transmitted infection (STI) test kits — including some that screen for HIV — and is warning the public to check packs for the CE quality mark and ensure they are sold by an approved supplier.
All of the products were purchased online in the past three years, the Medicines and Healthcare products Regulatory Agency (MHRA) revealed.
It is also advising the public against using any test kits which arrive in unsealed or damaged packing or have passed their expiry date, because of the increased risk they pose in providing unreliable results.
The warning comes as MHRA research revealed that 25% of people aged 18–30 years have bought medical products online in the past 12 months, despite suspecting that they were counterfeit.
Of the 1,009 young people surveyed, 63% said they had decided to buy an STI home test kit after reading about them on a website, forum or blog; while 64% were attracted to self-test because they were too embarrassed to buy a kit in person.
Graeme Tunbridge, devices group manager for the MHRA, told the public on the 17 October 2018: “Look for the distance selling logo which means that the [online] supplier is registered with the MHRA to sell medicines to the public and has been accredited as a legitimate source. Make sure that the test is easy to use and has clear instructions.”
Michael Brady, medical director of the Terrence Higgins Trust charity, which campaigns on and provides services relating to HIV and sexual health, said that while it welcomed online testing as a means of potentially improving HIV prevention and reducing STIs, “it is extremely worrying to hear about illegal and fake test kits being advertised online … it’s really important for people to know that what they’re purchasing is safe, effective and will reliably diagnose or exclude STIs and HIV”.
The figures and research were released as part of the MHRA’s ongoing #FakeMeds campaign .