Shutterstock.com
Adults who have at least one weight-related comorbidity and a body mass index (BMI) of 35kg/m2 or above could soon be prescribed semaglutide (Wegovy; Novo Nordisk), in line with final guidance from the National Institute for Health and Care Excellence (NICE).
The guidance, published on 8 March 2023, states that the glucagon-like peptide (GLP-1) receptor agonist be prescribed alongside a reduced-calorie diet and increased physical activity through specialist NHS weight management services.
The recommendation is based on evidence from the randomised controlled trial ‘Semaglutide treatment effect in people with obesity’ (STEP) 1, the results of which found that participants taking semaglutide lost an average of 12% more of their body weight compared with placebo.
The evidence also shows that semaglutide is more effective than liraglutide, another GLP-1 receptor agonist, for weight loss and may decrease the risk of cardiovascular disease.
People with a BMI of between 30.0–34.9 kg/m2, with one weight-related comorbidity, such as type 2 diabetes mellitus or hypertension, who are eligible for referral to specialist weight management services could also be prescribed the drug.
A lower BMI threshold has been recommended for people from South Asian, Chinese, other Asian, Middle Eastern, Black African or African-Caribbean family backgrounds.
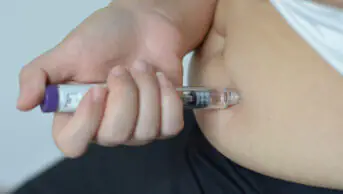
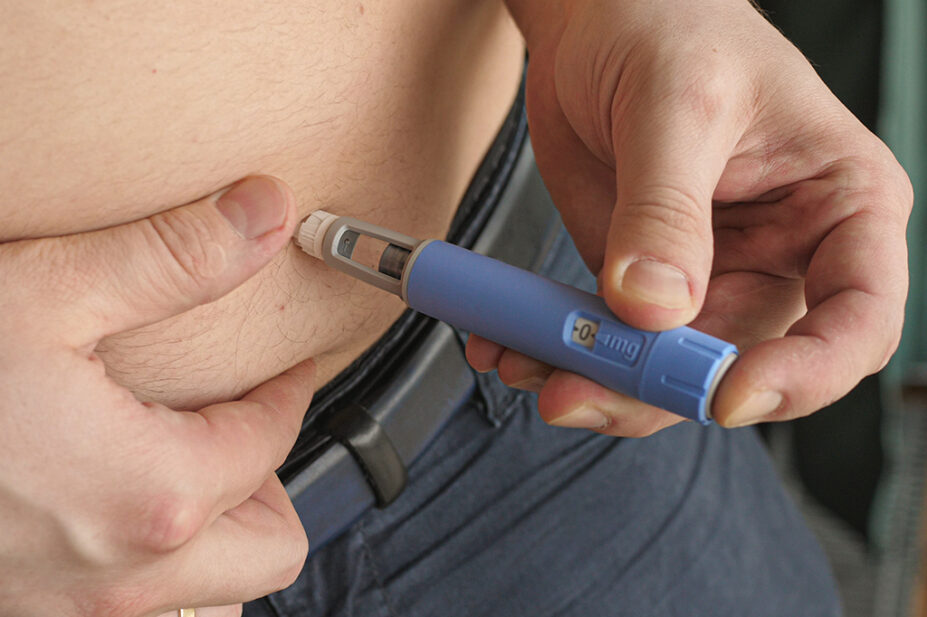
Semaglutide, which is administered via self-injection every week, works by suppressing a person’s appetite by mimicking the hormone GLP-1, which is released after eating. It makes people using it feel full, thereby resulting in people eating less and reducing their overall calorie intake.
Draft guidance for the drug was published in February 2022; however, in a press release published on 8 March 2023, NICE said it had been “unable” to publish final guidance “until today” because the product had not been scheduled to launch in England.
NICE’s statement added that the drug would be available to NHS patients “soon” when the launch of the drug in England was confirmed by its manufacturer.
The guidance says semaglutide can only be prescribed for a maximum of two years within a specialist weight management service, providing multidisciplinary management of overweight or obese patients; this, it says includes, but is not limited to, tier 3 and 4 weight management services.
In a statement dated 8 March 2023, Helen Knight, director of medicines evaluation at NICE, said: “Our committee has made specific recommendations to ensure it remains value for money for the taxpayer, and it can only be used for a maximum of two years.”
Commenting on the guidance, Hannah Beba, consultant diabetes pharmacist for NHS Leeds Clinical Commissioning Group, said she had “some reservations” around the often relapsing nature of obesity and the provision of only a two-year access to the medication.
“I also have concerns about capacity within current systems of care to cope with increased demand once this product is launched,” she added.
“I believe there is no planned aligned funding from NHS England at the moment to help with redesign of weight management services and this is likely to be needed to ensure access meets the demands we expect.”
Beba also highlighted imminent plans to provide the drug through high-street pharmacies and said that this had the potential to “drive inequalities in care”.
“We know that obesity is particularly prevalent in low IMD areas and those most in need may not be able to afford to self-fund such a medication,” she said.
“This is somewhat offset by the convenience of supply, particularly for those that may not engage with specialist healthcare provision. I look forward to working with our community colleagues to discuss benefits, concerns and understand more about how access is proposed.”
Carel le Roux, an obesity physician at University College Dublin, said the NICE committee had done “a very good job” at balancing the best evidence on the health-economic impact of semaglutide and the clinical need to the drug available to the UK population.
“I think the findings of NICE will be replicated in many other jurisdictions because it was balanced and sensible given the high-quality data from randomised controlled trials but also the current high price of the medication.
Responding to the recommendation that the drug can only be prescribed for a maximum of two years, le Roux highlighted that five-year data would soon be available via the ‘Semaglutide effects on heart disease and stroke in patients With overweight or obesity‘ (SELECT) study.